Advanced Biginelli Reaction Technology for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen heterocyclic compounds, particularly dihydropyrimidinones, which serve as critical scaffolds for calcium channel blockers and antitumor agents. Patent CN112812066A introduces a transformative synthesis method that addresses long-standing inefficiencies in the Biginelli reaction by utilizing a deep eutectic solvent (DES) system composed of choline chloride and ethylene glycol or glycerol. This innovation replaces hazardous volatile organic compounds with a stable, green solvent medium while employing p-toluenesulfonic acid as a cost-effective protonic acid catalyst. The technical breakthrough lies in the ability to achieve conversion rates exceeding 99% at a mild temperature of 50°C, coupled with a simplified workup procedure that relies on simple filtration rather than complex purification. For R&D directors and procurement specialists, this patent represents a significant opportunity to enhance the sustainability and economic viability of producing high-purity pharmaceutical intermediates without compromising on yield or structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of dihydropyrimidinones often relies on Lewis acid catalysts such as copper trifluorosulfonate or harsh protonic acids in volatile organic solvents, which present substantial drawbacks for industrial application. While Lewis acids can offer high yields, they introduce expensive transition metals that are difficult to remove completely from the final product, posing severe risks for residual metal impurities in pharmaceutical grades. Furthermore, conventional processes frequently require high temperatures and extensive downstream purification steps like column chromatography to isolate the product from the reaction mixture, leading to significant material loss and increased operational costs. The use of volatile organic solvents also raises environmental compliance issues and safety concerns regarding worker exposure and waste disposal, making these legacy methods increasingly unsustainable for modern green chemistry standards and large-scale commercial manufacturing requirements.
The Novel Approach
The novel approach detailed in the patent utilizes a deep eutectic solvent system that fundamentally alters the reaction environment to favor product precipitation and catalyst stability. By employing choline chloride mixed with ethylene glycol or glycerol, the reaction medium provides a unique hydrogen-bonding network that enhances the protonation degree of the reactants, thereby accelerating the condensation reaction without the need for expensive metal catalysts. This method allows the dihydropyrimidinone product to precipitate directly from the reaction mixture as it forms, enabling isolation through simple filtration while leaving the catalyst and unreacted starting materials dissolved in the solvent for reuse. This shift from homogeneous catalysis with difficult separation to a pseudo-heterogeneous system drastically simplifies the workflow, reduces solvent consumption, and eliminates the need for energy-intensive distillation or chromatographic purification steps.
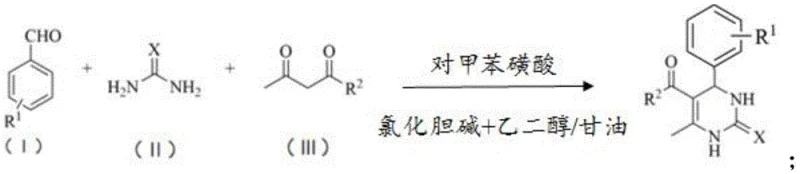
Mechanistic Insights into PTSA-Catalyzed Biginelli Condensation
The mechanistic advantage of this synthesis lies in the synergistic interaction between the p-toluenesulfonic acid catalyst and the deep eutectic solvent components. The p-toluenesulfonic acid acts as a strong proton donor, activating the carbonyl group of the aldehyde and the beta-keto ester to facilitate nucleophilic attack by the urea or thiourea. Simultaneously, the choline chloride-based solvent stabilizes the transition states through extensive hydrogen bonding, which lowers the activation energy required for the cyclization step. This dual activation mechanism ensures that the reaction proceeds efficiently at 50°C, a temperature significantly lower than typical thermal requirements for Biginelli reactions, thereby minimizing side reactions and thermal decomposition of sensitive functional groups on the aromatic rings.
Impurity control is inherently managed by the solubility characteristics of the product within the deep eutectic solvent matrix. As the dihydropyrimidinone compound forms, its low solubility in the DES causes it to crystallize out of the solution, effectively driving the equilibrium towards completion and preventing further side reactions or degradation. This precipitation mechanism ensures that the isolated filter cake possesses high purity, often exceeding 99% without additional recrystallization, as confirmed by NMR data showing no detectable impurities. For quality control teams, this means a more consistent impurity profile and reduced risk of genotoxic impurities or heavy metal contamination that are commonly associated with transition metal catalyzed processes in traditional synthetic routes.
