Advanced Manufacturing Strategies for Cilostazol Intermediates via Optimized Alkylation Routes
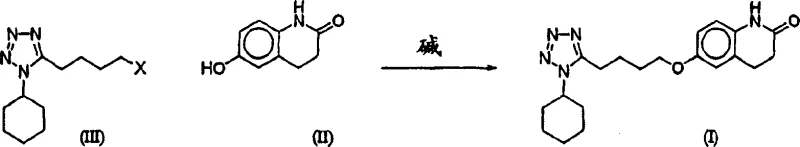
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical therapeutic agents, and the synthesis of Cilostazol stands as a prime example where process innovation drives commercial viability. Patent CN1469864A introduces a transformative approach to preparing 6-[4-(1-cyclohexyl-1H-tetrazol-5-yl)butoxy]-3,4-dihydro-2(1H)-quinolinone, addressing long-standing inefficiencies in alkylation chemistry. This technology shifts the paradigm from wasteful excess reagent usage to precise stoichiometric control, leveraging either biphasic phase transfer catalysis or buffered homogeneous inorganic base systems. For R&D directors and procurement specialists, understanding these mechanistic nuances is vital for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials at scale. The patent details how separating reactive species or buffering pH levels can drastically minimize degradation, offering a blueprint for cost-effective production that aligns with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
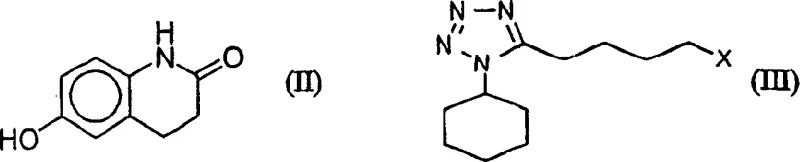
Historically, the synthesis of Cilostazol relied heavily on methods disclosed in earlier patents such as U.S. Patent No. 4,277,479, which utilized strong bases to alkylate 6-hydroxy-3,4-dihydroquinolinone (6-HQ). A significant drawback of these conventional routes was the inherent instability of the tetrazole alkylating agent, specifically 5-(4-halobutyl)-1-cyclohexyl-1H-tetrazole, when exposed to basic conditions. As illustrated in the reaction schemes, exposure to alkali metal hydroxides often triggered elimination and cyclization side reactions, generating impurities designated as compounds (IV) and (V). To compensate for this degradation, manufacturers were forced to employ a substantial molar excess of the tetrazole reagent, sometimes up to two equivalents, relative to the phenol substrate. This practice not only inflated raw material costs significantly but also complicated downstream purification, often necessitating column chromatography to remove unreacted starting materials and by-products, a step that is notoriously difficult to scale and environmentally burdensome due to solid waste generation.
The Novel Approach
The methodology presented in CN1469864A overcomes these historical bottlenecks by introducing two distinct yet complementary strategies that preserve the integrity of the sensitive tetrazole moiety. The first strategy employs a biphasic system where the base and phenol reside in an aqueous phase while the tetrazole remains in a water-immiscible organic solvent, mediated by a phase transfer catalyst. This physical separation prevents direct contact between the strong base and the alkylating agent until the moment of reaction at the interface or within the organic phase. Alternatively, the patent describes a homogeneous approach using a mixture of inorganic bases, such as potassium hydroxide and potassium carbonate, in an anhydrous hydroxylic solvent. This combination creates a buffered environment that maintains sufficient basicity for deprotonation without reaching the harsh pH levels that trigger decomposition. Both approaches enable the use of stoichiometric or even sub-stoichiometric amounts of the expensive tetrazole reagent, fundamentally altering the economic model of production by maximizing atom economy and simplifying isolation through recrystallization rather than chromatography.
