Revolutionizing Cilostazol Production: A Green Aqueous Route for Commercial Scale-Up
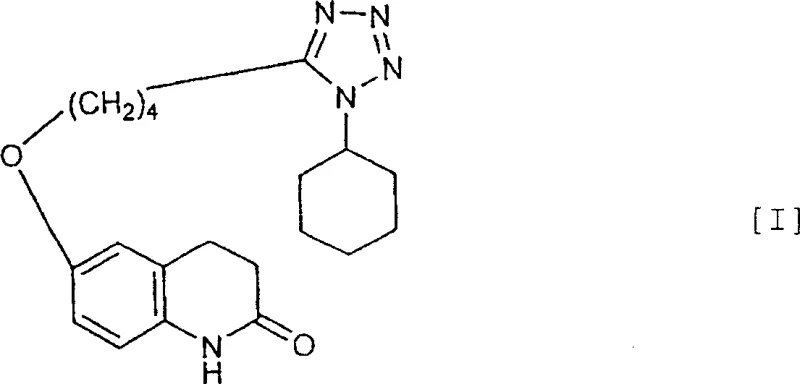
The pharmaceutical industry is currently undergoing a paradigm shift towards greener, more sustainable manufacturing processes, particularly for high-volume cardiovascular agents like Cilostazol. A pivotal advancement in this domain is detailed in Chinese Patent CN1257902C, which discloses a novel production method achieving exceptional yield and purity through an aqueous reaction system. This innovation addresses long-standing challenges in the synthesis of 6-[4-(1-cyclohexyl-1H-tetrazol-5-yl)butoxy]-3,4-dihydroquinolone, traditionally plagued by low conversion rates and difficult purification protocols. By replacing hazardous organic solvents with water and utilizing specific inorganic base ratios, this technology offers a robust pathway for producing pharmaceutical-grade intermediates. For R&D directors and procurement specialists seeking a reliable cilostazol supplier, understanding the mechanistic advantages of this aqueous route is critical for securing a stable, cost-effective supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cilostazol has relied heavily on organic solvents and phase transfer catalysts, methodologies that introduce significant operational and environmental burdens. Traditional protocols, such as those referenced in JP-A-56-49378, typically involve reacting the quinolone derivative with the tetrazole derivative in non-aqueous media. A major drawback of these legacy methods is the competitive nucleophilic attack that occurs not only at the desired phenolic oxygen but also at the nitrogen atom of the quinolone ring. This side reaction generates N-1 alkylated impurities that are structurally similar to the target molecule, making them notoriously difficult to remove via standard crystallization. Consequently, manufacturers often face yields ranging merely from 50% to 74%, necessitating complex, multi-step purification processes that erode profit margins and extend lead times. Furthermore, the reliance on volatile organic solvents creates substantial safety hazards and requires expensive waste treatment infrastructure to meet increasingly stringent environmental regulations.
The Novel Approach
In stark contrast, the method disclosed in CN1257902C leverages water as the primary reaction medium, fundamentally altering the reaction kinetics to favor the desired O-alkylation over N-alkylation. By strictly controlling the water-to-substrate weight ratio between 3 to 7 times and employing a specific molar excess of inorganic bases (1 to 6 mol/mol), the process achieves a dramatic suppression of byproduct formation. This aqueous environment stabilizes the transition state for the phenolic substitution while rendering the tetrazole derivative sufficiently reactive without decomposition. The result is a streamlined process that delivers yields exceeding 90% with purity levels consistently above 99.5%, effectively eliminating the need for column chromatography. This approach not only simplifies the downstream processing but also drastically reduces the environmental footprint, positioning it as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Aqueous Nucleophilic Substitution
The success of this aqueous synthesis hinges on the precise modulation of nucleophilicity and solubility dynamics within the reaction matrix. In conventional organic solvents, the lipophilic nature of the tetrazole derivative facilitates its interaction with multiple nucleophilic sites on the quinolone scaffold. However, in the aqueous system described, the solvation shell formed around the inorganic base and the substrate creates a unique microenvironment. The hydroxide or carbonate ions deprotonate the phenolic hydroxyl group of the 6-hydroxy-3,4-dihydroquinolone to form a phenoxide anion, which is a potent nucleophile. Crucially, the water solvent restricts the conformational freedom of the reactants, sterically hindering the approach of the bulky tetrazole alkyl halide to the less accessible N-1 position of the quinolone ring. This steric and electronic discrimination ensures that the alkylation occurs almost exclusively at the oxygen atom, thereby preventing the formation of the persistent N-1 impurity.
Furthermore, the patent highlights the importance of mechanical agitation through a continuous disperser, which plays a vital role in maintaining reaction homogeneity. Since the tetrazole derivative has limited solubility in water, the reaction occurs at the interface of the solid or suspended organic phase and the aqueous phase. The continuous circulation and pulverization of the reaction mixture prevent the agglomeration of the forming Cilostazol crystals. If these crystals were to clump together, they would trap unreacted starting materials and impurities within the lattice, compromising the final purity. By keeping the particle size small and uniform throughout the heating cycle (typically 50°C to 150°C), the process ensures that the product precipitates in a highly pure crystalline form, ready for simple filtration and washing. This mechanistic control is what allows the process to achieve high-purity cilostazol without the need for recrystallization from toxic solvents.
