Advanced Green Synthesis of Dihydropyrimidinones for Pharmaceutical Intermediates Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing critical heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN112812066B, which discloses a novel synthetic method for dihydropyrimidinone compounds. This technology represents a paradigm shift from traditional harsh conditions to a greener, more economical approach utilizing Deep Eutectic Solvents (DES). By employing a mixture of choline chloride with ethylene glycol or glycerol as the reaction medium, and p-toluenesulfonic acid as a benign catalyst, this method achieves exceptional conversion rates under mild thermal conditions. For R&D directors and procurement specialists alike, this patent offers a compelling solution for producing high-purity pharmaceutical intermediates while addressing growing environmental compliance pressures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydropyrimidinones via the Biginelli reaction has relied heavily on Lewis acid catalysts such as copper triflate or harsh protonic acids in volatile organic solvents. While Lewis acids can drive the reaction, they present severe downstream processing challenges, particularly regarding the removal of transition metal residues which are strictly regulated in active pharmaceutical ingredients (APIs). Furthermore, traditional methods often require high temperatures and generate significant amounts of hazardous waste, complicating the supply chain and increasing the cost of waste disposal. The difficulty in separating the catalyst from the final product often necessitates complex purification steps like column chromatography, which are not feasible for large-scale commercial production.
The Novel Approach
The methodology described in patent CN112812066B overcomes these hurdles by introducing a recyclable Deep Eutectic Solvent system combined with p-toluenesulfonic acid. This approach leverages the unique hydrogen-bonding network of the DES to enhance the protonation of reactants, thereby accelerating the condensation reaction at a mild temperature of 50°C. Unlike traditional methods where the product remains dissolved requiring extraction, this novel process allows the product to precipitate directly from the reaction mixture. This physical property change simplifies the workup to a mere filtration step, eliminating the need for energy-intensive distillation or solvent extraction processes.
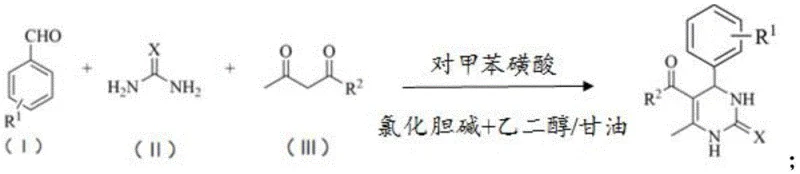
Mechanistic Insights into DES-Promoted Biginelli Condensation
The core innovation lies in the synergistic effect between the p-toluenesulfonic acid catalyst and the choline chloride-based solvent. The deep eutectic solvent is not merely an inert medium; it actively participates in the reaction mechanism by stabilizing the transition states through extensive hydrogen bonding. This interaction increases the effective acidity of the protonic catalyst, facilitating the formation of the N-acyliminium ion intermediate, which is the rate-determining step in the Biginelli reaction. Consequently, the reaction proceeds with high efficiency even at lower temperatures, minimizing side reactions such as polymerization or decomposition of sensitive functional groups on the aromatic aldehyde substrates.
Furthermore, the solubility characteristics of the DES play a crucial role in impurity control and product isolation. As the reaction progresses, the dihydropyrimidinone product reaches its saturation point within the DES matrix and crystallizes out. This spontaneous precipitation acts as a driving force for the reaction equilibrium, pushing the conversion towards completion. The unreacted starting materials and the catalyst remain dissolved in the mother liquor, ensuring that the isolated solid is of exceptionally high purity. This mechanism effectively decouples the reaction kinetics from the separation thermodynamics, providing a robust platform for synthesizing diverse derivatives.
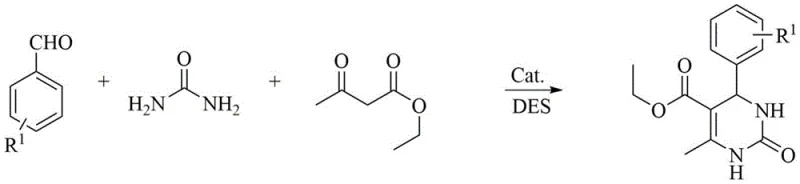
The versatility of this catalytic system is demonstrated by its compatibility with various substrates, including those containing electron-withdrawing or electron-donating groups. Whether utilizing ethyl acetoacetate to form ester-substituted products or acetylacetone for ketone-substituted analogs, the system maintains high yields. Additionally, the method accommodates the use of thiourea to synthesize dihydropyrimidine-2-thiones, expanding the chemical space accessible to medicinal chemists. The ability to switch between urea and thiourea without altering the fundamental process parameters highlights the robustness of this green chemistry platform for generating diverse chemical libraries.
