Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
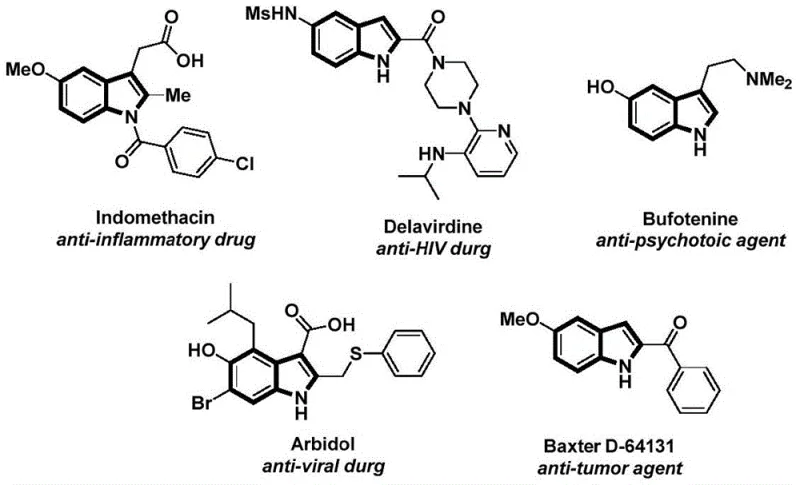
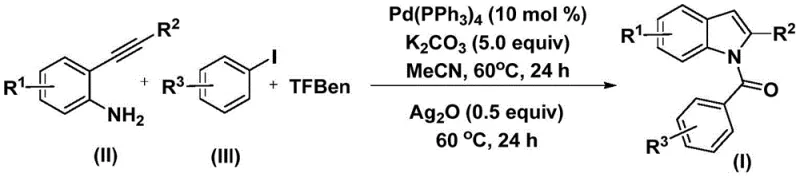
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to anti-HIV drugs such as Delavirdine. As depicted in the diverse pharmacological landscape, the ability to efficiently functionalize the indole nitrogen is critical for drug discovery and process development. Patent CN112898192B introduces a groundbreaking preparation method for N-acyl indole compounds that addresses long-standing challenges in synthetic efficiency and operational safety. This novel approach utilizes a palladium-catalyzed carbonylation cyclization strategy, leveraging 2-alkynyl aniline and aryl iodides as robust starting materials. By replacing hazardous gaseous carbon monoxide with a solid surrogate, this technology offers a safer, more controllable pathway for generating high-value pharmaceutical intermediates, positioning it as a vital tool for modern organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing N-acyl indole scaffolds often rely on direct acylation or classical carbonylation reactions that present significant logistical and safety hurdles. Conventional carbonylation typically requires the handling of carbon monoxide gas, which is highly toxic and necessitates specialized high-pressure equipment and rigorous safety protocols, thereby inflating capital expenditure and operational complexity. Furthermore, many existing methods suffer from poor atom economy, requiring harsh reaction conditions such as extreme temperatures or strong bases that can degrade sensitive functional groups on the substrate. These limitations frequently result in lower yields, complex impurity profiles that are difficult to separate, and a restricted scope of compatible substrates, ultimately hindering the rapid scale-up required for commercial pharmaceutical production.
The Novel Approach
In stark contrast, the methodology disclosed in CN112898192B employs a sophisticated yet operationally simple tandem reaction sequence. By utilizing phenol 1,3,5-tricarboxylate (TFBen) as a solid carbon monoxide substitute, the process eliminates the need for gas cylinders while maintaining high reaction efficiency. The reaction proceeds in a single pot using acetonitrile as the solvent at a mild temperature of 60°C, significantly reducing energy consumption. The introduction of silver oxide in the second stage promotes a smooth cyclization, ensuring high conversion rates even with sterically hindered or electronically diverse substrates. This dual-catalyst system (Pd/Ag) not only streamlines the workflow but also broadens the functional group tolerance, allowing for the synthesis of complex derivatives that were previously difficult to access.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The core of this innovation lies in the elegant orchestration of palladium and silver catalysis to construct the indole framework. The mechanism initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide, generating a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of the TFBen surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amine group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The final transformation is driven by the addition of silver oxide, which facilitates the intramolecular cyclization of the alkyne moiety, aromatizing the system to furnish the target N-acyl indole product with high regioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages for process chemists. The use of a solid CO source ensures a steady, controlled release of carbon monoxide, preventing the formation of side products associated with local concentration spikes of gas. Moreover, the mild thermal conditions (60°C) minimize thermal degradation pathways and polymerization of the alkyne starting material. The high substrate compatibility noted in the patent data suggests that the catalytic cycle is robust against common interfering groups such as halogens, methoxy, and trifluoromethyl substituents. This robustness translates directly to a cleaner crude reaction profile, reducing the burden on downstream purification units and ensuring that the final active pharmaceutical ingredient (API) intermediate meets stringent purity specifications with minimal effort.
