Revolutionizing 1H-1,2,3-Triazole Production: A One-Pot Strategy for Scalable API Intermediate Manufacturing
Introduction to Advanced Triazole Synthesis
The pharmaceutical industry continuously seeks robust and scalable pathways for critical heterocyclic building blocks, and 1H-1,2,3-triazole stands out as a pivotal intermediate in the synthesis of complex bioactive molecules, including the beta-lactamase inhibitor tazobactam. Recent intellectual property developments, specifically patent CN111320587A, have unveiled a transformative one-pot methodology that addresses longstanding inefficiencies in triazole manufacturing. This novel approach leverages a unified ethanol solvent system to execute reduction, cyclization, and deamination sequentially, thereby eliminating the logistical burdens of solvent swapping and intermediate isolation. For R&D directors and procurement strategists, this represents a significant leap forward in process intensification, offering a route that is not only chemically elegant but also inherently safer and more environmentally compliant than legacy technologies. The ability to produce high-purity 1H-1,2,3-triazole without generating hazardous solid wastes or relying on explosive diazonium chemistry positions this technology as a cornerstone for modern API intermediate supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1H-1,2,3-triazole has been plagued by multi-step complexities and hazardous reagent profiles that inflate both operational costs and safety liabilities. One prominent legacy route initiates with o-phenylenediamine, which undergoes diazotization and cyclization to form benzotriazole, followed by aggressive oxidation with potassium permanganate to yield 1H-1,2,3-triazole-4,5-dicarboxylic acid, and finally decarboxylation using copper powder in dioxane. 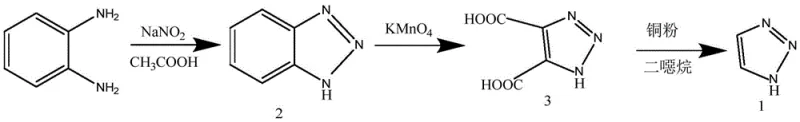 This pathway is fundamentally flawed for large-scale production due to its reliance on dioxane, a solvent with significant toxicity and regulatory scrutiny, alongside a low overall yield that necessitates expensive purification protocols. Furthermore, alternative routes utilizing p-toluenesulfonyl chloride introduce severe handling challenges, as this reagent is a potent lachrymator and moisture-sensitive solid that complicates reactor charging and requires stringent atmospheric controls. Other methods involving nitrite-mediated deamination pose unacceptable explosion risks due to the transient formation of diazonium salts, creating a safety bottleneck that limits batch sizes and increases insurance and containment costs for manufacturing facilities.
This pathway is fundamentally flawed for large-scale production due to its reliance on dioxane, a solvent with significant toxicity and regulatory scrutiny, alongside a low overall yield that necessitates expensive purification protocols. Furthermore, alternative routes utilizing p-toluenesulfonyl chloride introduce severe handling challenges, as this reagent is a potent lachrymator and moisture-sensitive solid that complicates reactor charging and requires stringent atmospheric controls. Other methods involving nitrite-mediated deamination pose unacceptable explosion risks due to the transient formation of diazonium salts, creating a safety bottleneck that limits batch sizes and increases insurance and containment costs for manufacturing facilities.