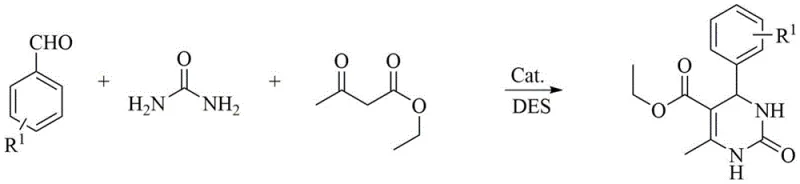
How to Synthesize Dihydropyrimidinone Efficiently
Implementing this synthesis route requires precise control over the molar ratios of the deep eutectic solvent components and the reactants to ensure optimal reaction kinetics and yield. The process begins with the preparation of the solvent by mixing choline chloride with ethylene glycol in a 1:2 molar ratio or glycerol in a 3:4 ratio, followed by the addition of benzaldehyde derivatives, ethyl acetoacetate, and urea. The reaction is catalyzed by p-toluenesulfonic acid at a molar ratio of 5 to 20:1 relative to the substrate and maintained at 50°C with stirring for 3 to 20 hours depending on the specific substituents. Detailed standardized synthesis steps see the guide below.
- Prepare the deep eutectic solvent by mixing choline chloride with ethylene glycol or glycerol in specific molar ratios.
- Combine benzaldehyde derivatives, ethyl acetoacetate, and urea in the solvent with p-toluenesulfonic acid catalyst.
- Heat the mixture to 50°C, stir for 3-20 hours, and isolate the product via simple filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers profound advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediate manufacturing. The elimination of expensive transition metal catalysts removes the need for costly scavenging resins or complex purification protocols, directly lowering the bill of materials and processing time. Additionally, the ability to recycle the solvent and catalyst system for at least 15 cycles without significant loss in performance drastically reduces raw material consumption and waste disposal costs, contributing to substantial long-term operational savings. This efficiency translates into a more resilient supply chain where production is less dependent on volatile raw material markets and more focused on sustainable, repeatable processes that ensure consistent availability of critical intermediates.
- Cost Reduction in Manufacturing: The replacement of Lewis acid catalysts with p-toluenesulfonic acid eliminates the expense associated with precious or transition metals, which are subject to significant price fluctuations and supply constraints. Furthermore, the simplified isolation process via filtration removes the need for large volumes of organic solvents used in extraction and chromatography, significantly reducing solvent procurement and recovery costs. The overall process intensity is lowered, meaning less energy is required for heating and cooling, which cumulatively results in a drastically simplified cost structure for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the deep eutectic solvent system ensures that production can continue even if specific batches of reactants vary slightly, as the solvent buffers the reaction conditions effectively. The recyclability of the catalyst and solvent means that the facility does not need to constantly replenish these critical processing aids, reducing the frequency of procurement orders and minimizing the risk of supply disruptions. This stability is crucial for maintaining continuous manufacturing operations and meeting strict delivery timelines for downstream API synthesis without unexpected delays caused by material shortages.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward because it avoids the handling hazards associated with volatile organic solvents and toxic metal catalysts. The green nature of the choline chloride-based solvent aligns with increasingly stringent environmental regulations, reducing the regulatory burden and potential fines associated with hazardous waste generation. The ability to operate at mild temperatures of 50°C also reduces the engineering requirements for reactor cooling and heating systems, making it easier to adapt existing infrastructure for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield consistency, recycling capabilities, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios and ensuring alignment with quality and sustainability goals.
Q: How does the DES solvent system improve product purity compared to traditional methods?
A: The deep eutectic solvent system facilitates product precipitation during the reaction, allowing for simple filtration that separates the product from the catalyst and unreacted materials without needing column chromatography, resulting in purity exceeding 99%.
Q: Can the catalyst and solvent be recycled in this synthesis process?
A: Yes, the p-toluenesulfonic acid catalyst and the choline chloride-based solvent remain in the filtrate after product separation and can be reused for at least 15 cycles without significant loss in conversion efficiency.
Q: What are the temperature conditions required for this Biginelli reaction?
A: The reaction proceeds efficiently at a mild temperature of 50°C, which is significantly lower than many conventional high-temperature protocols, reducing energy consumption and thermal degradation risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyrimidinone Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthesis technology to support your drug development and commercial manufacturing needs. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch of dihydropyrimidinone intermediate meets the highest global standards, providing you with a reliable pharmaceutical intermediates supplier partner who understands the critical nature of your supply chain.
We invite you to engage with our technical procurement team to discuss how this green synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this DES-based protocol. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and cost-efficiency of your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
