Advanced Synthesis of Benzoyl Acrylic Podophyllotoxin Esters for Scalable Oncology Intermediates
Advanced Synthesis of Benzoyl Acrylic Podophyllotoxin Esters for Scalable Oncology Intermediates
The pharmaceutical industry continuously seeks novel scaffolds that balance potent bioactivity with manageable toxicity profiles, particularly in the realm of oncology therapeutics. Patent CN113185529B introduces a groundbreaking preparation method for benzoyl-containing acrylic acid podophyllotoxin ester derivatives, addressing the critical limitations of native podophyllotoxin. This technology leverages a semi-synthetic approach to modify the C-4 position of the podophyllotoxin backbone, introducing benzoyl acrylic moieties that significantly alter the pharmacological profile. For R&D directors and procurement specialists, this represents a viable pathway to develop next-generation antitumor agents with improved therapeutic indices. The disclosed methodology not only enhances inhibitory activity against resistant cell lines such as HGC-27 and MCF-7 but also drastically reduces off-target toxicity on normal renal epithelial cells. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthetic route is essential for securing a stable supply of high-value oncology precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional utilization of podophyllotoxin as a lead compound has been severely hampered by its inherent non-selective cytotoxicity, which causes severe gastrointestinal responses and damage to normal tissues. Conventional derivatives like etoposide, while clinically successful, still suffer from resistance issues and significant side effects due to their mechanism of action which often lacks specificity for tumor tissue. Furthermore, direct modification of the podophyllotoxin skeleton often requires harsh reaction conditions or complex protection-deprotection strategies that lower overall yield and increase production costs. The lack of selectivity in prior art methods means that achieving a therapeutic window wide enough for clinical safety often requires extensive structural optimization, leading to prolonged development timelines. Additionally, the purification of podophyllotoxin derivatives from complex reaction mixtures can be challenging, often resulting in impurity profiles that fail to meet stringent regulatory standards for API intermediates. These factors collectively create a bottleneck in the cost reduction in API manufacturing, necessitating a more efficient and selective synthetic strategy.
The Novel Approach
The innovative strategy outlined in the patent data involves the introduction of a benzoyl acrylic group at the C-4 position of the podophyllotoxin skeleton through a streamlined esterification process. This structural modification capitalizes on the physiological activities of beta-aryl acrylic acid derivatives, known for their antibacterial and antitumor properties, to create a synergistic effect with the lignan lactone core. By connecting corresponding benzene sulfonamide phenylacetic acid or benzoylacrylic acid derivatives to podophyllotoxin, the new compounds exhibit strong inhibitory activity on tumor cell strains while demonstrating markedly lower toxicity to normal cells. The synthesis avoids excessive steps by utilizing direct coupling methods under mild conditions, thereby preserving the stereochemical integrity of the sensitive podophyllotoxin core. This approach allows for the rapid generation of a diverse library of derivatives, facilitating structure-activity relationship studies to identify lead candidates like Compound 12c. 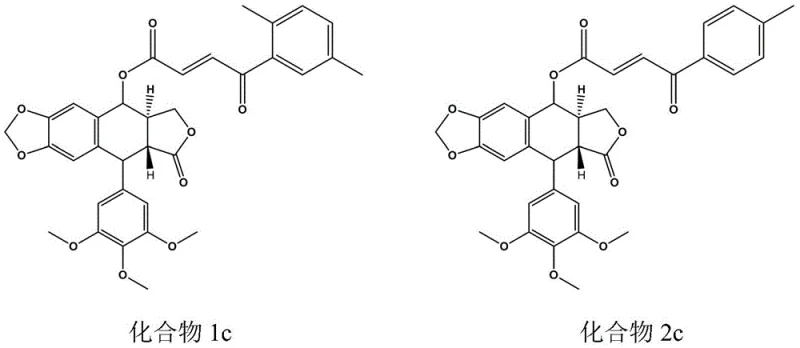
Mechanistic Insights into DCC-Mediated Esterification and Friedel-Crafts Acylation
The core of this synthetic route relies on two key chemical transformations: the preparation of substituted benzoylacrylic acids via Friedel-Crafts acylation and the subsequent esterification with podophyllotoxin. The acid intermediate synthesis utilizes anhydrous aluminum chloride as a Lewis acid catalyst to activate maleic anhydride, which then reacts with substituted xylenes. This electrophilic aromatic substitution proceeds efficiently at room temperature, generating the requisite cinnamic acid derivatives with high regioselectivity. The reaction mixture is carefully quenched with diluted hydrochloric acid to pH 6-7, followed by extraction and drying, ensuring the removal of aluminum salts that could interfere with downstream processing. The subsequent purification involves dissolution in sodium bicarbonate and re-precipitation at pH 2, a classic acid-base workup that effectively removes neutral organic impurities and unreacted starting materials. This meticulous purification of the acid precursor is critical, as any residual impurities could complicate the subsequent coupling reaction or introduce difficult-to-remove contaminants in the final API intermediate.
