Scalable Green Synthesis of Pyrimidine Derivatives Using Sulfonic Acid Ionic Liquids
Scalable Green Synthesis of Pyrimidine Derivatives Using Sulfonic Acid Ionic Liquids
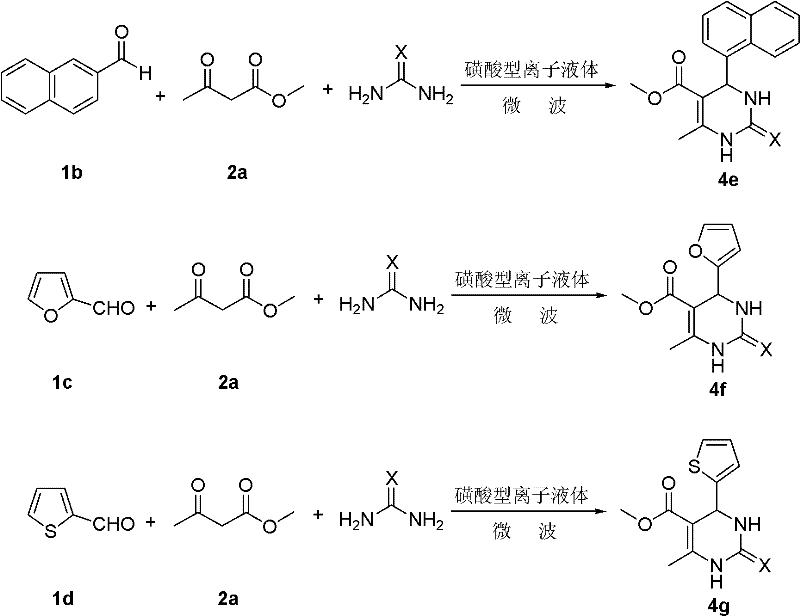
The pharmaceutical and agrochemical industries are increasingly demanding sustainable manufacturing processes that minimize environmental impact while maximizing efficiency and product purity. A pivotal advancement in this domain is detailed in patent CN102241654A, which discloses an environmentally friendly synthetic method for a broad class of pyrimidine derivatives. This technology leverages the unique properties of sulfonic acid-type ionic liquids as dual-function catalysts and solvents, operating under solvent-free conditions promoted by microwave irradiation. By replacing traditional corrosive mineral acids and volatile organic solvents with a recyclable ionic liquid system, this process addresses critical pain points in modern chemical manufacturing, including equipment corrosion, hazardous waste generation, and energy consumption. The methodology demonstrates exceptional versatility, accommodating a wide range of aromatic aldehydes, acetoacetates, and urea or thiourea substrates to produce high-value intermediates with impressive yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrimidine derivatives, particularly via the Biginelli three-component condensation reaction, has relied heavily on traditional homogeneous acid catalysts such as hydrochloric acid, trifluoroacetic acid, or Lewis acids like ferric chloride and indium trichloride. While effective in laboratory settings, these conventional methodologies present severe drawbacks when translated to industrial scales. The use of strong mineral acids necessitates the use of specialized corrosion-resistant reactor materials, significantly inflating capital expenditure and maintenance costs. Furthermore, these catalysts are often difficult to separate from the reaction mixture, leading to complex workup procedures that generate substantial amounts of acidic wastewater. The reliance on volatile organic solvents to facilitate these reactions exacerbates environmental concerns, contributing to atmospheric pollution and requiring expensive solvent recovery systems. Additionally, many traditional Lewis acid catalysts are moisture-sensitive, requiring stringent anhydrous conditions that complicate operational protocols and increase energy usage for drying reagents and solvents.
The Novel Approach
The innovative approach described in the patent data fundamentally re-engineers the reaction landscape by introducing a sulfonic acid-functionalized ionic liquid, specifically [(HSO3-p)2Im][CF3SO3], as a robust and reusable catalyst. This novel system operates under solvent-free conditions, effectively eliminating the hazards and costs associated with volatile organic compounds. The integration of microwave irradiation serves as a powerful promoter, enabling rapid dielectric heating that accelerates reaction kinetics far beyond what is achievable with conventional conductive heating. This combination allows the reaction to proceed at moderate temperatures ranging from 50°C to 120°C within remarkably short timeframes of 10 to 50 minutes. The result is a streamlined process that not only achieves high yields and purity but also simplifies product isolation through simple filtration after quenching in ice water, thereby drastically reducing the complexity of downstream processing.
Mechanistic Insights into Sulfonic Acid Ionic Liquid Catalysis
The efficacy of this synthetic route is rooted in the dual functionality of the sulfonic acid ionic liquid, which acts as both a Brønsted acid catalyst and a reaction medium. The presence of the sulfonic acid groups (-SO3H) on the imidazolium cation provides strong proton-donating capability, which is essential for activating the carbonyl group of the aromatic aldehyde. This activation facilitates the initial nucleophilic attack by the urea or thiourea, forming the key intermediate. Simultaneously, the ionic liquid stabilizes the transition states through electrostatic interactions, lowering the activation energy barrier for the subsequent cyclization steps. Unlike traditional Lewis acids which may deactivate in the presence of moisture or coordinating solvents, this functionalized ionic liquid maintains its catalytic integrity under the reaction conditions, ensuring consistent performance across multiple cycles. The microwave field further enhances this mechanism by selectively heating the polar ionic species, creating localized superheating effects that drive the equilibrium towards product formation more efficiently than bulk heating methods.
