Scalable Synthesis of High-Potency 4-Beta-Carbon Substituted Podophyllotoxin Derivatives for Oncology
Scalable Synthesis of High-Potency 4-Beta-Carbon Substituted Podophyllotoxin Derivatives for Oncology
The pharmaceutical landscape for antineoplastic agents continues to evolve, driven by the need for compounds with broader spectra of activity and reduced toxicity profiles compared to legacy drugs like Etoposide. Patent CN1101819C introduces a significant advancement in this domain by disclosing a novel series of 4-beta-carbon substituted-4-deoxy-4'-demethylepipodophyllotoxin derivatives. These compounds represent a strategic structural modification of the classic podophyllotoxin scaffold, specifically targeting the C4 position to enhance biological potency. The patent outlines a robust synthetic methodology that not only achieves high stereoselectivity for the bioactive beta-configuration but also utilizes key intermediates that are amenable to large-scale manufacturing. For R&D directors and procurement specialists, this technology offers a pathway to next-generation oncology intermediates with improved cost-efficiency and supply reliability.
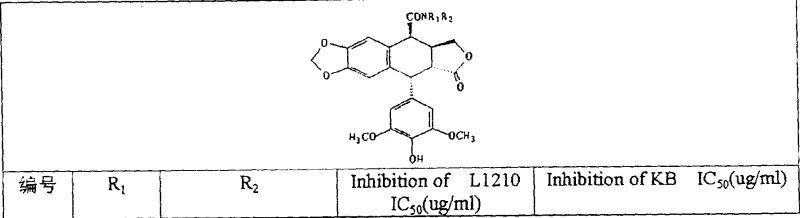
The core innovation lies in the precise insertion of carbon atoms between the C4 position and various heteroatom substituents (Nitrogen, Oxygen, or Sulfur), creating a new class of amides, esters, and thioesters. As illustrated in the structural framework, the versatility of the R1 and R2 groups allows for extensive structure-activity relationship (SAR) exploration. This flexibility is crucial for optimizing pharmacokinetic properties and minimizing side effects. The patent explicitly details the synthesis of key intermediates, namely 4-beta-cyano-4-deoxy-4'-demethylepipodophyllotoxin and its corresponding carboxylic acid derivative, which serve as the pivotal branching points for generating the final library of active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the C4 position in podophyllotoxin derivatives has been fraught with challenges regarding stereocontrol and reaction efficiency. Traditional nucleophilic substitution methods often suffer from poor yields and a lack of specificity, leading to mixtures of alpha and beta isomers that require difficult and costly separation processes. Furthermore, the use of hazardous reagents like solid potassium cyanide in conventional cyanation protocols presents significant safety risks and environmental burdens in a commercial setting. In the specific context of this patent's comparative data, the use of potassium cyanide resulted in a dismal yield of merely 8% for the key cyano-intermediate, rendering the process economically unviable for industrial scale-up. Such low efficiency translates directly to excessive waste generation and inflated raw material costs, creating a bottleneck for reliable supply chains.
The Novel Approach
The methodology described in CN1101819C overcomes these hurdles through the implementation of a Lewis acid-catalyzed system utilizing trimethylsilyl cyanide (Me3SiCN) and boron trifluoride etherate. This modern approach fundamentally shifts the reaction dynamics, facilitating a highly stereospecific attack that favors the formation of the desired 4-beta-cyano configuration. The impact on process efficiency is profound; the patent data reports a dramatic increase in yield to 76.3% when employing trimethylsilyl cyanide compared to the traditional potassium cyanide route. This nearly tenfold improvement in conversion efficiency is a game-changer for manufacturing economics. Additionally, the liquid nature of trimethylsilyl cyanide simplifies handling and dosing in reactor systems, enhancing operational safety and reducing the complexity of waste treatment protocols associated with solid cyanide salts.
Mechanistic Insights into Lewis Acid Catalyzed Stereospecific Cyanation
The success of this synthetic route hinges on the precise activation of the substrate by the Lewis acid catalyst, boron trifluoride etherate. In this mechanism, the Lewis acid coordinates with the oxygen functionality at the C4 position of the 4'-demethylepipodophyllotoxin starting material, effectively generating a highly reactive oxocarbenium ion intermediate. This activation lowers the energy barrier for nucleophilic attack, allowing the cyanide source to displace the leaving group with high fidelity. The steric environment of the rigid aryltetralin lignan scaffold, combined with the specific coordination geometry imposed by the catalyst, directs the incoming nucleophile to attack exclusively from the beta-face. This stereoelectronic control is critical, as the biological activity of podophyllotoxin derivatives is exquisitely sensitive to the spatial orientation of the C4 substituent.
