Revolutionizing Benzofuran Production: A One-Step Lewis Acid Catalyzed Route for Commercial Scale-Up
The chemical industry is constantly seeking more efficient pathways to synthesize high-value heterocyclic scaffolds, and the recent disclosure in patent CN109503531B offers a transformative approach to producing benzofuran compounds. This intellectual property details a robust preparation method that bypasses the cumbersome multi-step sequences traditionally associated with benzofuran synthesis. By leveraging a direct cyclization strategy using furan compounds as the sole carbon source, the process achieves remarkable selectivity rates reaching up to 99 percent under mild thermal conditions. The methodology utilizes an aqueous acetic acid solvent system combined with Lewis acid catalysts, creating a reaction environment that is both economically viable and environmentally considerate. For R&D directors and process chemists, this represents a significant leap forward in accessing complex aromatic systems without the burden of harsh reagents or difficult purification protocols. The ability to tune the final product structure by simply varying the substitution pattern on the starting furan ring provides a versatile platform for generating diverse libraries of bioactive intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzofuran cores has been plagued by inefficiencies that drive up costs and extend lead times for pharmaceutical and agrochemical developers. Traditional routes often rely on the condensation of phenols with 2-haloketones, a pathway that necessitates the procurement of specialized and often expensive halogenated starting materials which are not always readily available in bulk quantities. Furthermore, these classical methods typically involve multiple discrete reaction steps, including initial ether formation followed by acid-catalyzed dehydration, each step introducing potential yield losses and requiring intermediate isolation. Another common approach involves starting from salicylaldehyde and substituted bromoacetates, which similarly suffers from operational complexity and high process costs due to the stoichiometric generation of salt byproducts. These legacy processes often demand severe reaction conditions and rigorous safety controls, creating bottlenecks in supply chains that struggle to meet the growing demand for high-purity heterocyclic building blocks. Consequently, manufacturers face significant challenges in scaling these routes while maintaining competitive pricing and consistent quality standards.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent data utilizes a direct, one-step cyclization of furan derivatives to construct the benzofuran skeleton. This method eliminates the need for pre-functionalized phenolic precursors, instead relying on abundant furan compounds that can be sourced from renewable biomass feedstocks. The reaction proceeds smoothly in an acetic acid aqueous solution, which acts as both a solvent and a proton source, facilitating the rearrangement and aromatization required to form the fused ring system. By operating at moderate temperatures ranging from 80°C to 160°C, the process avoids the energy-intensive conditions often required for traditional cyclizations. The simplicity of charging the furan substrate, acetic acid, and a Lewis acid catalyst into a single vessel drastically reduces the operational footprint and minimizes the risk of human error during manufacturing. This streamlined workflow not only accelerates the timeline from raw material to finished product but also enhances the overall atom economy of the synthesis, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this technological breakthrough lies in the precise activation of the furan ring by Lewis acid species within a protic solvent medium. When a furan compound, such as those represented by the general formulas in the patent, is introduced to the reaction system, the Lewis acid catalyst coordinates with the oxygen atom or the pi-system of the furan ring. 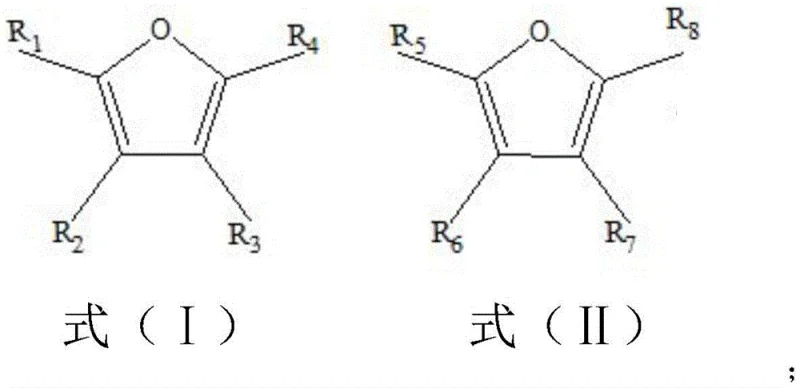 This coordination increases the electrophilicity of the furan ring, making it susceptible to intramolecular attack or dimerization pathways that lead to ring expansion and subsequent aromatization. The presence of water in the acetic acid solvent plays a critical role in modulating the acidity and solubility of the catalyst, ensuring a homogeneous reaction phase that promotes consistent kinetics throughout the batch. The specific selection of catalysts like aluminum chloride or ferric chloride allows for fine-tuning the reaction rate, preventing over-reaction or polymerization which could otherwise degrade the yield. Understanding this mechanistic nuance is vital for process engineers aiming to optimize the reaction parameters for different substituted furan substrates.
