Revolutionizing Cilostazol Production: High-Yield Aqueous Synthesis for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the synthesis of critical cardiovascular agents like cilostazol. Patent CN1553908A introduces a transformative production method that fundamentally shifts the paradigm from traditional organic solvent-based systems to an innovative aqueous medium. This technical breakthrough addresses long-standing challenges in the alkylation of carbostyril derivatives, specifically targeting the suppression of regioisomeric impurities that have historically plagued manufacturing efficiency. By utilizing water as the primary solvent in conjunction with specific inorganic basic compounds, this process achieves exceptional conversion rates and purity profiles that were previously unattainable with conventional techniques. For R&D directors and process chemists, this represents a significant leap forward in reaction engineering, offering a cleaner, safer, and more economically viable route to high-quality active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cilostazol has relied heavily on the use of organic solvents such as dimethylformamide (DMF), toluene, or acetone, often necessitating the addition of phase transfer catalysts to facilitate the reaction between the phenolic substrate and the alkyl halide. These conventional methodologies are fraught with significant drawbacks, primarily stemming from poor selectivity and environmental hazards. A critical failure mode in these traditional routes is the concurrent alkylation at the nitrogen atom of the quinolone ring, leading to the formation of N-1 substituted byproducts that are structurally similar to the target molecule and notoriously difficult to separate. Furthermore, the reliance on volatile organic compounds (VOCs) imposes severe burdens on waste management infrastructure and increases the overall carbon footprint of the manufacturing process. The yields associated with these older methods are frequently suboptimal, often ranging between 50% and 74%, which translates to substantial material loss and increased cost of goods sold for manufacturers operating at scale.
The Novel Approach
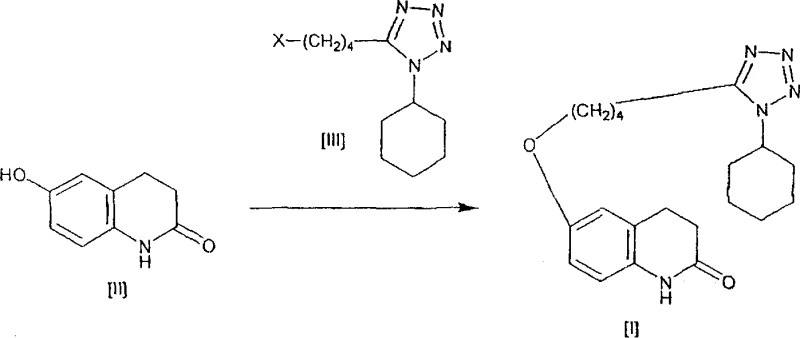
In stark contrast, the novel approach detailed in the patent leverages the unique properties of water as a reaction medium to drive the O-alkylation selectively while suppressing unwanted N-alkylation. By carefully controlling the ratio of water to the carbostyril derivative—specifically maintaining a 3 to 7-fold weight ratio—and employing inorganic bases like potassium carbonate or sodium hydroxide, the reaction environment is optimized for high efficiency. This aqueous system not only eliminates the need for expensive and toxic organic solvents but also simplifies the downstream processing significantly. The absence of phase transfer catalysts removes a potential source of metallic contamination, thereby enhancing the final purity of the API. This method demonstrates that what was once considered a solubility limitation—the insolubility of the tetrazole derivative in water—can be overcome through precise stoichiometric control and thermal management, resulting in a streamlined process that delivers yields exceeding 90% with purity levels approaching 99.7%.
Mechanistic Insights into Aqueous Nucleophilic Substitution
The success of this aqueous synthesis hinges on the delicate interplay between the inorganic base and the solvent matrix to direct the nucleophilic attack exclusively to the phenolic oxygen. In the presence of strong inorganic bases such as potassium carbonate or sodium hydroxide, the phenolic hydroxyl group of the 6-hydroxy-3,4-dihydroquinolinone is deprotonated to form a phenoxide anion. In an aqueous environment, the solvation shell surrounding this anion plays a crucial role in modulating its reactivity. Unlike in polar aprotic organic solvents where the nucleophile might be too aggressive or poorly solvated leading to side reactions, the water molecules stabilize the transition state in a manner that favors O-alkylation over N-alkylation. The tetrazole derivative, despite its limited solubility, reacts efficiently at the interface or within the micellar-like structures formed under these specific conditions, ensuring that the alkyl chain is attached precisely at the 6-position oxygen atom without attacking the lactam nitrogen at the 1-position.
Furthermore, the control of impurity formation is intrinsically linked to the choice of base and the absence of organic co-solvents that might stabilize the N-anion. The patent data indicates that by avoiding organic solvents and phase transfer catalysts, the energetic barrier for N-alkylation remains prohibitively high under the specified reaction conditions (50°C to 150°C). The use of a continuous disperser in the preferred embodiment further enhances mass transfer, ensuring that the solid tetrazole derivative is constantly exposed to the reactive phenoxide species in the aqueous phase. This mechanical agitation prevents the localized accumulation of reactants that could lead to hot spots and subsequent degradation or side reactions. Consequently, the impurity profile is drastically simplified, with the major contaminant being unreacted starting material rather than inseparable structural analogs, facilitating a straightforward crystallization and washing protocol to achieve pharmaceutical-grade purity.
