Scalable Synthesis of Chiral Rivastigmine Intermediates via Advanced Ruthenium Catalysis
Scalable Synthesis of Chiral Rivastigmine Intermediates via Advanced Ruthenium Catalysis
The pharmaceutical industry continuously seeks robust methodologies for producing chiral active pharmaceutical ingredients (APIs) with exceptional optical purity and economic viability. Patent CN102381988B introduces a groundbreaking preparation method for the key intermediate of Rivastigmine, specifically targeting the synthesis of compound VII, (S)-3-(1-(N,N-dimethylamino)ethyl)phenol. This technology represents a significant leap forward from traditional chiral separation techniques, utilizing a sophisticated asymmetric catalytic hydrogenation strategy that ensures high chemical and optical purity. By leveraging a specialized Ruthenium-chiral phosphine-chiral amine complex, this process achieves enantiomeric excess values exceeding 99%, addressing the critical demand for high-quality neurodegenerative disease therapeutics. The strategic implementation of this route not only enhances product quality but also streamlines the supply chain for global pharmaceutical manufacturers seeking reliable sources of complex chiral building blocks.
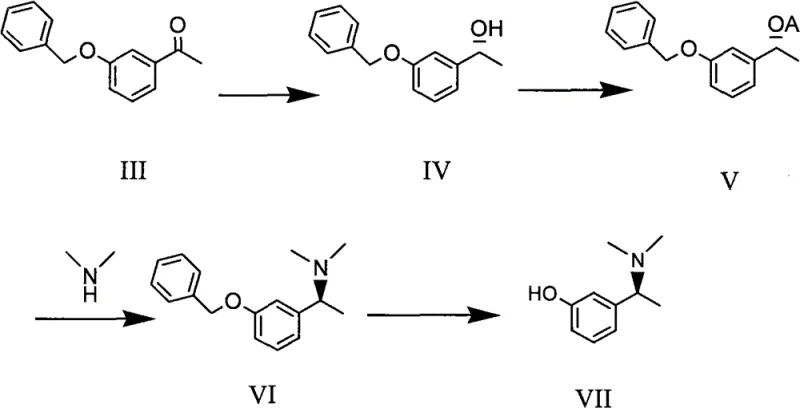
Rivastigmine, known commercially as EXELON, is a potent acetylcholinesterase inhibitor used effectively in the treatment of presenile dementia and Alzheimer's disease. The biological activity of this molecule is strictly dependent on its stereochemistry, with the S-configuration being the active constituent. As illustrated in the structural representation, the presence of a chiral carbon atom necessitates precise control during synthesis to avoid the formation of inactive or potentially harmful R-isomers. The patent discloses a comprehensive pathway that begins with simple aromatic ketones and proceeds through a series of highly controlled transformations, culminating in the target phenol derivative. This approach underscores the importance of modern catalytic science in overcoming the limitations of classical organic synthesis, providing a scalable solution for the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral amines like the Rivastigmine intermediate has relied heavily on chiral resolution technologies or stoichiometric chiral induction. Traditional resolution methods, which utilize agents such as tartrate or camphorsulfonic acid to separate enantiomers, suffer from an inherent theoretical maximum yield of only 50%. This inefficiency results in the wastage of half the produced material, leading to increased raw material consumption and higher disposal costs for the unwanted isomer. Furthermore, processes involving stoichiometric chiral auxiliaries often require expensive reagents and generate substantial chemical waste, complicating the environmental footprint of the manufacturing process. These conventional routes are frequently plagued by harsh reaction conditions and difficult purification steps, making them less attractive for large-scale industrial applications where cost and sustainability are paramount concerns for procurement and supply chain leaders.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN102381988B employs asymmetric catalytic hydrogenation, a technique that offers superior atom economy and efficiency. By utilizing a chiral transition metal catalyst, the synthesis directs the formation of the desired S-configuration directly from a prochiral ketone precursor, theoretically allowing for 100% conversion of the starting material into the desired enantiomer. This method eliminates the need for wasteful separation steps and significantly reduces the overall number of unit operations required. The process is characterized by mild reaction conditions, often proceeding at moderate temperatures and pressures, which enhances operational safety and reduces energy consumption. The simplicity of the post-reaction workup, often involving mere filtration and concentration, further distinguishes this novel approach as a highly viable candidate for cost-effective commercial manufacturing of complex pharmaceutical intermediates.