The esterification step employs N,N-Dicyclohexylcarbodiimide (DCC) as a dehydrating coupling agent and 4-Dimethylaminopyridine (DMAP) as a nucleophilic catalyst to drive the formation of the ester bond at the C-4 hydroxyl group of podophyllotoxin. The reaction is conducted in dichloromethane under ice-bath conditions to minimize side reactions such as epimerization of the sensitive lignan stereocenters. DCC activates the carboxylic acid to form an O-acylisourea intermediate, which is then attacked by the C-4 alcohol of podophyllotoxin, facilitated by DMAP. A crucial aspect of this mechanism is the formation of dicyclohexylurea (DCU) as a byproduct, which is insoluble in many organic solvents and can be removed by filtration after freezing the reaction mixture at -20°C. This physical removal of DCU simplifies the purification process significantly compared to methods requiring aqueous washes that might degrade the product. The final product is isolated via column chromatography using ethyl acetate and petroleum ether, yielding high-purity benzoyl acrylic podophyllotoxin ester derivatives suitable for biological evaluation. 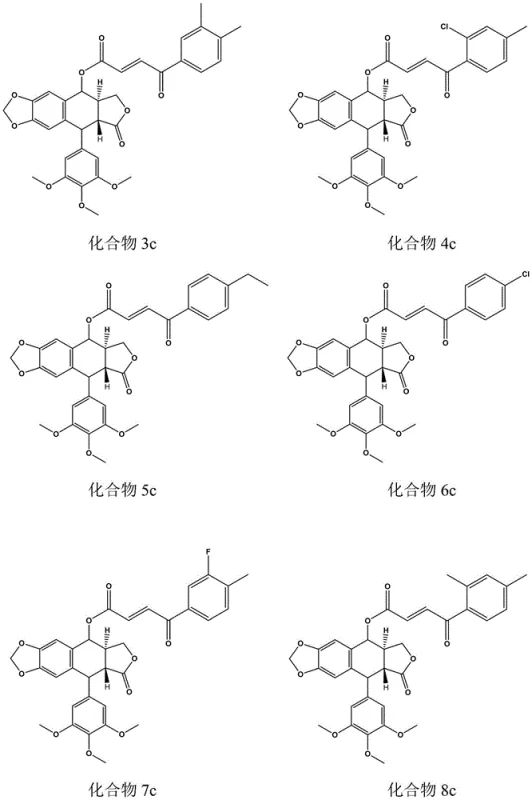
How to Synthesize Benzoyl Acrylic Podophyllotoxin Ester Derivatives Efficiently
The synthesis protocol described in the patent offers a robust framework for producing these high-value intermediates, emphasizing operational simplicity and reproducibility. The process begins with the preparation of the acid side chain, followed by the coupling with the natural product core, ensuring that each step is optimized for yield and purity. Detailed standard operating procedures for the synthesis, isolation, and purification of intermediates like 2a through 2l are established to guarantee consistency across batches. The use of common laboratory equipment such as rotary evaporators and separatory funnels indicates that the process is readily transferable from bench scale to pilot plant operations without requiring specialized high-pressure or cryogenic reactors. For process chemists, the key to success lies in the strict control of reaction temperatures during the coupling phase and the thorough removal of the DCU byproduct to prevent contamination.
- Preparation of substituted benzoylacrylic acid intermediates via Friedel-Crafts acylation of xylenes with maleic anhydride using anhydrous aluminum chloride catalyst.
- Purification of the acid intermediate through pH-controlled precipitation using sodium bicarbonate and dilute hydrochloric acid to ensure high purity.