Mechanistic Insights into Phase Transfer and Buffered Alkylation
To fully appreciate the technical superiority of this process, one must delve into the mechanistic interactions that govern selectivity and yield. In the biphasic variation, the phase transfer catalyst, typically a quaternary ammonium salt like Aliquat 336 or tetrabutylammonium bromide, plays a critical role in shuttling the 6-HQ phenoxide anion from the aqueous layer into the organic layer. Once in the organic phase, the lipophilic ion pair reacts efficiently with the dissolved tetrazole derivative. This mechanism ensures that the bulk aqueous base never directly attacks the alkyl halide chain of the tetrazole, thereby suppressing the formation of elimination by-products. The efficiency of this transfer can be further enhanced by adding reaction accelerators like sodium sulfate to the aqueous phase, which increases ionic strength and drives the organic species out of the water, effectively pushing the equilibrium toward the product. This level of control allows for near-quantitative conversion of the tetrazole, a metric that is crucial for cost management in large-scale manufacturing.
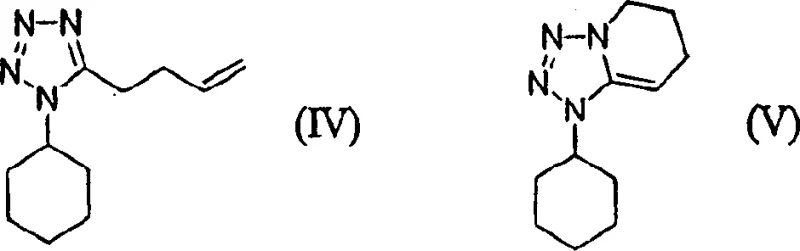
In the homogeneous variation, the suppression of side reactions is achieved through careful pH modulation and water control. The patent highlights that the tetrazole intermediate is susceptible to hydrolysis and elimination in the presence of water and strong bases. By utilizing a mixture of hydroxide and carbonate bases, the system buffers the reaction medium, preventing localized spikes in alkalinity that could degrade the reagent. Furthermore, the optional use of molecular sieves to remove water generated during the deprotonation of 6-HQ ensures that the reaction environment remains anhydrous. This is particularly important because water can facilitate the hydrolysis of the halide or promote the cyclization side reactions shown in the degradation pathways. By maintaining strict control over moisture and base strength, the process minimizes the formation of dimerized by-products and ensures that the nucleophilic attack occurs exclusively at the phenolic oxygen rather than the lactam nitrogen, resulting in a cleaner crude product profile that is amenable to simple crystallization techniques.
How to Synthesize Cilostazol Efficiently
Implementing this patented technology requires precise adherence to reaction parameters to achieve the reported improvements in yield and purity. The process generally involves the preparation of distinct phases or a carefully buffered single phase, followed by controlled mixing and thermal treatment. Operators must select appropriate solvents that offer the right balance of solubility for the reactants and insolubility for the product to facilitate isolation. For instance, in the biphasic method, toluene or methylene chloride serves as the organic carrier, while the homogeneous method relies on anhydrous alcohols like n-butanol. The choice of phase transfer catalyst and its loading is also critical, with sub-stoichiometric amounts often proving sufficient to drive the reaction to completion. Detailed standard operating procedures regarding the order of addition, particularly the portionwise addition of base in the homogeneous method to prevent exotherms and dimerization, are essential for reproducibility. The following guide outlines the generalized steps derived from the patent examples to assist technical teams in replicating this high-efficiency route.
- Prepare a biphasic system by dissolving 6-HQ and inorganic base in water, and the tetrazole derivative in a water-immiscible organic solvent like toluene.
- Add a quaternary ammonium phase transfer catalyst such as Aliquat 336 to facilitate the transfer of the phenoxide anion into the organic phase.