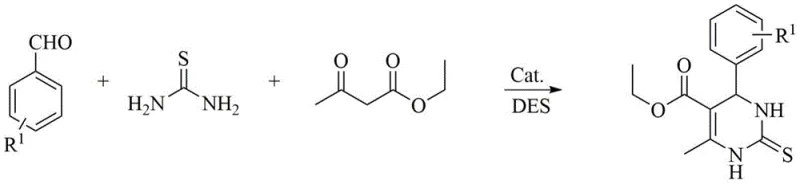
How to Synthesize Dihydropyrimidinone Efficiently
To implement this synthesis effectively, one must carefully control the molar ratios of the deep eutectic solvent components and the catalyst loading. The process begins with the preparation of the solvent mixture, followed by the addition of the three components: the aldehyde, the beta-keto ester, and the urea source. Detailed standard operating procedures regarding temperature ramping, stirring speeds, and filtration techniques are critical for maximizing yield and ensuring batch-to-batch consistency. For a comprehensive guide on the exact experimental parameters and troubleshooting tips, please refer to the standardized synthesis steps outlined below.
- Prepare the Deep Eutectic Solvent (DES) by mixing choline chloride with ethylene glycol or glycerol in specific molar ratios.
- Dissolve benzaldehyde derivatives, ethyl acetoacetate (or acetylacetone), and urea (or thiourea) in the DES solvent.
- Add p-toluenesulfonic acid catalyst and stir at 50°C for 3 to 20 hours, then filter to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology addresses several critical pain points in the supply chain for pharmaceutical intermediates. The elimination of transition metal catalysts removes the need for expensive and time-consuming metal scavenging steps, which are often bottlenecks in API manufacturing. By replacing volatile organic solvents with non-volatile, biodegradable deep eutectic solvents, the process significantly reduces the environmental footprint and lowers the costs associated with solvent recovery and waste treatment. This aligns perfectly with the industry's shift towards greener manufacturing practices and stricter regulatory compliance.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the reusability of the reaction medium. Since the catalyst and solvent remain in the filtrate after product isolation, they can be directly reused for subsequent batches without complex regeneration. This circular economy approach drastically reduces the consumption of raw materials and minimizes waste generation. Furthermore, the simplicity of the workup—requiring only filtration and drying—lowers labor costs and reduces the demand for specialized purification equipment, leading to substantial overall cost savings in dihydropyrimidinone manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. The mild temperature requirement of 50°C reduces energy consumption and mitigates safety risks associated with high-pressure or high-temperature reactors. Additionally, the high purity of the crude product means that lead times for quality control testing and release are shortened. Suppliers can respond more quickly to demand fluctuations because the production cycle is streamlined, ensuring a steady flow of high-quality intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: Scaling up this process is inherently safer and more straightforward than traditional methods. The absence of toxic volatile solvents simplifies the engineering controls required for large-scale production, making it easier to transition from pilot plant to commercial scale. The non-toxic nature of the choline chloride-based solvents ensures that the process meets stringent environmental regulations regarding emissions and effluent discharge. This compliance advantage future-proofs the manufacturing process against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this green synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of the method for industrial partners.
Q: What are the advantages of using Deep Eutectic Solvents (DES) over traditional organic solvents in Biginelli reactions?
A: DES systems, such as choline chloride mixed with ethylene glycol, offer superior stability and low viscosity compared to traditional solvents. They facilitate higher protonation degrees through hydrogen bonding effects, significantly improving reaction yields without the need for toxic volatile organic compounds.
Q: Can the catalyst and solvent system be recycled in this synthesis process?
A: Yes, the patent data indicates that the p-toluenesulfonic acid catalyst and the DES solvent can be reused for at least 15 cycles. The product precipitates out, allowing the catalyst and unreacted materials to remain in the filtrate for subsequent batches, drastically reducing waste.
Q: Is purification required after the reaction is complete?
A: No complex purification is typically needed. The product forms a precipitate during the reaction which can be separated by simple filtration. The resulting filter cake, once dried, exhibits high purity (>99%) without requiring column chromatography or recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyrimidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the one described in CN112812066B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards for pharmaceutical applications.
We invite procurement leaders and R&D teams to collaborate with us to leverage this advanced synthesis route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both cost-efficiency and sustainability.