This coordination increases the electrophilicity of the furan ring, making it susceptible to intramolecular attack or dimerization pathways that lead to ring expansion and subsequent aromatization. The presence of water in the acetic acid solvent plays a critical role in modulating the acidity and solubility of the catalyst, ensuring a homogeneous reaction phase that promotes consistent kinetics throughout the batch. The specific selection of catalysts like aluminum chloride or ferric chloride allows for fine-tuning the reaction rate, preventing over-reaction or polymerization which could otherwise degrade the yield. Understanding this mechanistic nuance is vital for process engineers aiming to optimize the reaction parameters for different substituted furan substrates.
Furthermore, the structural versatility of this method is evident in the wide range of products accessible through simple variations in the starting material. As illustrated by the product formulas, the substituents on the initial furan ring are largely retained or rearranged in a predictable manner to form the final benzofuran structure. 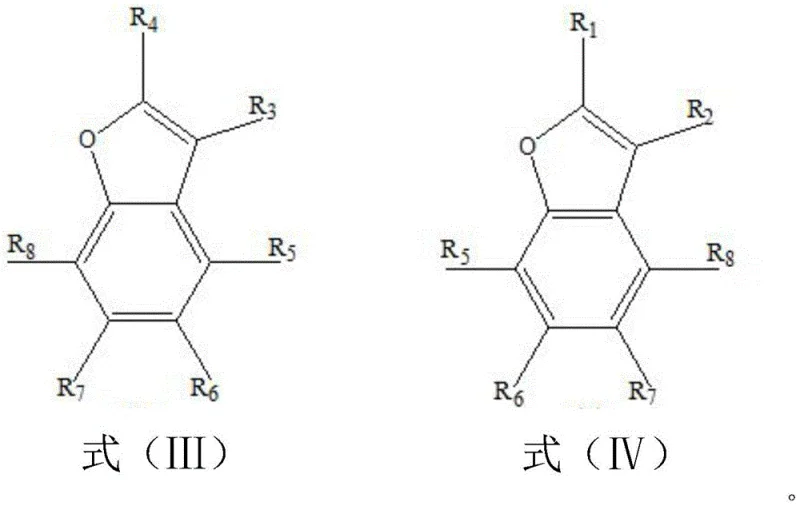 For instance, when specific hydrogen atoms on the furan ring are replaced by alkyl or thiol groups, the resulting benzofuran compounds exhibit corresponding functionalization at the 2, 4, or 7 positions. This predictability allows chemists to design synthetic routes for specific target molecules with high confidence in the regioselectivity of the outcome. The high selectivity reported, often exceeding 99 percent for the parent benzofuran, suggests that the transition state favors the formation of the thermodynamically stable aromatic system over competing side reactions. This level of control is essential for minimizing impurity profiles, thereby reducing the burden on downstream purification units and ensuring the final API intermediate meets stringent regulatory specifications.
For instance, when specific hydrogen atoms on the furan ring are replaced by alkyl or thiol groups, the resulting benzofuran compounds exhibit corresponding functionalization at the 2, 4, or 7 positions. This predictability allows chemists to design synthetic routes for specific target molecules with high confidence in the regioselectivity of the outcome. The high selectivity reported, often exceeding 99 percent for the parent benzofuran, suggests that the transition state favors the formation of the thermodynamically stable aromatic system over competing side reactions. This level of control is essential for minimizing impurity profiles, thereby reducing the burden on downstream purification units and ensuring the final API intermediate meets stringent regulatory specifications.
How to Synthesize Benzofuran Compounds Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction medium and the precise control of thermal parameters to maximize conversion. The process begins with the formulation of an aqueous acetic acid solution, where the ratio of acid to water can be adjusted between 1:0 and 1:5 to optimize catalyst solubility and reaction homogeneity. Once the solvent system is ready, the furan substrate and the Lewis acid catalyst are charged into the reactor, with the catalyst loading typically maintained between 1 percent and 8 percent by mass to balance activity and cost. The mixture is then heated to a target temperature within the 80°C to 160°C window and held for a duration ranging from 0.5 to 24 hours, depending on the specific reactivity of the furan derivative. Following the reaction completion, the crude mixture undergoes a straightforward separation and purification sequence, often involving rectification to recover the valuable acetic acid solvent and isolate the pure benzofuran product. Detailed standardized operating procedures for specific substrates are outlined in the technical guide below.