The Novel Approach
In stark contrast to these fragmented and hazardous legacy processes, the methodology disclosed in CN111320587A introduces a cohesive one-pot strategy that streamlines the entire synthetic sequence into a single reaction vessel using ethanol as the sole solvent. This innovative route begins with the condensation of glyoxal and hydrazine hydrate to form a glyoxal dihydrazone intermediate, which is subsequently subjected to oxidative cyclization using hydrogen peroxide rather than heavy metal oxidants. 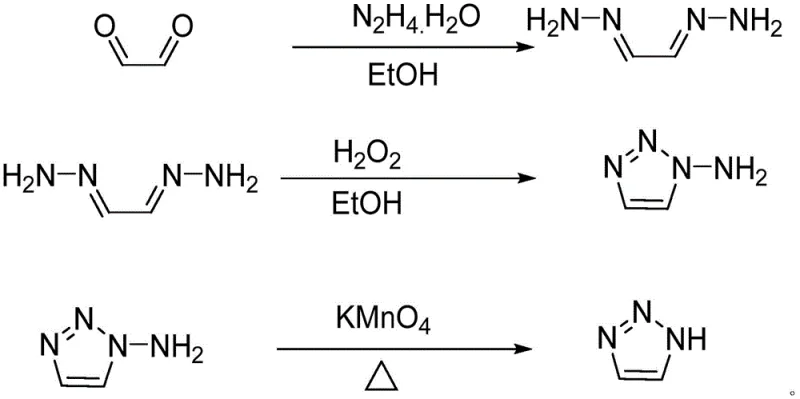 The final deamination step utilizes potassium permanganate under controlled thermal conditions to cleave the amino group, yielding the target triazole without the need for intermediate isolation or solvent exchange. By maintaining a consistent ethanol environment throughout the reduction, cyclization, and deamination phases, this process drastically reduces energy consumption associated with distillation and solvent recovery, while simultaneously minimizing the generation of solid waste residues that typically burden downstream waste management teams. The elimination of toxic solvents like dioxane and hazardous gases like ammonia further enhances the environmental profile, making this a highly attractive option for sustainable chemical manufacturing.
The final deamination step utilizes potassium permanganate under controlled thermal conditions to cleave the amino group, yielding the target triazole without the need for intermediate isolation or solvent exchange. By maintaining a consistent ethanol environment throughout the reduction, cyclization, and deamination phases, this process drastically reduces energy consumption associated with distillation and solvent recovery, while simultaneously minimizing the generation of solid waste residues that typically burden downstream waste management teams. The elimination of toxic solvents like dioxane and hazardous gases like ammonia further enhances the environmental profile, making this a highly attractive option for sustainable chemical manufacturing.
Mechanistic Insights into Oxidative Cyclization and Deamination
The core chemical innovation of this process lies in the strategic selection of oxidants for the cyclization and deamination steps, which fundamentally alters the impurity profile and reaction kinetics compared to traditional methods. In the cyclization phase, hydrogen peroxide acts as a clean oxidant that converts the glyoxal dihydrazone into 1-amino-1,2,3-triazole with water as the primary byproduct, avoiding the formation of insoluble manganese dioxide sludge that characterizes older permanganate-based cyclizations. This homogenous reaction environment ensures efficient mass transfer and prevents the occlusion of product within solid waste matrices, leading to higher recovery rates and simplified filtration operations. The subsequent deamination step employs potassium permanganate not for ring formation, but specifically for the oxidative removal of the N-amino group, a transformation that proceeds through a nitroso intermediate before hydrolytic cleavage of the N-N bond. This mechanistic pathway is significantly faster and safer than acid-catalyzed diazotization, as it bypasses the high-energy diazonium species that are prone to thermal runaway and detonation.
From an impurity control perspective, the one-pot nature of the reaction minimizes exposure to atmospheric moisture and oxygen between steps, which are common sources of degradation in multi-vessel syntheses. The use of ethanol, a polar protic solvent, facilitates the solubility of both the hydrazine starting materials and the triazole intermediates, ensuring that side reactions such as polymerization or over-oxidation are kinetically suppressed. Moreover, the stoichiometric control of hydrogen peroxide and potassium permanganate allows for precise tuning of the oxidation potential, preventing the over-oxidation of the triazole ring which can lead to ring-opening byproducts. The final workup involves a straightforward extraction with dichloromethane followed by vacuum distillation, a purification technique that effectively separates the volatile triazole product from non-volatile inorganic salts and residual oxidants, ensuring that the final API intermediate meets stringent pharmacopeial specifications for heavy metals and organic impurities.
How to Synthesize 1H-1,2,3-Triazole Efficiently
The operational simplicity of this patented method makes it an ideal candidate for technology transfer from laboratory scale to commercial production, requiring only standard stainless steel reactors equipped with temperature control and dosing systems. The process is designed to be robust against minor variations in reagent addition rates, providing a wide operating window that accommodates different manufacturing setups.