- Coupling of the purified acid with podophyllotoxin using DCC and DMAP in dichloromethane under ice-bath conditions to form the target ester derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this synthetic route offers substantial benefits regarding raw material availability and process scalability, directly impacting the reliability of the supply chain for high-purity antitumor intermediates. The starting materials, including podophyllotoxin, maleic anhydride, and various substituted xylenes, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The reaction conditions are mild, predominantly occurring at room temperature or under simple ice-bath cooling, which eliminates the need for energy-intensive heating or deep-freeze infrastructure often required for cryogenic chemistry. This translates to significantly reduced operational expenditures and a lower carbon footprint for the manufacturing process, aligning with modern green chemistry initiatives. Furthermore, the workup procedures rely on standard liquid-liquid extraction and crystallization techniques rather than complex distillation or preparative HPLC, facilitating easier scale-up and reducing solvent consumption. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the demanding timelines of pharmaceutical development projects.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of stoichiometric reagents like DCC and DMAP, which are cost-effective and easily sourced, drives down the overall cost of goods sold. The simplified purification strategy, which leverages the insolubility of the DCU byproduct for removal, reduces the need for expensive chromatographic resins or extensive solvent exchanges. By avoiding harsh acidic or basic conditions that could degrade the expensive podophyllotoxin starting material, the process maximizes atom economy and yield, further enhancing cost efficiency. Additionally, the ability to recycle solvents like dichloromethane and petroleum ether after distillation contributes to long-term savings in material costs. These cumulative efficiencies allow for a competitive pricing structure for the final high-purity podophyllotoxin derivatives without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production schedules are not disrupted by shortages of exotic reagents or specialized catalysts. The robustness of the synthetic route, demonstrated by its tolerance to standard laboratory variations, ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance in the pharmaceutical sector. The modular nature of the synthesis, where different substituted acids can be coupled to the same podophyllotoxin core, allows for flexible manufacturing campaigns to produce various analogues based on market demand. This flexibility enables suppliers to respond rapidly to changes in client requirements or clinical trial needs, reducing lead time for high-purity antitumor intermediates. Moreover, the stability of the intermediates allows for strategic stockpiling, providing a buffer against potential logistical disruptions in the global supply network.
- Scalability and Environmental Compliance: The process is inherently scalable, as the exothermic nature of the Friedel-Crafts reaction is manageable at larger scales with appropriate cooling capacity, and the esterification step does not generate hazardous gaseous byproducts. The waste streams generated are primarily organic solvents and solid urea derivatives, which are well-characterized and can be treated using standard industrial waste management protocols. The absence of heavy metals in the catalyst system simplifies the environmental clearance process and reduces the burden on wastewater treatment facilities. Scaling from gram to kilogram quantities involves straightforward linear expansion of reactor volumes without the need for re-optimizing complex kinetic parameters. This ease of commercial scale-up of complex lignan esters ensures that the technology can support the transition from preclinical research to full-scale commercial production seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel podophyllotoxin derivatives. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their drug development pipelines. The answers are derived directly from the experimental data and beneficial effects reported in the patent literature, ensuring accuracy and relevance.
Q: What is the primary advantage of the C-4 modification in podophyllotoxin derivatives?
A: The C-4 modification with benzoyl acrylic groups significantly reduces cytotoxicity towards normal cells while maintaining or enhancing antitumor activity against strains like HGC-27 and MCF-7.
Q: How is the purity of the final ester derivative ensured during synthesis?
A: Purity is controlled through rigorous workup procedures including low-temperature precipitation to remove urea byproducts and column chromatography using ethyl acetate and petroleum ether systems.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes standard reagents like DCC and DMAP under mild conditions (room temperature to ice bath), making it highly adaptable for kilogram-to-ton scale manufacturing without exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Podophyllotoxin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the benzoyl acrylic podophyllotoxin ester synthesis route in advancing oncology drug discovery. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped to handle the specific requirements of lignan chemistry, including stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify structural integrity and impurity profiles. We understand that the consistency of chiral intermediates is paramount, and our manufacturing processes are designed to maintain the stereochemical fidelity of the podophyllotoxin backbone throughout the synthesis. By partnering with us, you gain access to a team of experts dedicated to optimizing yield and minimizing environmental impact while adhering to the highest international quality standards.
We invite you to collaborate with us to leverage this innovative synthetic technology for your next-generation antitumor programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We encourage you to reach out to request specific COA data for our available analogues and comprehensive route feasibility assessments to determine the optimal path forward for your development needs. Together, we can accelerate the delivery of safer and more effective cancer therapies to patients worldwide by overcoming the traditional limitations of podophyllotoxin-based treatments.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