From an impurity control perspective, the mild yet efficient nature of this catalytic system offers significant advantages. Traditional acid-catalyzed reactions often suffer from side reactions such as polymerization of the aldehyde or hydrolysis of the ester moiety due to harsh conditions and prolonged reaction times. In contrast, the rapid kinetics enabled by microwave promotion and the specific acidity of the ionic liquid minimize the residence time of reactive intermediates, thereby suppressing the formation of by-products. The solvent-free environment also prevents solvolysis side reactions that might occur in alcoholic or aqueous media. Consequently, the crude product obtained after filtration exhibits high purity, often requiring minimal recrystallization to meet stringent pharmaceutical specifications. This inherent selectivity reduces the burden on purification units and improves the overall mass balance of the manufacturing process.
How to Synthesize Pyrimidine Derivatives Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemists aiming to optimize production workflows. The procedure involves a straightforward one-pot mixing of the aromatic aldehyde, acetoacetate ester, and urea or thiourea in the presence of the ionic liquid catalyst. The mixture is then subjected to microwave irradiation, after which the product precipitates upon addition to ice water. This direct isolation method bypasses the need for extraction and extensive washing steps typically required in liquid-liquid separations. For a comprehensive understanding of the specific stoichiometric ratios, temperature profiles, and workup details validated by experimental data, please refer to the standardized synthesis guide below.
- Combine the sulfonic acid ionic liquid catalyst [(HSO3-p)2Im][CF3SO3] with aromatic aldehyde, methyl acetoacetate (or ethyl ester), and urea (or thiourea) in a reaction vessel.
- Subject the mixture to microwave irradiation at temperatures between 50°C and 120°C for a duration of 10 to 50 minutes under solvent-free conditions.
- Pour the reaction mixture into ice-water, stir to precipitate the product, filter the solid cake, and dry to obtain the high-purity pyrimidine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-mediated synthesis offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of volatile organic solvents represents a major cost avoidance opportunity, removing the expenses associated with solvent purchasing, storage, and hazardous waste disposal. Furthermore, the reusability of the ionic liquid catalyst introduces a circular economy element to the process, where the catalytic medium can be recovered from the filtrate and recycled for subsequent batches, effectively amortizing the initial cost of the catalyst over a long operational lifespan. This significantly lowers the variable cost per kilogram of the final API intermediate, enhancing margin potential in competitive markets.
- Cost Reduction in Manufacturing: The transition to a solvent-free system drastically simplifies the material bill of quantities by removing the need for bulk solvents. Additionally, the ability to recover and reuse the expensive ionic liquid catalyst multiple times without significant loss of activity means that the effective catalyst cost per unit of production becomes negligible over time. The reduction in reaction time from hours to minutes also translates to higher throughput per reactor volume, allowing existing infrastructure to produce more product without capital expansion, thereby optimizing asset utilization and reducing fixed cost allocation per unit.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis—aromatic aldehydes, acetoacetates, and urea—are commodity chemicals with robust global supply chains, minimizing the risk of raw material shortages. The mild reaction conditions reduce the stress on reactor equipment, leading to lower maintenance frequencies and higher plant availability. Moreover, the simplified workup procedure, which relies on filtration rather than complex distillation or chromatography, reduces the dependency on specialized separation equipment and skilled labor, making the supply chain more resilient to operational disruptions.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, this green synthesis method positions manufacturers favorably for compliance. The absence of solvent vapors eliminates the need for extensive VOC abatement systems, and the non-corrosive nature of the ionic liquid extends the lifespan of standard stainless steel reactors. The process generates minimal waste, primarily consisting of the aqueous wash which is easily treated, aligning with modern sustainability goals and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid-catalyzed pyrimidine synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: Can the sulfonic acid ionic liquid catalyst be recovered and reused?
A: Yes, the patent data indicates that the filtrate containing the ionic liquid can be concentrated after filtration and directly reused for subsequent batches without significant loss in catalytic activity or product yield.
Q: What are the advantages of using microwave irradiation in this synthesis?
A: Microwave irradiation provides rapid and uniform heating, significantly reducing reaction times to as little as 10 minutes compared to traditional reflux methods, while maintaining high conversion rates and product purity.
Q: Is this method suitable for large-scale industrial production?
A: The method is highly suitable for scale-up due to its solvent-free nature, which eliminates the need for large volumes of volatile organic compounds, and the use of a reusable catalyst which simplifies downstream processing and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the sulfonic acid ionic liquid catalysis system in driving the next generation of pharmaceutical manufacturing. Our team of process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the laboratory are faithfully reproduced at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pyrimidine intermediate meets the exacting standards required for downstream drug synthesis, providing our partners with unwavering quality assurance.
We invite forward-thinking organizations to collaborate with us to leverage this advanced synthetic route for their specific pipeline needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this eco-friendly methodology can optimize your supply chain and reduce your overall cost of goods sold.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
