Advanced Quinolone And Naphthyridonecarboxylic Acid Derivatives For Commercial Pharmaceutical Production
The pharmaceutical landscape is constantly evolving to combat the rising threat of antimicrobial resistance, necessitating the development of next-generation antibacterial agents with superior efficacy profiles. Patent CN1075499C introduces a groundbreaking class of quinolone-and naphthyridonecarboxylic acid derivatives that redefine the structural boundaries of traditional fluoroquinolones. Unlike conventional agents that rely on simple piperazinyl substitutions, this technology incorporates complex bicyclic amido structures, specifically diazabicyclo[4.3.0]nonane derivatives, at the critical 7-position of the quinolone nucleus. This strategic molecular modification is not merely an incremental change but a fundamental shift that enhances lipophilicity and cellular penetration, allowing the compound to effectively target dormant and resistant microorganisms that typically evade standard therapies. The core innovation lies in the precise stereochemical control of these bicyclic amines, ensuring that the final active pharmaceutical ingredient possesses the optimal spatial configuration for receptor binding. 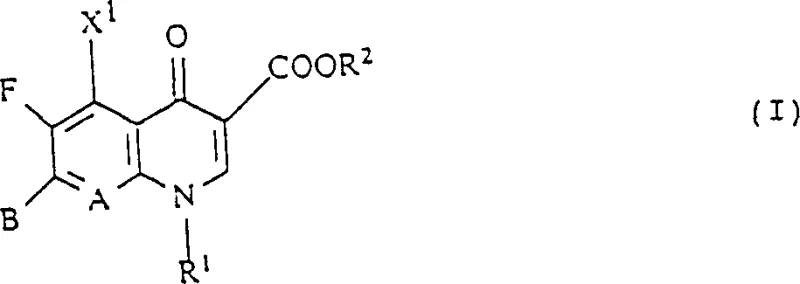
For research and development directors evaluating new chemical entities, understanding the mechanistic advantages of this novel scaffold is paramount for project feasibility. The limitations of conventional methods often stem from the use of racemic mixtures or less rigid cyclic amines that fail to maintain the necessary conformational stability within the bacterial DNA gyrase complex. Traditional synthetic routes frequently struggle with low enantiomeric excess, leading to impurities that complicate downstream purification and reduce overall therapeutic index. In contrast, the novel approach detailed in the patent leverages advanced chiral resolution techniques and asymmetric synthesis to produce enantiomerically pure intermediates. This ensures that every molecule in the final batch contributes to the desired pharmacological effect, eliminating the metabolic burden and potential toxicity associated with the inactive enantiomer. Furthermore, the robustness of the bicyclic structure provides enhanced metabolic stability, prolonging the half-life of the drug in vivo and reducing dosing frequency for patients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-substituted quinolones has relied heavily on nucleophilic displacement reactions using simple secondary amines, which often results in products with limited spectrum activity against anaerobic bacteria and resistant strains. The rigidity of the substituent is a critical factor; flexible chains can adopt multiple conformations, only one of which may be biologically active, thereby reducing the effective concentration at the target site. Moreover, conventional processes often lack efficient methods for separating optical isomers, forcing manufacturers to either sell racemic drugs with lower potency or incur significant costs in chiral chromatography. The presence of impurities from incomplete reactions or side products like N-alkylated byproducts further complicates the regulatory approval process, requiring extensive validation to prove safety. These technical bottlenecks create substantial barriers to entry for generic manufacturers seeking to produce high-quality antibacterial agents that meet modern pharmacopeial standards.
The Novel Approach
The methodology presented in CN1075499C overcomes these hurdles by introducing a highly structured bicyclic amine framework that locks the molecule into its bioactive conformation. By utilizing diazabicyclo[4.3.0]nonane derivatives, the synthesis achieves a level of steric precision that maximizes interaction with bacterial topoisomerases. The patent describes versatile synthetic pathways, including the reaction of fluorinated quinolone esters with chiral amines under mild conditions, which preserves the integrity of sensitive functional groups. This approach not only improves the yield of the desired isomer but also simplifies the purification process, as the distinct physical properties of the diastereomers allow for efficient crystallization. The result is a manufacturing process that is both chemically elegant and industrially viable, offering a clear pathway to high-purity intermediates that satisfy the rigorous demands of global regulatory agencies.
