Advanced Manufacturing of Doxifluridine: A Technical Breakthrough for Commercial Scale Production
Advanced Manufacturing of Doxifluridine: A Technical Breakthrough for Commercial Scale Production
The global demand for high-purity oncology therapeutics continues to drive innovation in the synthesis of critical nucleoside analogues. Specifically, the production of 5'-deoxy-5-fluorouridine, widely known by its International Nonproprietary Name Doxifluridine, has historically faced challenges regarding reagent toxicity and process complexity. Patent CN1184228C introduces a transformative methodology that addresses these longstanding industrial bottlenecks by replacing hazardous phosphorus-based reagents with a streamlined sulfonate activation pathway. This technical insight report analyzes the proprietary process detailed in the patent, highlighting its potential to redefine supply chain standards for reliable API intermediate suppliers. By shifting from stoichiometric phosphorus reagents to catalytic hydrogenation, manufacturers can achieve superior purity profiles while mitigating the environmental liabilities associated with traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Doxifluridine relied heavily on methodologies described in earlier patents such as US 4,071,680 and EP 21,231, which presented significant operational hazards. The conventional approach typically involved the use of triphenylphosphite methoiodide as a chemical iodinating agent to substitute the 5'-hydroxyl group. While chemically effective, this reagent generates toxic reaction by-products that necessitate rigorous safety protocols and expensive waste treatment procedures. Furthermore, alternative routes described in the literature often employed bromine substitution followed by catalytic hydrogenation under harsh conditions, frequently requiring strong inorganic bases like potassium hydroxide. These aggressive conditions not only pose risks to equipment integrity but also complicate the purification process, often leading to lower overall yields and increased formation of degradation impurities that are difficult to remove during downstream processing.
The Novel Approach
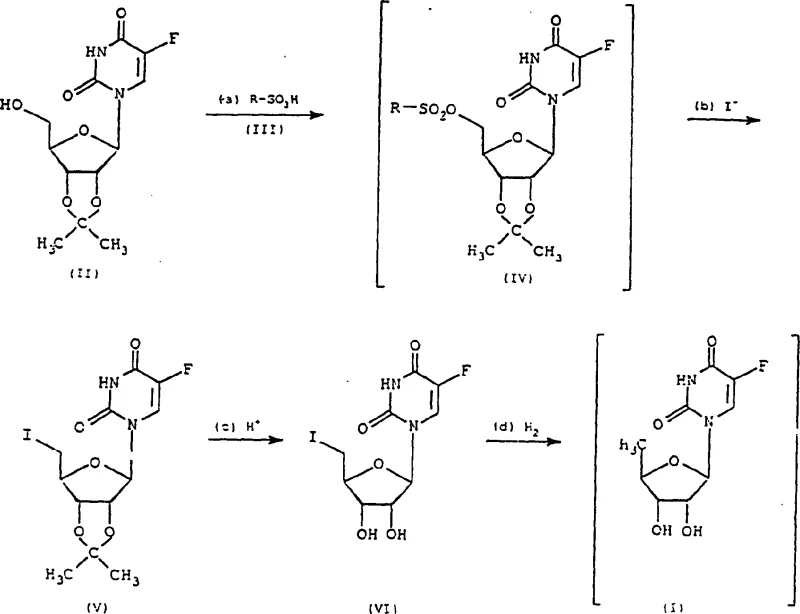
The innovative process disclosed in CN1184228C circumvents these issues by introducing a mild, four-step sequence that completely eliminates the need for phosphorus compounds. The strategy begins with the activation of the 5'-hydroxyl group of 2',3'-O-isopropylidene-5-fluorouridine using a sulfonic acid derivative, such as p-toluenesulfonyl chloride. This activated intermediate is then subjected to nucleophilic substitution with an alkaline iodide, such as sodium iodide, to install the iodine atom cleanly. Crucially, the final deoxygenation is achieved through catalytic hydrogenation or the use of hydrogen donors, avoiding the harsh basic conditions of previous methods. This approach not only enhances operator safety but also simplifies the workup procedure, as the by-products are easily removed by filtration or washing, resulting in a much cleaner crude product profile ready for final crystallization.
Mechanistic Insights into Sulfonate Activation and Catalytic Reduction
The core of this technological advancement lies in the precise control of the nucleoside sugar moiety through selective functional group manipulation. The mechanism initiates with the sulfonylation of the primary hydroxyl group at the 5'-position, converting a poor leaving group into an excellent sulfonate ester. This activation is critical for the subsequent displacement by iodide ions, which proceeds via an SN2 mechanism with high stereospecificity, ensuring the integrity of the chiral centers in the ribose ring is maintained. Following iodination, the isopropylidene protecting group is hydrolyzed under acidic conditions, exposing the 2' and 3' hydroxyl groups necessary for biological activity. The final step involves the reductive removal of the iodine atom, which acts as a temporary placeholder for the hydrogen atom, utilizing a palladium on carbon catalyst to facilitate the hydrogenolysis efficiently.
