Scalable Synthesis of 1H-1,2,3-Triazoles Using Recyclable Porous Copper Catalysts
The rapid expansion of the pharmaceutical and agrochemical sectors has intensified the demand for robust, scalable synthetic routes for heterocyclic scaffolds, particularly 1H-1,2,3-triazoles, which serve as critical bioisosteres in modern drug design. A significant technological breakthrough in this domain is documented in patent CN102603660A, which discloses a novel preparation method for 1H-1,2,3-triazole compounds utilizing a heterogeneous porous copper catalyst. This innovation addresses long-standing challenges in click chemistry by replacing expensive, hard-to-remove homogeneous catalysts with a recyclable, commercially available porous copper material. The process operates under mild conditions, typically between 25-80°C, and notably employs deionized water as the primary reaction solvent, marking a substantial departure from traditional organic solvent-heavy protocols. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this methodology offers a compelling value proposition by merging high chemical efficiency with environmental sustainability and cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles via the copper-catalyzed azide-alkyne cycloaddition (CuAAC) has relied heavily on homogeneous copper sources such as copper sulfate paired with reducing agents like sodium ascorbate, or soluble copper(I) salts. While effective on a small laboratory scale, these conventional methods present severe bottlenecks for industrial manufacturing. The primary drawback lies in the difficulty of removing residual copper from the final product, which is a critical quality attribute for active pharmaceutical ingredients (APIs) due to strict regulatory limits on heavy metals. Furthermore, homogeneous catalysts are single-use, leading to significant material costs and generating large volumes of copper-contaminated waste streams that require expensive treatment. The reliance on organic solvents such as dichloromethane or DMF further exacerbates environmental liabilities and operational costs, making cost reduction in pharmaceutical intermediate manufacturing a persistent challenge for supply chain heads.
The Novel Approach
The methodology outlined in patent CN102603660A introduces a transformative one-pot strategy that utilizes porous copper as a heterogeneous catalyst, effectively circumventing the limitations of traditional homogeneous systems. By employing a solid-state catalyst with a specific pore structure (optimally 5-20μm), the reaction facilitates easy separation of the catalyst from the reaction mixture simply by filtration, allowing the copper to be reused multiple times without significant loss of activity. This approach not only adheres to the principles of atom economy but also drastically simplifies the downstream processing workflow. The ability to conduct the reaction in deionized water, a non-toxic and inexpensive solvent, eliminates the need for volatile organic compounds (VOCs), thereby reducing fire hazards and solvent recovery costs. This novel route provides a clear pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring high purity and consistent quality while minimizing the environmental footprint.
Mechanistic Insights into Porous Copper-Catalyzed Cycloaddition
The catalytic mechanism of this process relies on the unique surface properties of the porous copper, which acts as a source of active copper(I) species essential for the 1,3-dipolar cycloaddition. Unlike soluble salts, the porous structure provides a high surface-area-to-volume ratio, enhancing the interaction between the solid catalyst and the dissolved reactants (halide, alkyne, and sodium azide). The reaction initiates with the in situ generation of an organic azide from the halide and sodium azide, which then undergoes cycloaddition with the terminal alkyne on the copper surface. The pore size of the copper is a critical parameter; experimental data indicates that smaller pore sizes (around 5μm) yield significantly higher conversion rates compared to larger pores (50-150μm), likely due to increased accessibility of active sites. This heterogeneous nature ensures that the bulk of the copper remains in the solid phase, preventing leaching into the product stream and ensuring that the final triazole compound meets stringent purity specifications required for sensitive biological applications.
Impurity control is another pivotal aspect of this mechanistic advantage. In traditional homogeneous catalysis, side reactions such as Glaser coupling (oxidative dimerization of alkynes) can occur, leading to difficult-to-separate byproducts. The controlled environment provided by the porous copper surface, combined with the use of water as a solvent, suppresses these competing pathways. The mild reaction temperatures (55-60°C) further minimize thermal degradation of sensitive functional groups often present in complex drug intermediates. For R&D teams, this means a cleaner crude reaction profile, which translates to fewer purification steps and higher overall yields. The robustness of the catalyst allows it to tolerate a wide range of substrates, including electron-rich and electron-deficient aromatic alkynes, as well as aliphatic chains, demonstrating the versatility required for diverse synthetic campaigns in both agrochemical and medicinal chemistry.