Mechanistic Insights into Diazabicyclo Nonane Substitution
The biological superiority of these compounds is rooted in the unique electronic and steric properties of the diazabicyclo nonane moiety. The bridgehead nitrogen atoms in the bicyclic system create a specific electron density distribution that facilitates stronger hydrogen bonding with the amino acid residues in the enzyme active site. 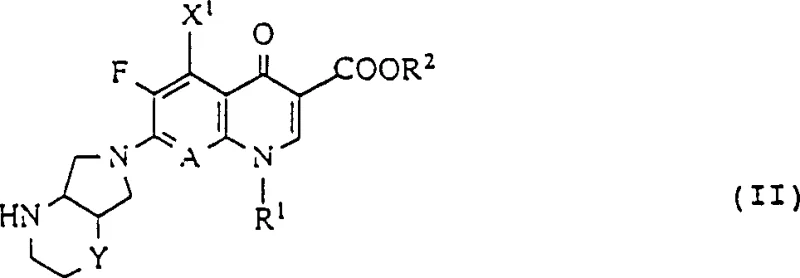 This enhanced binding affinity translates directly into lower minimum inhibitory concentrations (MICs) against pathogens such as Staphylococcus aureus and Pseudomonas aeruginosa. Furthermore, the rigid structure hinders the access of bacterial efflux pumps, which are a common mechanism of resistance against older fluoroquinolones. By physically blocking the expulsion of the drug from the bacterial cell, the compound accumulates to therapeutic levels more rapidly, ensuring rapid bactericidal action even in hostile biological environments. The patent data indicates that these derivatives maintain activity against strains that have developed resistance to penicillins, cephalosporins, and tetracyclines, making them invaluable assets for treating multidrug-resistant infections.
This enhanced binding affinity translates directly into lower minimum inhibitory concentrations (MICs) against pathogens such as Staphylococcus aureus and Pseudomonas aeruginosa. Furthermore, the rigid structure hinders the access of bacterial efflux pumps, which are a common mechanism of resistance against older fluoroquinolones. By physically blocking the expulsion of the drug from the bacterial cell, the compound accumulates to therapeutic levels more rapidly, ensuring rapid bactericidal action even in hostile biological environments. The patent data indicates that these derivatives maintain activity against strains that have developed resistance to penicillins, cephalosporins, and tetracyclines, making them invaluable assets for treating multidrug-resistant infections.
From an impurity control perspective, the synthesis strategy employs specific protecting groups and reaction conditions that minimize the formation of regioisomers. The use of acid-binding agents and controlled temperature profiles during the coupling reaction prevents the degradation of the quinolone core, which is susceptible to hydrolysis under harsh conditions. The detailed examples in the patent demonstrate that by selecting the appropriate solvent system, such as acetonitrile or dimethylformamide, manufacturers can achieve conversion rates that maximize throughput while minimizing waste. This level of process control is essential for maintaining a consistent impurity profile across different production batches, a key requirement for any supplier aiming to serve the regulated pharmaceutical market. The ability to produce salts with improved solubility and stability, such as hydrochlorides or mesylates, further enhances the commercial viability of these intermediates for final dosage form development.
How to Synthesize Quinolone Derivatives Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction engineering and quality control to ensure reproducibility at scale. The process begins with the preparation of the chiral amine precursor, followed by a carefully monitored nucleophilic substitution with the fluorinated quinolone ester. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results. Adhering to these protocols ensures that the critical quality attributes of the final product, such as optical purity and residual solvent levels, remain within specified limits. It is crucial for technical teams to validate each step of the workflow, particularly the crystallization stages, as these determine the final particle size distribution and flow properties of the powder.
- Preparation of enantiomerically pure diazabicyclo[4.3.0]nonane intermediates via chiral resolution or asymmetric synthesis.
- Nucleophilic displacement reaction between the chiral amine intermediate and fluorinated quinolone carboxylic acid esters.