From an impurity control perspective, this route offers distinct advantages over bromine-based alternatives. The use of iodide as the leaving group precursor allows for milder reduction conditions, significantly reducing the risk of debasing the fluorouracil base or causing epimerization at the anomeric center. The patent data reports HPLC purities exceeding 99% for the intermediates and the final product, demonstrating the robustness of this chemical pathway. By avoiding the formation of phosphine oxides or other organophosphorus residues, the downstream purification burden is drastically reduced. This mechanistic elegance ensures that the final active pharmaceutical ingredient meets stringent regulatory specifications for residual solvents and heavy metals, a critical factor for procurement managers evaluating long-term supply partners for high-purity pharmaceutical intermediates.
How to Synthesize 5'-Deoxy-5-fluorouridine Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the sulfonylation and iodination phases to maximize yield. The process is designed to be flexible, allowing for the isolation of intermediates or a telescoped one-pot operation for the first three steps, which can significantly reduce cycle times in a manufacturing setting. Operators should utilize polar aprotic solvents like acetone or dimethylformamide for the substitution steps to ensure complete solubility of the inorganic iodide salts. For the final reduction, the choice between catalytic hydrogenation and chemical hydrogen donors depends on facility capabilities, though catalytic methods are generally preferred for green chemistry compliance. Detailed standardized operating procedures regarding reagent addition rates and quenching protocols are essential to maintain the high reproducibility reported in the patent examples.
- React 2',3'-O-isopropylidene-5-fluorouridine with a sulfonic acid derivative (e.g., p-toluenesulfonyl chloride) to form the 5'-sulfonyloxy intermediate.
- Treat the sulfonyloxy derivative with an alkali metal iodide (e.g., sodium iodide) in a ketone solvent to substitute the sulfonate group with iodine.
- Hydrolyze the isopropylidene protecting group in acidic media to yield 5'-deoxy-5'-iodo-5-fluorouridine.
- Perform catalytic hydrogenation using Pd/C or a hydrogen donor to reduce the iodo group to hydrogen, yielding the final Doxifluridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The elimination of specialized, toxic phosphorus reagents removes a significant variable from the raw material sourcing matrix, relying instead on commodity chemicals like tosyl chloride and sodium iodide which are globally available and price-stable. This shift mitigates the risk of supply disruptions caused by the limited availability of niche reagents. Furthermore, the simplified workup procedures, which often involve simple filtration and solvent evaporation rather than complex chromatographic separations, lead to a drastic simplification of the manufacturing workflow. This efficiency gain directly correlates to reduced production cycles and lower utility consumption, providing a compelling argument for cost reduction in pharmaceutical manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The removal of expensive and toxic phosphorus-based reagents eliminates the need for costly waste disposal and specialized containment infrastructure. By utilizing common solvents such as ethyl acetate and acetone, which are easily recovered and recycled, the overall solvent cost per kilogram of product is significantly optimized. Additionally, the high yields reported in the patent, often exceeding 90% for individual steps, mean that less raw material is wasted, directly improving the cost of goods sold (COGS) and enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous starting materials ensures a more resilient supply chain capable of withstanding market fluctuations. The ability to perform multiple steps in a telescoped manner reduces the number of unit operations and intermediate storage requirements, thereby shortening the overall lead time for high-purity nucleoside intermediates. This agility allows manufacturers to respond more rapidly to changes in market demand, ensuring consistent delivery schedules for downstream API producers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard industrial equipment without the need for exotic reactors. The absence of heavy metal contaminants (other than the recoverable palladium catalyst) and organophosphorus waste aligns perfectly with modern environmental, health, and safety (EHS) regulations. This compliance reduces the regulatory burden on the manufacturing site and minimizes the risk of production shutdowns due to environmental violations, ensuring long-term supply continuity for critical oncology medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. Understanding these details is crucial for R&D teams evaluating technology transfer and for quality assurance teams establishing specification limits. The answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: Why is this new synthesis route preferred over traditional phosphorus-based methods?
A: Traditional methods often utilize toxic phosphorus reagents like triphenylphosphite methoiodide, which generate hazardous byproducts requiring complex disposal. The patented process eliminates phosphorus compounds entirely, utilizing safer sulfonate activation and catalytic hydrogenation, significantly reducing environmental impact and operational toxicity risks.
Q: What yields can be expected from this manufacturing process?
A: The patent data indicates exceptionally high efficiency, with individual steps (a), (b), and (c) achieving yields generally higher than 90% of theory. The overall recovery yield for the key intermediate (Formula VI) reaches 83-85%, and the final reduction step yields approximately 89%, ensuring robust material throughput for commercial production.
Q: Is this process suitable for large-scale industrial application?
A: Yes, the process is designed for scalability. It allows for a 'one-pot' implementation of the first three steps without isolating intermediates, drastically simplifying the workflow. Furthermore, the use of common solvents like acetone and ethyl acetate, along with standard catalysts like Pd/C, facilitates easy technology transfer from laboratory to multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doxifluridine Supplier
The technical potential of the sulfonate-activation route for Doxifluridine represents a significant opportunity for optimizing the global supply of this critical oncology intermediate. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalytic requirements of this process, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency is paramount in the pharmaceutical industry, and our commitment to process validation guarantees that the high yields and purity levels described in the patent are reproducible on an industrial scale.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how this advanced manufacturing method can enhance your supply chain resilience. Whether you require pilot-scale quantities for clinical trials or multi-ton volumes for commercial launch, our dedicated team is ready to support your project with the highest standards of quality and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