- Heat the mixture to reflux to drive the alkylation to completion, followed by cooling and recrystallization to isolate high-purity Cilostazol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the processes described in CN1469864A offers substantial strategic advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediates manufacturing. The most immediate impact is the drastic reduction in raw material costs associated with the tetrazole building block. By eliminating the need for a twofold molar excess of this complex and expensive reagent, manufacturers can significantly lower the bill of materials per kilogram of finished API intermediate. Furthermore, the ability to isolate the product via recrystallization rather than column chromatography removes a major bottleneck in production throughput. Chromatography is not only slow and labor-intensive but also consumes vast quantities of silica gel and solvents, creating significant hazardous waste disposal costs. Replacing this with a crystallization step streamlines the workflow, reduces cycle times, and minimizes the environmental footprint, aligning with increasingly stringent regulatory requirements for sustainable chemical manufacturing.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the optimization of reagent stoichiometry. In traditional methods, the instability of the tetrazole reagent forced the use of large excesses to drive the reaction, meaning a significant portion of high-value raw material was wasted as degradation by-products. The new methods stabilize the reaction environment, allowing for the use of stoichiometric or even slight sub-stoichiometric amounts of the tetrazole. This directly translates to a lower cost of goods sold (COGS) as less expensive starting material is required to produce the same amount of product. Additionally, the avoidance of chromatographic purification eliminates the recurring costs of stationary phases and the extensive solvent recovery operations associated with them, further enhancing the overall profit margin for the manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable inorganic bases such as potassium carbonate and sodium hydroxide, which replace more specialized or hazardous organic bases like DBU used in older methods. These inorganic reagents are commodity chemicals with robust global supply chains, reducing the risk of procurement delays. Moreover, the simplified work-up procedure, which often involves simple filtration and washing, reduces the dependency on specialized equipment or skilled labor for complex separations. This operational simplicity ensures that production schedules are more predictable and less prone to disruptions caused by equipment fouling or extended purification cycles, thereby guaranteeing more consistent delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but the biphasic and buffered homogeneous systems described here are inherently scalable. The biphasic system, in particular, benefits from excellent heat transfer characteristics, allowing for safer management of exothermic events during large-batch production. From an environmental standpoint, the reduction in solvent usage and the elimination of silica waste from chromatography columns significantly lower the E-factor (mass of waste per mass of product). This makes the process more attractive for facilities operating under strict environmental permits and reduces the liability associated with hazardous waste disposal. The ability to recycle solvents like toluene or n-butanol further enhances the sustainability profile, making the supply of these intermediates more secure in a regulatory landscape that increasingly penalizes wasteful chemical practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific problem-solution dynamics presented in the patent documentation, focusing on the practical implications for quality control and process engineering. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the empirical data and mechanistic explanations provided within the intellectual property, ensuring that expectations regarding yield, purity, and operational complexity are grounded in verified scientific principles.
Q: Why is the tetrazole intermediate unstable in conventional synthesis methods?
A: In conventional methods using strong aqueous bases, the 5-(4-halobutyl)-tetrazole intermediate undergoes elimination and cyclization side reactions, forming unwanted by-products like compounds (IV) and (V), which necessitates the use of excess reagents.
Q: How does the phase transfer catalysis method improve yield?
A: The biphasic system physically separates the base-sensitive tetrazole in the organic phase from the aqueous base, allowing the phenoxide anion to transfer and react selectively without degrading the expensive alkylating agent.
Q: Is chromatography required for purification in this new process?
A: No, the optimized process allows for high-purity isolation through simple recrystallization from solvents like n-butanol or acetone, eliminating the need for costly and waste-generating column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cilostazol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated alkylation techniques described in CN1469864A can be seamlessly transferred to industrial reactors. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical methods to monitor for the specific elimination by-products and dimers that this process is designed to avoid. Our commitment to quality assurance means that every batch of Cilostazol intermediate meets the exacting standards required by global regulatory bodies, providing our partners with the confidence needed to advance their own drug development pipelines without supply chain interruptions.
We invite potential partners to engage with our technical procurement team to discuss how these process optimizations can be tailored to your specific volume requirements. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis that quantifies the potential reductions in raw material and waste disposal expenses for your project. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of this high-efficiency synthesis with your current quality frameworks. Let us collaborate to secure a sustainable and cost-effective supply of high-purity pharmaceutical intermediates that drive your business forward.
