Scalable Base-Catalyzed Synthesis of N-Acyl Indoles for Pharmaceutical Manufacturing
Scalable Base-Catalyzed Synthesis of N-Acyl Indoles for Pharmaceutical Manufacturing
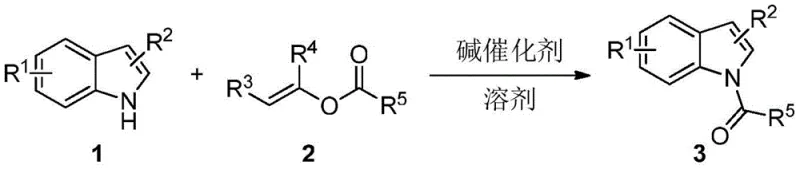
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance high efficiency with environmental sustainability. Patent CN108752256B introduces a transformative approach to the preparation of N-acyl indole compounds, utilizing carboxylic acid alkenyl esters as acylating agents under mild base catalysis. This technology addresses critical bottlenecks in the production of these vital organic synthesis intermediates, which serve as foundational building blocks for numerous active pharmaceutical ingredients (APIs) and agrochemicals. By shifting away from traditional, hazardous acylating reagents, this method offers a greener pathway that maintains exceptional selectivity and yield. For R&D directors and procurement specialists, understanding the mechanistic advantages of this vinyl ester-based system is crucial for optimizing supply chains and reducing the total cost of ownership for complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has relied heavily on the use of acyl halides or acid anhydrides as the primary acylating reagents. While these classical methods are well-documented, they suffer from significant inherent drawbacks that complicate large-scale manufacturing. The high reactivity of acid halides often leads to poor chemoselectivity, resulting in competitive acylation at the C3 position of the indole ring rather than the desired nitrogen atom. Furthermore, these reactions typically require harsh conditions and generate stoichiometric amounts of corrosive acidic byproducts, necessitating complex neutralization and waste treatment protocols. The difficulty in controlling the reaction trajectory often leads to lower yields and extensive purification challenges, which directly impacts the economic viability of producing high-purity pharmaceutical intermediates. Additionally, the sensitivity of acid halides to moisture requires stringent anhydrous conditions, further increasing operational costs and safety risks in an industrial setting.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes carboxylic acid alkenyl esters, such as vinyl acetate, as the acyl source, fundamentally altering the reaction thermodynamics. This approach leverages the unique tautomerization capability of the leaving group; upon nucleophilic attack by the indole nitrogen, the expelled enol spontaneously tautomerizes into a stable aldehyde or ketone. This irreversible transformation provides a powerful thermodynamic driving force that pushes the equilibrium towards the product, effectively preventing the reverse acylation reaction. Consequently, this system achieves superior N-selectivity without the need for expensive transition metal catalysts or aggressive reagents. The mild reaction conditions, ranging from ambient temperature to moderate heating, ensure excellent functional group tolerance, allowing for the successful acylation of indoles bearing sensitive substituents like cyano, nitro, or halogen groups without degradation.
Mechanistic Insights into Base-Catalyzed Vinyl Ester Acylation
The core of this technological advancement lies in the elegant simplicity of the base-catalyzed mechanism. The reaction initiates with the deprotonation of the indole nitrogen by a mild organic or inorganic base, generating a nucleophilic indolyl anion. This anion then attacks the carbonyl carbon of the vinyl ester, forming a tetrahedral intermediate. Unlike traditional acylation where a halide ion is expelled, this process eliminates a vinyloxy group which rapidly rearranges into a stable carbonyl compound, such as acetaldehyde in the case of vinyl acetate. This irreversible step is the key to the method's high efficiency and selectivity. The absence of metal catalysts eliminates the risk of heavy metal contamination, a critical parameter for API manufacturing, while the use of simple amines or carbonate salts significantly lowers the catalyst cost profile. This mechanistic clarity allows for precise tuning of reaction parameters to maximize throughput.
Furthermore, the impurity profile of this reaction is remarkably clean due to the high specificity of the nitrogen attack. In conventional methods, C3-acylation is a persistent side reaction that requires difficult chromatographic separation. However, the steric and electronic properties of the vinyl ester system, combined with the specific basicity of the catalyst, favor N-acylation almost exclusively. This high selectivity simplifies the downstream processing, as the crude reaction mixture contains fewer structural isomers. For quality control teams, this means a more consistent impurity spectrum that is easier to characterize and control. The ability to use a wide variety of solvents, from polar aprotic solvents like DMSO to non-polar options like toluene, provides additional flexibility to optimize solubility and reaction kinetics for different substrate combinations, ensuring robust performance across a diverse library of indole derivatives.
