Advanced Synthesis of Carbazole-Modified BODIPY Dyes for High-Efficiency Laser Applications
Advanced Synthesis of Carbazole-Modified BODIPY Dyes for High-Efficiency Laser Applications
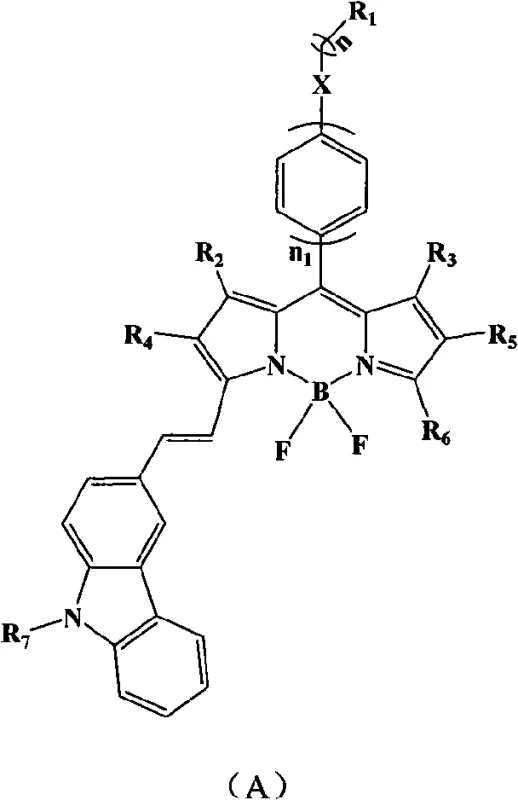
The development of high-performance fluorescent materials remains a cornerstone of innovation in the optoelectronics and bio-imaging sectors. Patent CN101565554A introduces a groundbreaking class of strong-fluorescence boron dipyrromethene (BODIPY) dyes that incorporate a carbazole structure, addressing long-standing challenges in achieving efficient red-emitting laser dyes. Traditional BODIPY derivatives often struggle with limited Stokes shifts or reduced quantum yields when modified for longer wavelength emission. This patented technology overcomes these limitations by strategically extending the conjugated system through the attachment of substituted carbazole aldehydes. The result is a compound family that exhibits a remarkable red shift in absorption spectra by approximately 80 nanometers and emission spectra by 90 nanometers, all while preserving a high fluorescence quantum yield of 0.67 and achieving laser efficiencies exceeding 30 percent in ethyl acetate. For R&D directors and procurement specialists seeking reliable fluorescent material suppliers, this synthesis route represents a significant leap forward in creating stable, high-efficiency optical standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of red-emitting BODIPY dyes has been fraught with complexity and inefficiency. Conventional methods often rely on harsh conditions or multi-step sequences that introduce impurities difficult to remove, leading to batch-to-batch variability that is unacceptable for precision optical applications. Many existing protocols utilize transition metal catalysts which, while effective for coupling, introduce the risk of heavy metal contamination requiring expensive downstream purification processes. Furthermore, achieving a significant red shift in the emission spectrum typically comes at the cost of fluorescence quantum yield; as the conjugation length increases to push wavelengths into the red region, non-radiative decay pathways often dominate, quenching the fluorescence. This trade-off has limited the commercial viability of many red BODIPY dyes for laser applications, where both high intensity and specific wavelength output are critical. The instability of some precursors and the sensitivity of the BODIPY core to acidic or basic degradation during functionalization further complicate the manufacturing landscape, driving up costs and extending lead times for high-purity electronic chemicals.
The Novel Approach
The methodology outlined in patent CN101565554A offers a streamlined and robust alternative that directly addresses these synthetic bottlenecks. By employing a condensation reaction between a boron dipyrromethene core containing active methylene groups and various substituted carbazole aldehydes, the process achieves extensive conjugation in a single strategic step. The use of piperidine as a mild organic base catalyst avoids the need for expensive and toxic transition metals, simplifying the impurity profile and reducing environmental compliance burdens. Crucially, the inclusion of 4-angstrom molecular sieves as a dehydrating agent drives the equilibrium forward effectively, ensuring high conversion rates even with sterically hindered substrates. This approach allows for the modular introduction of diverse substituents on the carbazole ring, enabling fine-tuning of solubility and solid-state packing properties without compromising the core photophysical performance. For supply chain heads, this modularity means that a single platform technology can generate a library of specialized dyes, enhancing supply continuity and reducing the need for multiple distinct manufacturing lines.
Mechanistic Insights into Piperidine-Catalyzed Condensation
The core chemical transformation in this patent is a Knoevenagel-type condensation, facilitated by the unique electronic properties of the BODIPY core and the carbazole aldehyde. The reaction initiates with the deprotonation of the active methyl or methylene group on the BODIPY pyrrole ring by the piperidine catalyst, generating a nucleophilic carbanion stabilized by the adjacent electron-withdrawing boron-nitrogen complex. This nucleophile then attacks the electrophilic carbonyl carbon of the carbazole aldehyde, forming a beta-hydroxy intermediate. Under the elevated reaction temperatures of 100 to 150 degrees Celsius, this intermediate undergoes dehydration to form the final carbon-carbon double bond, extending the pi-conjugation system across the carbazole and BODIPY units. The presence of the carbazole unit is particularly advantageous as it acts as a strong electron donor, pushing electron density into the BODIPY acceptor core, which is the fundamental driver for the observed bathochromic shifts in absorption and emission. Understanding this mechanism is vital for process chemists aiming to optimize reaction kinetics and minimize side reactions such as polymerization or over-condensation.