Following the formation of the 4-beta-cyano intermediate, the pathway proceeds through a controlled hydrolysis step to generate the 4-beta-carboxylic acid. This transformation is achieved under acidic conditions using mineral acids like hydrochloric or sulfuric acid in polar protic solvents such as acetic acid. The resulting carboxylic acid serves as a versatile handle for further derivatization. By converting this acid into an acid chloride using thionyl chloride, the process enables a broad scope of downstream coupling reactions. This modularity allows manufacturers to readily attach diverse amines, alcohols, or thiols to construct the final amide, ester, or thioester libraries. The ability to access these distinct chemical classes from a single common intermediate streamlines the production workflow and minimizes the need for multiple distinct synthetic routes.
How to Synthesize 4-Beta-Carbon Substituted Podophyllotoxin Derivatives Efficiently
The synthesis of these high-value antineoplastic intermediates follows a logical three-stage sequence designed for maximum yield and purity. The process begins with the stereospecific cyanation of the starting lignan, followed by hydrolysis to the acid, and concludes with coupling to the desired side chain. This streamlined approach minimizes unit operations and purification steps, which is essential for maintaining cost competitiveness in the fine chemical sector. The detailed standardized synthesis steps for implementing this patented technology are outlined in the guide below.
- Perform stereospecific cyanation of 4'-demethylepipodophyllotoxin using trimethylsilyl cyanide and boron trifluoride etherate to obtain the 4-beta-cyano intermediate with high yield.
- Hydrolyze the 4-beta-cyano intermediate under acidic conditions (HCl or H2SO4 in acetic acid) to generate the key 4-beta-carboxyl intermediate.
- Activate the carboxyl group using thionyl chloride and couple with specific amines, alcohols, or thiols to form the final amide, ester, or thioester derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic improvement in process mass intensity and overall throughput. By shifting from a low-yielding cyanation protocol to one that achieves high conversion rates, manufacturers can significantly reduce the consumption of expensive starting materials per kilogram of final product. This efficiency gain directly correlates to a substantial reduction in the cost of goods sold (COGS), making the final API intermediates more price-competitive in the global market. Furthermore, the elimination of solid cyanide handling reduces the regulatory burden and insurance costs associated with hazardous material storage and transport.
- Cost Reduction in Manufacturing: The transition to a high-yield catalytic system eliminates the need for excessive recycling of unreacted starting materials, which is often a hidden cost driver in low-efficiency processes. The use of trimethylsilyl cyanide, while potentially higher in unit cost than potassium cyanide, is offset by the massive gain in yield and the reduction in downstream purification costs. The simplified workup procedures, which avoid complex chromatographic separations required for isomer mixtures, further contribute to operational expenditure savings. Consequently, the overall manufacturing footprint is reduced, allowing for higher production volumes within existing facility constraints without the need for capital-intensive expansion.
- Enhanced Supply Chain Reliability: The robustness of the Lewis acid catalyzed reaction ensures consistent batch-to-batch quality, which is a critical metric for qualifying suppliers in the pharmaceutical industry. High stereoselectivity means that the risk of producing off-spec material containing the inactive alpha-isomer is minimized, thereby reducing the likelihood of batch rejection and supply disruptions. Additionally, the reagents employed, such as boron trifluoride etherate and thionyl chloride, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages. This reliability allows downstream drug manufacturers to plan their production schedules with greater confidence and maintain leaner inventory levels.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from laboratory gram-scale experiments to multi-kilogram pilot runs and eventually to commercial tonnage production. The reaction conditions, operating at moderate temperatures and pressures, are compatible with standard glass-lined or stainless steel reactors found in most fine chemical plants. From an environmental perspective, the higher atom economy of the new method results in less chemical waste per unit of product. The avoidance of heavy metal catalysts and the use of manageable solvent systems simplify effluent treatment, ensuring compliance with increasingly stringent environmental regulations and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these podophyllotoxin derivatives. The answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your existing product portfolio or supply network.
Q: Why is trimethylsilyl cyanide preferred over potassium cyanide in this synthesis?
A: According to patent data, using trimethylsilyl cyanide (Me3SiCN) with BF3·Et2O increases the yield of the key cyano-intermediate from approximately 8% (with KCN) to over 76%, while also offering safer handling characteristics compared to solid cyanide salts.
Q: What is the biological significance of the 4-beta configuration?
A: The patent highlights that the beta-configuration at the C4 position is critical for biological activity. The described Lewis acid catalyzed method ensures high stereoselectivity for the beta-isomer, which exhibits superior antitumor properties compared to alpha-isomers or other substitutions.
Q: How do these derivatives compare to Etoposide in terms of potency?
A: Pharmacological data indicates that specific derivatives, particularly certain aliphatic esters (e.g., compound II-10), demonstrate antitumor activity exceeding that of Etoposide by two orders of magnitude against L1210 leukemia cells, suggesting a potential for enhanced therapeutic efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Podophyllotoxin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-purity intermediates for the development of next-generation oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1101819C can be translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 4-beta-carbon substituted derivatives meets the exacting standards required for clinical and commercial applications. Our commitment to quality assurance ensures that your drug development timeline remains on track without delays caused by material inconsistencies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Whether you require custom synthesis of specific analogs or bulk supply of the key cyano- or carboxyl-intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall project costs.