How to Synthesize N-Acyl Indole Compounds Efficiently
The operational protocol for this synthesis is designed for ease of implementation in both laboratory and pilot plant environments. The process involves the sequential addition of the indole substrate, a selected base catalyst, and the vinyl ester acylating agent into a reactor charged with an appropriate organic solvent. The reaction mixture is then heated to a temperature between 25°C and 130°C, depending on the reactivity of the specific substrates, and maintained for a period of 8 to 36 hours. This broad window allows operators to balance reaction speed with energy consumption. Following the completion of the reaction, indicated by the consumption of the starting material, the solvent is removed under reduced pressure. The resulting residue is typically purified using standard silica gel column chromatography with common eluent systems like petroleum ether and ethyl acetate. Detailed standardized synthesis steps are provided in the guide below.
- Sequentially add indole substrate, alkali catalyst (e.g., triethylamine or potassium carbonate), and carboxylic alkenyl ester into a reactor containing an organic solvent.
- Heat the reaction mixture to a temperature between 25°C and 130°C and maintain stirring for 8 to 36 hours to ensure complete conversion.
- Remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the pure N-acyl indole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this vinyl ester-based acylation technology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material supply chain. Vinyl esters are commodity chemicals produced on a massive scale for the polymer industry, making them significantly cheaper and more readily available than specialized acid halides or anhydrides. This abundance ensures supply continuity and protects against price volatility often seen with finer chemical reagents. Moreover, the elimination of transition metal catalysts removes the need for expensive scavenging resins and rigorous metal testing, which are costly and time-consuming steps in the production of pharmaceutical grades. The overall process intensity is reduced, leading to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the replacement of high-cost acylating agents with low-cost vinyl esters and the use of inexpensive inorganic bases like potassium carbonate or sodium hydroxide. By avoiding the need for cryogenic conditions or strict anhydrous environments required by acid chlorides, energy consumption is significantly lowered. The high atom economy of the reaction means less waste is generated per kilogram of product, reducing disposal costs. Furthermore, the simplified workup procedure reduces labor hours and solvent usage during purification, contributing to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Relying on commodity-grade vinyl esters and common bases mitigates the risk of supply disruptions associated with specialty reagents. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, as the process is not limited by the stability of sensitive reagents. The wide substrate scope demonstrated in the patent data indicates that a single set of operating conditions can be adapted for various substituted indoles, streamlining inventory management. This flexibility enables manufacturers to respond quickly to changing demand for different API precursors without requalifying entirely new synthetic routes or sourcing obscure starting materials.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a significant improvement over traditional acylation. The absence of corrosive hydrogen halide byproducts reduces the burden on scrubber systems and wastewater treatment facilities. The mild temperatures and lack of exothermic hazards associated with acid halide additions enhance process safety, facilitating safer scale-up from gram to ton quantities. The use of environmentally friendlier solvents and the generation of benign carbonyl byproducts align with green chemistry principles, helping companies meet increasingly stringent regulatory standards and sustainability goals without compromising on yield or purity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this acylation technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation. They are intended to provide clarity on the operational parameters and scope of the method for technical decision-makers evaluating this route for potential adoption. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What is the primary advantage of using vinyl esters over acid halides for indole acylation?
A: Using vinyl esters avoids the harsh conditions and low selectivity associated with acid halides. The reaction byproduct is an enol that tautomerizes to a stable carbonyl compound, providing a thermodynamic driving force that prevents reverse reactions and ensures high N-selectivity.
Q: Which catalysts are compatible with this synthesis method?
A: The process is highly versatile and supports a wide range of inexpensive organic and inorganic bases, including triethylamine, pyridine, potassium carbonate, sodium hydroxide, and potassium phosphate, allowing for flexible cost optimization.
Q: Does this method tolerate sensitive functional groups on the indole ring?
A: Yes, the mild reaction conditions exhibit excellent functional group compatibility, successfully accommodating substrates with cyano, methoxy, halogen, and acetyl groups without degradation or side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the potential of this base-catalyzed vinyl ester acylation route and is prepared to leverage its advantages for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acyl indole intermediate delivered meets the highest international standards for pharmaceutical application.
We invite you to collaborate with us to optimize your supply chain for these essential heterocyclic building blocks. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how this innovative chemistry can be integrated into your existing production workflows. Let us help you achieve greater efficiency and cost-effectiveness in your API synthesis programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