How to Synthesize Cilostazol Efficiently
The synthesis of cilostazol via this aqueous route is designed for operational simplicity and robustness, making it highly attractive for technology transfer and scale-up. The process begins with the suspension of the carbostyril derivative and the tetrazole alkyl halide in water, followed by the addition of the inorganic base. The reaction mixture is then heated, typically to around 90°C to 100°C, and maintained under agitation for several hours to ensure complete conversion. A key operational feature is the potential use of a continuous disperser to circulate the reaction mixture, which prevents the agglomeration of product crystals and maintains a homogeneous reaction environment despite the heterogeneous nature of the starting materials.
- Prepare the reaction mixture by combining 6-hydroxy-3,4-dihydroquinolinone and the tetrazole derivative in water with an inorganic base such as potassium carbonate.
- Heat the mixture to between 50°C and 150°C, optionally circulating through a disperser to prevent aggregation, and maintain for 1 to 20 hours.
- Cool the reaction, filter the crude crystals, and purify via washing with water and alcohol to obtain high-purity cilostazol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. The most immediate impact is seen in the drastic reduction of raw material costs associated with solvent procurement and disposal. By replacing expensive, regulated organic solvents with water, manufacturers can significantly lower their variable production costs and mitigate the risks associated with volatile solvent pricing and supply chain disruptions. Additionally, the elimination of phase transfer catalysts and the reduction in purification steps translate to shorter cycle times and higher throughput capacity, allowing facilities to meet market demand more responsively. The high yield and purity achieved directly contribute to a more efficient use of starting materials, reducing the overall material intensity of the process and minimizing waste generation, which is a critical metric for modern sustainable supply chains.
- Cost Reduction in Manufacturing: The shift to an aqueous system eliminates the capital and operational expenditures related to solvent recovery units and explosion-proof infrastructure required for organic solvents. Since water is non-flammable and inexpensive, the safety protocols can be streamlined, reducing insurance and compliance costs. Furthermore, the high selectivity of the reaction means that less raw material is wasted on byproduct formation, effectively lowering the cost per kilogram of the final API. The simplified work-up procedure, which often requires only filtration and washing rather than complex chromatographic separations or multiple recrystallizations from organic solvents, further drives down labor and utility costs associated with downstream processing.
- Enhanced Supply Chain Reliability: Relying on water as a primary solvent decouples the manufacturing process from the volatility of the petrochemical solvent market. Organic solvents are subject to price fluctuations and availability issues driven by upstream oil and gas dynamics; water, conversely, is a stable and universally available resource. This stability ensures consistent production scheduling and reduces the risk of shutdowns due to solvent shortages. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and base concentrations, provides operational flexibility that allows manufacturers to adapt quickly to feedstock variations or equipment constraints without compromising product quality, thereby securing a steady supply of high-purity intermediates for downstream formulation.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently safer and easier to scale. The absence of toxic VOCs simplifies permitting processes and reduces the burden on wastewater treatment facilities, as the effluent is primarily aqueous and easier to treat than mixed organic waste streams. The use of inorganic bases like potassium carbonate generates benign salt byproducts that are easier to manage than complex organic residues. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major global pharmaceutical companies committed to reducing their Scope 3 emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced aqueous synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process viability. Understanding these details is crucial for stakeholders assessing the risk and reward profile of adopting this new manufacturing standard.
Q: Why is the aqueous method superior to traditional organic solvent methods for cilostazol?
A: Traditional methods often suffer from low yields (50-74%) and the formation of difficult-to-remove N-1 substituted impurities. The aqueous method described in CN1553908A suppresses this side reaction, achieving yields over 90% and purity exceeding 99.5% without complex purification.
Q: What are the environmental benefits of this production process?
A: This process replaces hazardous organic solvents and phase transfer catalysts with water and inorganic bases. This significantly reduces VOC emissions, lowers waste disposal costs, and aligns with green chemistry principles for sustainable pharmaceutical manufacturing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly describes conditions suitable for industrial scale-up, including the use of continuous dispersers to manage crystal growth and prevent aggregation, ensuring consistent quality and operational safety at commercial volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cilostazol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous synthesis pathway described in CN1553908A for the production of high-quality cilostazol. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and mixing requirements of this aqueous process, including the integration of continuous dispersers for optimal crystal morphology control. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for cardiovascular therapeutics, delivering a product with minimal impurity profiles and consistent physicochemical properties.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and environmentally superior manufacturing route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this aqueous technology for your specific supply chain needs. We encourage you to reach out today to discuss specific COA data, route feasibility assessments, and how our expertise in green chemistry can help you secure a reliable, high-purity cilostazol supply that aligns with your sustainability goals and commercial objectives.