- Perform reduction reaction by adding glyoxal to hydrazine hydrate in ethanol at -5 to 25°C to form glyoxal dihydrazone.
- Execute oxidative cyclization by introducing hydrogen peroxide to the solution at 5-45°C to generate 1-amino-1,2,3-triazole.
- Complete deamination by adding potassium permanganate and heating at 5-55°C, followed by dichloromethane extraction and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this one-pot synthesis route offers compelling economic and logistical advantages that directly impact the bottom line and operational resilience. The consolidation of three distinct chemical transformations into a single vessel eliminates the need for multiple reactor campaigns, intermediate storage tanks, and the associated cleaning validation protocols, resulting in a drastically simplified production schedule.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of solvent exchange steps and the use of commodity-grade reagents. By utilizing ethanol throughout the entire sequence, manufacturers avoid the capital and operational expenditures associated with recovering and recycling multiple specialized solvents like dioxane or dichloromethane at intermediate stages. Furthermore, the replacement of expensive and hazardous reagents like p-toluenesulfonyl chloride with readily available glyoxal and hydrazine hydrate reduces raw material procurement costs and mitigates the risk of supply disruptions caused by the limited availability of specialty sulfonyl chlorides. The reduction in solid waste generation also translates to lower disposal fees and reduced liability, contributing to a leaner cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals such as ethanol, hydrogen peroxide, and glyoxal ensures a stable and diversified supply base, reducing dependency on single-source suppliers of exotic reagents. Ethanol is globally produced in massive quantities for fuel and beverage industries, guaranteeing consistent availability and price stability even during market fluctuations. Additionally, the simplified process flow shortens the overall cycle time from raw material intake to finished goods, allowing manufacturers to respond more agilely to demand spikes from downstream pharmaceutical clients. The absence of complex intermediate isolation steps also reduces the physical footprint required for production, enabling higher throughput within existing facility constraints and improving overall asset utilization rates.
- Scalability and Environmental Compliance: From a regulatory and sustainability standpoint, this process aligns perfectly with modern green chemistry principles, facilitating easier permitting and compliance with increasingly strict environmental regulations. The avoidance of carcinogenic solvents like dioxane and lachrymatory agents like tosyl chloride simplifies occupational health and safety monitoring, reducing the need for specialized containment equipment and personal protective gear. The aqueous nature of the hydrogen peroxide oxidation step minimizes the generation of hazardous organic waste streams, lowering the burden on wastewater treatment facilities. This environmental compatibility not only future-proofs the manufacturing site against regulatory tightening but also enhances the brand reputation of the supplier as a responsible partner in the sustainable pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis method, drawing directly from the experimental data and beneficial effects outlined in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for large-scale intermediate production.
Q: Why is the one-pot ethanol method superior to traditional dioxane-based routes?
A: The one-pot ethanol method eliminates the need for solvent exchange and avoids toxic dioxane, significantly reducing waste treatment costs and environmental hazards while simplifying the operational workflow.
Q: How does this process mitigate safety risks associated with diazonium salts?
A: By utilizing potassium permanganate for deamination instead of nitrites, the process avoids the formation of unstable diazonium intermediates, thereby removing the explosion risks inherent in traditional diazotization methods.
Q: What are the purity implications of using hydrogen peroxide for cyclization?
A: Hydrogen peroxide offers high oxidation efficiency with water as the only byproduct, preventing the generation of solid manganese dioxide waste and resulting in a cleaner reaction profile with fewer impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process engineering capabilities. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot synthesis are fully realized in an industrial setting. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating every batch against the highest international standards, guaranteeing that your supply of 1H-1,2,3-triazole remains uninterrupted and compliant. We understand the critical nature of API intermediates in the drug development timeline and are committed to delivering consistent quality that supports your regulatory filings and clinical trials.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain to drive efficiency and reduce total landed costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the specific economic impacts relevant to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-purity 1H-1,2,3-triazole that meets your exacting specifications. Let us collaborate to secure a resilient and cost-effective supply of this vital pharmaceutical building block.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