Mechanistic Insights into Ru-Catalyzed Asymmetric Hydrogenation
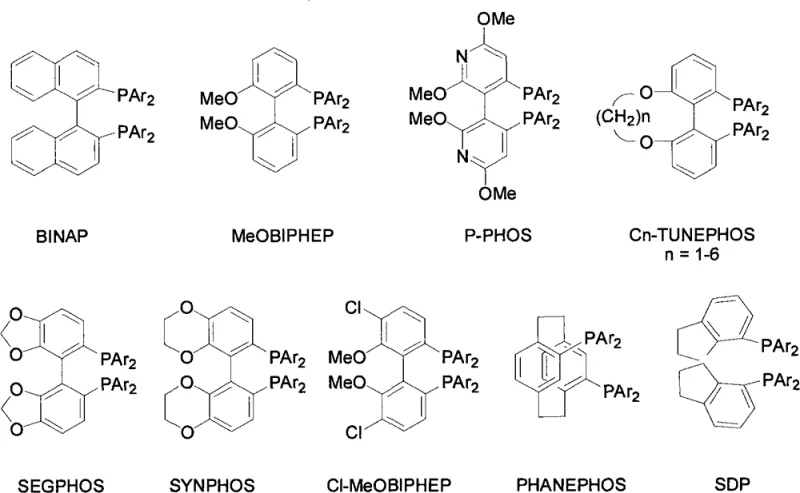
The core of this technological breakthrough lies in the utilization of a specific Ruthenium-based catalyst system, typically comprising a chiral diphosphine ligand and a chiral diamine co-ligand. The patent highlights the effectiveness of complexes such as RuCl2(s)xylBinap(s)-Daipen, where the interplay between the bulky biaryl phosphine ligand and the chiral diamine creates a highly stereoselective environment around the metal center. During the hydrogenation of the benzyloxy-substituted acetophenone derivative, the catalyst facilitates the transfer of hydrogen to the carbonyl group with exceptional facial selectivity. The steric and electronic properties of ligands like BINAP, SEGPHOS, or SDP, as shown in the library of potential ligands, are critical in dictating the orientation of the substrate within the catalytic pocket, thereby ensuring the exclusive formation of the desired alcohol stereoisomer.
Beyond the primary hydrogenation step, the mechanism ensures rigorous control over impurity profiles, which is a key concern for R&D directors focused on regulatory compliance. The high turnover number (TON) of the catalyst, reported to reach levels as high as 50,000 in optimized embodiments, indicates that the metal complex remains active and stable throughout the reaction cycle without decomposing into species that could contaminate the product. This stability minimizes the presence of residual heavy metals in the final API, simplifying the purification burden. Furthermore, the subsequent steps involving the activation of the hydroxyl group via mesylation or tosylation, followed by nucleophilic displacement with dimethylamine, proceed with retention or predictable inversion of configuration, preserving the optical integrity established in the initial hydrogenation step. This mechanistic robustness guarantees a final product with an enantiomeric excess (ee) value consistently above 99%, meeting the stringent quality standards required for neurological medications.
How to Synthesize Rivastigmine Intermediate Efficiently
The synthesis of the target compound involves a logical sequence of reactions designed to maximize yield and purity while minimizing operational complexity. The process initiates with the protection of the phenolic hydroxyl group in 3-hydroxyacetophenone via etherification with benzyl chloride, creating a robust intermediate suitable for subsequent reduction. Following this, the pivotal asymmetric hydrogenation step installs the chiral center with high fidelity. The resulting chiral alcohol is then activated to a leaving group, such as a mesylate, which undergoes nucleophilic substitution with dimethylamine to introduce the amine functionality. The final step involves the removal of the benzyl protecting group through catalytic hydrogenolysis, yielding the free phenol intermediate ready for final carbamylation into Rivastigmine. For the detailed standardized operating procedures and specific reaction parameters, please refer to the guide below.