How to Synthesize 1H-1,2,3-Triazole Compounds Efficiently
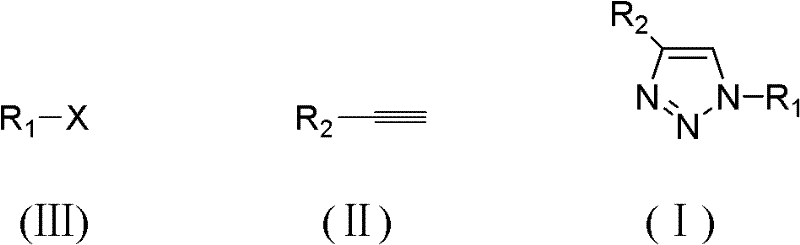
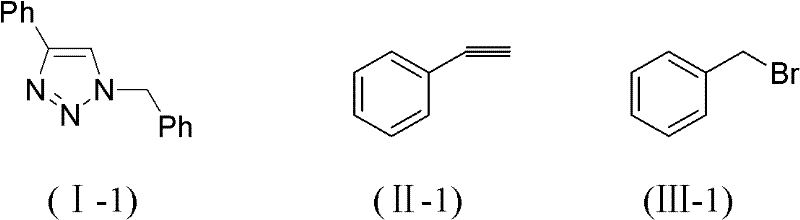
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and reaction parameters as defined in the patent literature. The process involves mixing a halide substrate (Formula III), a terminal alkyne (Formula II), and sodium azide in deionized water, followed by the addition of the porous copper catalyst. The detailed standardized synthesis steps below outline the optimal conditions for achieving maximum yield and purity, ensuring reproducibility from gram-scale development to multi-ton production. Following these guidelines allows manufacturers to leverage the full economic and technical benefits of this green chemistry approach.
- Mix halide (Formula III), terminal alkyne (Formula II), and sodium azide in deionized water.
- Add porous copper catalyst (5-150μm pore size) and heat the mixture to 25-80°C for 5-85 hours.
- Extract the reaction mixture with ethyl acetate, wash, dry, and purify via column chromatography to obtain the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this porous copper-catalyzed process offers tangible strategic advantages beyond mere technical feasibility. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of triazole production by eliminating the recurring expense of precious metal ligands and reducing waste disposal fees associated with heavy metal contamination. The use of water as a solvent not only lowers raw material costs but also simplifies regulatory compliance regarding VOC emissions. These factors collectively contribute to a more resilient and cost-efficient supply chain, capable of meeting the rigorous demands of global pharmaceutical markets without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The implementation of recyclable porous copper catalysts significantly lowers the cost of goods sold (COGS) by removing the need for stoichiometric amounts of expensive copper salts and ligands. Since the catalyst can be recovered and reused multiple times without extensive regeneration processes, the amortized cost per kilogram of product is drastically reduced. Furthermore, the elimination of organic solvents in favor of deionized water removes the substantial overhead associated with solvent purchase, storage, and distillation recovery, leading to substantial cost savings in utility and material consumption.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including simple halides, terminal alkynes, and sodium azide, are commodity chemicals with stable global supply chains, reducing the risk of production stoppages due to material shortages. The robustness of the porous copper catalyst, which is commercially available and does not require specialized synthesis, ensures that the critical catalytic component is always accessible. This reliability allows for consistent production scheduling and shorter lead times for high-purity pharmaceutical intermediates, enabling partners to respond swiftly to market fluctuations and urgent API demands.
- Scalability and Environmental Compliance: The one-pot nature of this reaction, combined with the use of water, makes it inherently scalable from pilot plants to large-scale commercial reactors without the need for complex engineering modifications. The process generates minimal hazardous waste, aligning with increasingly strict environmental regulations and corporate sustainability goals. By avoiding toxic organic solvents and minimizing heavy metal waste, manufacturers can reduce their environmental liability and streamline the permitting process for new production lines, facilitating faster time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the porous copper-catalyzed synthesis of 1H-1,2,3-triazoles. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical implementation and advantages of this technology for potential partners and stakeholders.
Q: What are the advantages of using porous copper over traditional homogeneous copper catalysts?
A: Porous copper acts as a heterogeneous catalyst, allowing for easy filtration and reuse without complex purification steps, significantly reducing heavy metal contamination in the final API intermediate.
Q: Can this synthesis method be performed in environmentally friendly solvents?
A: Yes, the patent specifically highlights deionized water as the preferred solvent, which eliminates the need for toxic organic solvents and lowers waste treatment costs.
Q: What is the typical yield range for this triazole synthesis?
A: Under optimized conditions using 5μm porous copper at 55-60°C, yields can exceed 95%, demonstrating high efficiency for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the porous copper-catalyzed synthesis route for producing high-quality 1H-1,2,3-triazole derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry method are fully realized in an industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of triazole intermediate meets the exacting standards required for downstream API synthesis. We are committed to delivering products that not only perform exceptionally in chemical reactions but also adhere to the highest safety and environmental protocols.
We invite pharmaceutical and agrochemical companies to collaborate with us to optimize their supply chains using this advanced technology. By leveraging our expertise, you can achieve significant process efficiencies and cost optimizations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your development goals and ensure a steady supply of critical intermediates for your global operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