How to Synthesize N-Acyl Indole Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for generating these valuable heterocycles. The process is designed to be user-friendly, requiring standard laboratory glassware and readily available reagents. The reaction is typically conducted in acetonitrile, which serves as an effective polar aprotic solvent to dissolve both the organic substrates and the inorganic bases. The sequential addition of reagents—first the palladium system for carbonylation, followed by silver oxide for cyclization—allows for precise control over the reaction trajectory. For detailed operational parameters and stoichiometry, refer to the standardized guide below.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, carbon monoxide substitute (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent like acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to induce cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic benefits beyond mere chemical elegance. The shift from gaseous reagents to solid surrogates fundamentally alters the risk profile and cost structure of the manufacturing process. By removing the dependency on specialized high-pressure reactors and toxic gas handling infrastructure, facilities can repurpose existing standard batch reactors for production, leading to substantial capital cost avoidance. Furthermore, the simplicity of the workup procedure, which involves basic filtration and chromatography, reduces the consumption of solvents and silica gel, directly lowering the variable costs associated with each production batch.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of cheap and commercially available starting materials such as aryl iodides and 2-alkynyl anilines. The elimination of expensive high-pressure equipment maintenance and the reduced need for specialized safety training for gas handling contribute to a leaner operational budget. Additionally, the high reaction efficiency and yield reported across various substrates mean less raw material is wasted, optimizing the overall material throughput and maximizing the return on investment for every kilogram of precursor purchased.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the accessibility of the reagents. Unlike specialized gaseous reagents that may face logistics bottlenecks or regulatory shipping restrictions, the solid components of this reaction (TFBen, Pd catalyst, Ag2O) are stable, easy to store, and widely sourced from global chemical suppliers. This stability ensures consistent production schedules without the risk of interruptions due to reagent shortages. The robust nature of the reaction also implies a lower rate of batch failures, guaranteeing a steady flow of intermediates to downstream formulation teams.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is straightforward due to the absence of exothermic gas evolution risks and the use of mild temperatures. The environmental footprint is minimized by avoiding the release of carbon monoxide into the atmosphere and reducing the generation of hazardous waste associated with harsh acidic or basic workups. This alignment with green chemistry principles simplifies regulatory compliance and waste disposal permitting, facilitating faster approval for commercial manufacturing sites and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acyl indole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing pipelines.
Q: What are the key advantages of this N-acyl indole synthesis method over traditional carbonylation?
A: This method avoids the use of toxic and high-pressure carbon monoxide gas by utilizing a solid CO surrogate (TFBen). It operates under mild conditions (60°C) and demonstrates excellent substrate compatibility with various functional groups, simplifying the purification process and enhancing operational safety.
Q: What is the role of Silver Oxide (Ag2O) in this reaction mechanism?
A: Silver oxide acts as a crucial promoter for the cyclization step. After the initial palladium-catalyzed formation of the amide intermediate, Ag2O facilitates the intramolecular cyclization to form the final N-acyl indole core structure efficiently.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable due to its simple one-pot operation, the use of commercially available starting materials, and the avoidance of hazardous gas handling. The mild reaction temperatures and standard workup procedures make it ideal for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has extensively evaluated the palladium-catalyzed carbonylation route described in CN112898192B and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acyl indole intermediate we deliver adheres to the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules, ensuring a secure and cost-effective supply chain for your critical pharmaceutical intermediates.