- Perform nucleophilic substitution between 3-hydroxyacetophenone and benzyl chloride to form the protected ketone intermediate.
- Execute asymmetric catalytic hydrogenation using a Ru-BINAP-Daipen complex to establish the chiral center with high enantiomeric excess.
- Activate the alcohol group via esterification or sulfonylation, followed by nucleophilic substitution with dimethylamine and final catalytic debenzylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic benefits regarding cost structure and supply reliability. The shift from resolution-based methods to asymmetric catalysis fundamentally alters the cost equation by eliminating the 50% yield penalty associated with discarding the wrong enantiomer. This dramatic improvement in atom economy means that less raw material is required to produce the same amount of active ingredient, directly lowering the variable cost of goods sold. Additionally, the high efficiency of the catalyst allows for minute loading levels, reducing the expenditure on expensive precious metals like Ruthenium. The simplified downstream processing, which often avoids complex chromatographic separations in favor of crystallization or extraction, further reduces solvent consumption and waste disposal costs, contributing to a leaner and more sustainable manufacturing operation.
- Cost Reduction in Manufacturing: The implementation of this high-TON catalytic system significantly lowers the cost burden associated with chiral reagents and metal catalysts. By achieving near-quantitative yields and high optical purity in fewer steps, the process reduces the overall consumption of solvents and energy. The elimination of resolution steps removes the need for expensive resolving agents and the associated recycling processes, leading to substantial operational savings. Furthermore, the robustness of the reaction conditions allows for the use of standard industrial equipment without the need for specialized low-temperature or high-vacuum infrastructure, capitalizing on existing manufacturing assets to drive down capital expenditure requirements.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials and robust catalytic systems ensures a stable and continuous supply of critical intermediates. Unlike enzymatic processes that may suffer from batch-to-batch variability or sensitivity to feedstock impurities, this chemical catalytic approach offers consistent performance across large scales. The ability to source ligands and metal precursors from established global suppliers mitigates the risk of single-source bottlenecks. Moreover, the high purity of the intermediate reduces the likelihood of production delays caused by failed quality control tests, ensuring that downstream API synthesis schedules are met with greater predictability and confidence by logistics planners.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been validated from gram-scale laboratory experiments to potential multi-ton production runs. The use of common solvents like isopropanol and methanol, which are easily recovered and recycled, aligns with green chemistry principles and facilitates compliance with increasingly strict environmental regulations. The low catalyst loading minimizes the generation of heavy metal waste, simplifying effluent treatment and reducing the environmental footprint of the facility. This alignment with sustainability goals not only future-proofs the supply chain against regulatory changes but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains. The answers reflect the balance between high-performance chemistry and practical industrial application.
Q: What is the primary advantage of the Ru-catalyzed method over traditional resolution?
A: The Ru-catalyzed asymmetric hydrogenation avoids the theoretical 50% yield loss inherent in chiral resolution, significantly improving atom economy and reducing raw material costs.
Q: How does the catalyst loading impact production costs?
A: The patent demonstrates extremely low catalyst loading (TON up to 50,000), which drastically reduces the cost contribution of expensive transition metals and simplifies downstream purification.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method utilizes robust conditions, common solvents like isopropanol, and simple workup procedures like filtration, making it highly adaptable for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivastigmine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of effective neurodegenerative therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic chemistry described in patent CN102381988B can be seamlessly transferred to an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and GC-MS instrumentation to guarantee that every batch of Rivastigmine intermediate meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence ensures that the high enantiomeric excess and chemical purity achieved in the lab are preserved and enhanced during commercial manufacturing.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains and reduce time-to-market for critical medications. By leveraging our expertise in asymmetric catalysis and process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your strategic partner in delivering high-quality, cost-effective solutions for the global healthcare market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