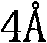
Impurity control in this synthesis is largely governed by the stoichiometry of the reactants and the efficiency of water removal. The patent specifies a molar ratio of boron dipyrromethene to carbazole aldehyde ranging from 1:2 to 1:5, ensuring that the aldehyde is in excess to drive the reaction to completion and minimize unreacted starting material. The 4-angstrom molecular sieves play a dual role: physically adsorbing the water byproduct to prevent the reverse hydrolysis reaction and potentially sequestering trace polar impurities that could interfere with the catalyst. Post-reaction purification typically involves silica gel column chromatography, which effectively separates the highly conjugated product from polar byproducts and residual catalyst. The structural rigidity imparted by the newly formed double bond and the fused ring systems contributes to the photochemical stability of the final dye, a key parameter for long-term reliability in laser devices and biological probes where photobleaching is a major failure mode.
How to Synthesize Carbazole-BODIPY Dyes Efficiently
The synthesis protocol described in the patent provides a clear pathway for producing these advanced fluorophores with high reproducibility. The process begins with the precise weighing of the boron dipyrromethene precursor and the selected N-substituted carbazole aldehyde, ensuring the molar ratios fall within the optimized range of 1:2 to 1:5. These reagents are suspended in a high-boiling organic solvent such as orthodichlorobenzene, toluene, or N-methyl pyrrolidone, chosen for their ability to dissolve both reactants and withstand the required reaction temperatures. The addition of piperidine catalyst and activated 4-angstrom molecular sieves is critical before heating commences. The detailed standardized synthesis steps for this process are provided in the guide below.
- Prepare the reaction mixture by combining the boron dipyrromethene core and substituted carbazole aldehyde in a molar ratio of 1: 2 to 1:5 within an organic solvent such as toluene or orthodichlorobenzene.
- Add piperidine as a catalyst (molar ratio 1: 0.01 to 1:0.20 relative to the core) and introduce 4-angstrom molecular sieves as a dehydrating agent to drive the equilibrium.
- Heat the mixture to 100-150°C under inert gas protection (argon or nitrogen) with stirring for 8 to 50 hours, followed by purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of cost structure and operational reliability. The elimination of precious metal catalysts such as palladium or platinum, which are common in cross-coupling alternatives, results in significant raw material cost savings and removes the regulatory burden associated with heavy metal residue limits in pharmaceutical or electronic grade products. The use of commodity chemicals like piperidine and widely available solvents ensures that the supply chain is not vulnerable to the geopolitical volatility often seen with specialized reagents. Furthermore, the robustness of the reaction conditions, which tolerate a range of temperatures and solvents, allows for flexibility in manufacturing site selection and equipment utilization, reducing the risk of production stoppages due to minor process deviations.
- Cost Reduction in Manufacturing: The process leverages inexpensive organocatalysts and avoids the need for cryogenic conditions or ultra-high vacuum systems, significantly lowering energy consumption and capital expenditure requirements. By utilizing a direct condensation strategy, the number of synthetic steps is minimized compared to multi-step functionalization routes, which inherently reduces labor costs, solvent usage, and waste generation. The high atom economy of the condensation reaction, combined with the ability to recover and reuse molecular sieves after regeneration, contributes to a leaner manufacturing process that aligns with green chemistry principles and reduces overall disposal costs.
- Enhanced Supply Chain Reliability: The starting materials, specifically the carbazole aldehydes and BODIPY cores, are derived from bulk petrochemical feedstocks, ensuring a stable and continuous supply stream that is less susceptible to shortages than bio-based or rare-earth-dependent alternatives. The modular nature of the synthesis allows manufacturers to switch between different carbazole substituents with minimal process revalidation, providing agility in responding to specific customer demands for varying emission wavelengths. This flexibility ensures that supply chain heads can maintain inventory levels of key intermediates while customizing the final product mix based on real-time market needs without disrupting the core production flow.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure using standard glass-lined or stainless steel reactors, facilitating straightforward scale-up from laboratory grams to multi-ton commercial production without complex engineering changes. The absence of halogenated byproducts or toxic metal waste simplifies effluent treatment processes, making it easier to meet stringent environmental regulations in major manufacturing hubs. The use of molecular sieves as a solid dehydrating agent eliminates the need for azeotropic distillation setups, reducing energy load and simplifying the reactor configuration, which is a critical factor for scaling complex electronic chemicals efficiently and safely.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole-modified BODIPY dyes. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for potential partners evaluating this technology for integration into their product lines.
Q: What is the primary advantage of introducing a carbazole structure to BODIPY dyes?
A: Introducing a carbazole structure significantly extends the conjugated pi-system, resulting in a substantial red shift of both absorption (approx. 80nm) and emission spectra (approx. 90nm) while maintaining high fluorescence quantum yields around 0.67.
Q: What role do molecular sieves play in this synthesis process?
A: 4-angstrom molecular sieves act as a critical dehydrating agent, removing water generated during the condensation reaction between the active methylene group and the aldehyde, thereby driving the reaction equilibrium towards the product and improving yield.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes robust conditions (100-150°C), common organic solvents like toluene or chlorobenzene, and inexpensive catalysts like piperidine, making it highly suitable for commercial scale-up without requiring exotic transition metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole-BODIPY Dye Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable chemical solutions. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical infrastructure to manufacture these complex fluorophores with stringent purity specifications required for high-end optical applications. Our rigorous QC labs are equipped to verify the photophysical properties, including quantum yield and emission maxima, ensuring that every batch meets the exacting standards necessary for laser dye and bio-probe formulations. We understand the critical nature of supply continuity in the electronics and pharmaceutical sectors and have established robust logistics networks to ensure timely delivery globally.
We invite you to collaborate with us to optimize your supply chain for next-generation fluorescent materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product performance while reducing total landed costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