- Prepare an aqueous acetic acid solution with a specific volume ratio of acetic acid to water ranging from 1: 0 to 1:5 to serve as the reaction medium.
- Charge the reaction vessel with the furan compound substrate and a Lewis acid catalyst such as aluminum chloride, ensuring a catalyst mass concentration between 1% and 8%.
- Heat the mixture to a temperature between 80°C and 160°C for a duration of 0.5 to 24 hours, followed by separation and purification via rectification to isolate the high-purity benzofuran product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method presents a compelling value proposition centered on cost efficiency and supply security. By shifting away from complex multi-step sequences that rely on scarce halogenated ketones, manufacturers can significantly reduce their exposure to volatile raw material markets. The ability to utilize biomass-derived furans, which are increasingly produced at scale for the bio-fuel and chemical sectors, ensures a stable and sustainable supply chain foundation. Moreover, the simplification of the process from multiple steps to a single pot reaction drastically cuts down on labor costs, equipment utilization time, and waste disposal expenses associated with intermediate handling. These operational efficiencies translate directly into a more competitive cost structure for the final benzofuran intermediates, allowing downstream partners to improve their margins without compromising on quality. The robustness of the reaction conditions also implies a lower risk of batch failures, further enhancing the reliability of supply for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of expensive and hard-to-source 2-haloketones or salicylaldehyde derivatives removes a major cost driver from the bill of materials. Additionally, the use of acetic acid as a solvent, which can be easily recovered and recycled via distillation, minimizes solvent purchase and waste treatment costs. The high selectivity of the reaction means that less raw material is wasted on byproducts, effectively increasing the yield per kilogram of input and lowering the overall unit cost of production. This economic advantage is compounded by the reduced need for complex purification steps, as the crude product profile is much cleaner than that of traditional methods.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit immensely from the use of furan-based feedstocks, which are derived from renewable agricultural resources rather than petrochemical chains prone to geopolitical instability. The simplified one-step process reduces the number of critical control points in the manufacturing workflow, thereby decreasing the likelihood of delays caused by equipment bottlenecks or intermediate quality issues. This streamlined operation enables manufacturers to respond more agilely to fluctuations in market demand, ensuring that customers receive their orders within tighter lead times. The scalability of the process from gram to ton scale further guarantees that supply can be ramped up quickly to support clinical trials or commercial launches without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of hazardous halogenated solvents make this process inherently safer and easier to scale in standard stainless steel reactors. From an environmental perspective, the use of aqueous acetic acid reduces the generation of toxic organic waste streams, simplifying compliance with increasingly stringent environmental regulations. The ability to recover and reuse the solvent system contributes to a lower carbon footprint for the manufacturing site, aligning with corporate sustainability goals. Furthermore, the high atom economy of the direct cyclization minimizes the E-factor of the process, making it an attractive option for companies looking to green their supply chains while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzofuran synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production portfolios. The responses cover aspects ranging from catalyst selection to product purity and scalability concerns.
Q: What are the primary advantages of this new benzofuran synthesis method over traditional routes?
A: Unlike conventional methods that require multiple steps and expensive halogenated ketones, this patent describes a direct one-step cyclization using readily available biomass-derived furans. This significantly simplifies the process operation and reduces raw material costs while achieving selectivity as high as 99%.
Q: Which catalysts are most effective for this transformation according to the patent data?
A: The patent identifies several effective Lewis acid catalysts, including aluminum chloride, boron trifluoride, ferric chloride, and antimony pentafluoride. Aluminum chloride is highlighted in multiple examples for its ability to facilitate the reaction at mild temperatures between 80°C and 160°C.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for industrial application. It utilizes simple raw materials, operates under relatively mild conditions, and employs acetic acid aqueous solutions which are easy to recover and recycle via rectification, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Lewis acid-catalyzed route for producing high-value benzofuran intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the lab bench to full-scale manufacturing. Our facilities are equipped with state-of-the-art reactors capable of handling the mild thermal conditions and corrosive nature of Lewis acid catalysts safely and efficiently. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzofuran compounds meets the highest international standards for pharmaceutical and fine chemical applications. Our team of expert process chemists is ready to optimize the reaction parameters specifically for your target molecule, maximizing yield and minimizing impurities.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined route. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your unique supply chain requirements. Let us help you secure a reliable supply of high-purity benzofurans while driving down your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