- Purification of the final quinolone derivative through crystallization and salt formation to ensure high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers significant strategic benefits beyond mere technical performance. The streamlined synthesis route reduces the number of unit operations required to reach the final intermediate, which directly correlates to lower capital expenditure and reduced operational complexity. By eliminating the need for expensive transition metal catalysts often used in alternative cross-coupling strategies, the process achieves substantial cost savings in raw material procurement. The reliance on readily available starting materials ensures a stable supply chain that is less vulnerable to geopolitical disruptions or raw material shortages. Furthermore, the high yields reported in the patent examples suggest that less waste is generated per kilogram of product, aligning with increasingly stringent environmental regulations and reducing disposal costs. 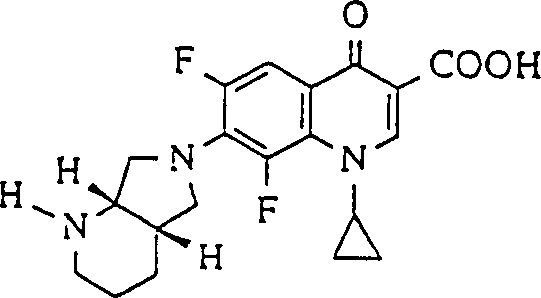
- Cost Reduction in Manufacturing: The elimination of complex chiral chromatography steps in favor of crystallization-based purification drastically lowers processing costs. By leveraging diastereomeric salt formation, manufacturers can achieve high enantiomeric purity without the need for expensive chiral columns or supercritical fluid chromatography systems. This shift from capital-intensive separation technologies to standard thermal processing equipment significantly improves the return on investment for production facilities. Additionally, the robust nature of the intermediates allows for longer storage times without degradation, reducing inventory write-offs and improving working capital efficiency.
- Enhanced Supply Chain Reliability: The synthetic pathways described utilize commodity chemicals and solvents that are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The flexibility of the chemistry allows for the production of various analogues using the same core infrastructure, enabling manufacturers to pivot quickly in response to market demand changes. This adaptability ensures continuous supply continuity even when specific raw material grades fluctuate in availability. The proven scalability of the reactions from gram to kilogram scale provides confidence that commercial volumes can be met without extensive re-engineering of the process.
- Scalability and Environmental Compliance: The processes operate at moderate temperatures and pressures, reducing energy consumption and enhancing workplace safety. The minimization of hazardous reagents and the ability to recycle solvents contribute to a greener manufacturing footprint, which is increasingly important for corporate sustainability goals. The high atom economy of the displacement reactions means that fewer byproducts are formed, simplifying wastewater treatment and reducing the environmental burden. This compliance with green chemistry principles facilitates smoother regulatory approvals and enhances the brand reputation of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel quinolone derivatives. These answers are derived directly from the experimental data and claims found within the patent documentation to ensure accuracy. Understanding these details is critical for stakeholders evaluating the integration of these intermediates into their existing product pipelines or new drug development programs.
Q: What distinguishes these quinolone derivatives from conventional fluoroquinolones?
A: These derivatives feature a novel bicyclic amido substitution at the 7-position, specifically utilizing diazabicyclo[4.3.0]nonane moieties. This structural modification significantly enhances activity against dormant and drug-resistant microorganisms compared to standard ciprofloxacin analogues.
Q: Are the chiral intermediates scalable for industrial production?
A: Yes, the patent outlines multiple methods for obtaining enantiomerically pure intermediates, including diastereomeric salt resolution and asymmetric hydrogenation. These methods utilize commercially available starting materials and standard unit operations suitable for multi-kilogram scale-up.
Q: What are the primary applications of these compounds?
A: Beyond human pharmaceuticals for treating systemic and local infections, these compounds exhibit potent preservative properties for organic materials and food additives due to their broad-spectrum antimicrobial activity against Gram-positive and Gram-negative bacteria.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a supply of high-quality intermediates for the development of next-generation antibiotics. Our team of expert chemists has extensively analyzed the synthetic routes outlined in CN1075499C and is fully prepared to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess. We understand that in the competitive landscape of antibacterial drug development, time-to-market is essential, and our optimized processes are designed to accelerate your timeline without compromising on quality or safety standards.
We invite you to collaborate with us to leverage these advanced chemical technologies for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities align with your strategic goals. By partnering with us, you gain access to a reliable supply chain partner dedicated to supporting the global fight against antimicrobial resistance through superior chemical innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