How to Synthesize Cilostazol Efficiently
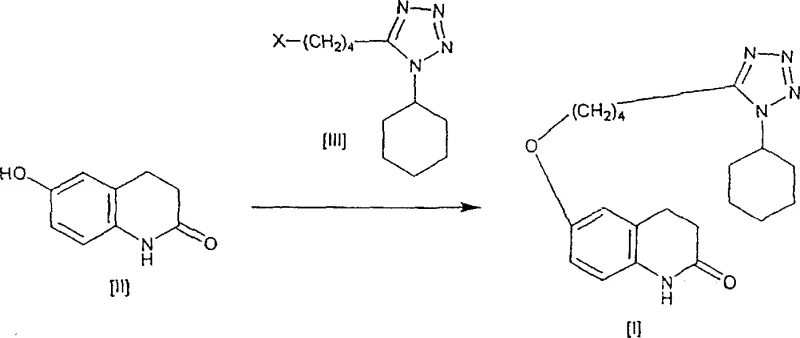
The implementation of this aqueous alkylation strategy requires careful attention to stoichiometry and thermal management to replicate the high yields observed in the patent examples. The process begins with the suspension of the quinolone precursor and the tetrazole alkylating agent in a controlled volume of water, followed by the addition of a mixed base system, often comprising potassium carbonate and sodium hydroxide. The reaction is then heated under reflux conditions while being continuously circulated through a disperser to maintain a homogeneous slurry. After the reaction reaches completion, typically indicated by HPLC analysis, the product is isolated via cooling and filtration. The crude solid is then subjected to a sequential washing protocol using water and lower alcohols like methanol to remove residual salts and unreacted materials. For a detailed breakdown of the specific temperatures, mixing rates, and work-up procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining 6-hydroxy-3,4-dihydroquinolone and 1-cyclohexyl-5-(4-chlorobutyl)-1H-tetrazole in water with an inorganic base.
- Heat the mixture to 50-150°C while circulating through a continuous disperser to prevent crystal aggregation and ensure uniform reaction kinetics.
- Isolate the crude product via filtration, followed by washing with water and alcohol, and final drying to achieve pharmaceutical-grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous synthesis route represents a significant opportunity to optimize total landed costs and mitigate supply risks. The elimination of organic solvents removes the need for complex solvent recovery systems, which are capital-intensive to install and maintain. This simplification of the plant infrastructure directly translates to lower overheads and reduced energy consumption, as there is no need for high-vacuum distillation to recover volatile organics. Additionally, the use of water as a solvent significantly lowers the fire hazard rating of the manufacturing facility, potentially reducing insurance premiums and easing regulatory compliance burdens related to VOC emissions. These factors combined create a more resilient and economically efficient production model.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the drastic reduction in raw material waste and solvent handling expenses. Traditional methods often require large volumes of expensive polar aprotic solvents like DMF or DMSO, which must be meticulously removed to meet residual solvent limits in the final API. By switching to water, the cost of solvent purchase, storage, and disposal is virtually eliminated. Moreover, the high selectivity of the reaction means that the yield loss due to byproduct formation is minimized, maximizing the output per kilogram of starting material. This efficiency gain allows for a substantial reduction in the cost of goods sold (COGS), providing a competitive pricing advantage in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on water as a solvent also mitigates risks associated with the supply volatility of specialty organic solvents. Organic solvents are often petrochemical derivatives subject to price fluctuations and supply disruptions. Water, being universally available and inexpensive, ensures that production can continue uninterrupted regardless of external market conditions. Furthermore, the simplified purification process reduces the turnaround time for each batch. With fewer unit operations required—specifically the removal of solvent stripping and complex chromatography steps—manufacturers can increase their batch frequency. This agility enables a more responsive supply chain capable of meeting sudden spikes in demand for cardiovascular medications, ensuring continuity of supply for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns perfectly with the growing corporate mandates for green chemistry. The aqueous effluent generated is far easier to treat than organic waste streams, reducing the load on wastewater treatment plants and minimizing the environmental impact of the manufacturing site. This compliance advantage is crucial for maintaining operating licenses in regions with strict environmental protections. Additionally, the process is inherently scalable; the use of continuous dispersers allows for easy translation from pilot plant to multi-ton commercial reactors without the heat transfer limitations often encountered in viscous organic solvent systems. This scalability ensures that the supplier can grow alongside the client's needs, supporting commercial scale-up of complex heterocyclic compounds with confidence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cilostazol using this advanced aqueous methodology. These insights are derived directly from the experimental data and claims found in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process adoption.
Q: How does the aqueous method improve purity compared to traditional organic solvent methods?
A: The aqueous method significantly suppresses the formation of N-1 alkylated impurities, which are common in organic solvents. By optimizing the base concentration and water ratio, the reaction selectively targets the phenolic hydroxyl group, resulting in purity levels exceeding 99.5% without complex chromatography.
Q: What are the environmental benefits of this specific Cilostazol synthesis route?
A: This route eliminates the need for volatile organic compounds (VOCs) and phase transfer catalysts. Using water as the primary solvent reduces hazardous waste generation and removes the energy-intensive steps associated with solvent recovery and distillation, aligning with modern green chemistry principles.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. The use of a continuous disperser prevents product agglomeration, ensuring consistent heat transfer and reaction efficiency even at multi-ton scales. The simplified work-up procedure further enhances throughput capabilities for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cilostazol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is essential for the future of the pharmaceutical industry. Our technical team has extensively analyzed the aqueous alkylation pathway described in CN1257902C and possesses the expertise to implement this technology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are realized in every batch we produce. Our facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every gram of Cilostazol we deliver meets the highest international pharmacopeial standards.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized aqueous process, we can offer a supply solution that balances economic efficiency with environmental responsibility. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality makes us the ideal strategic partner for your Cilostazol supply needs.
