Scalable Synthesis of High-Purity Rivastigmine Intermediates via Asymmetric Catalysis
Scalable Synthesis of High-Purity Rivastigmine Intermediates via Asymmetric Catalysis
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of active pharmaceutical ingredients (APIs) and their critical precursors. A significant breakthrough in this domain is detailed in patent CN102381988A, which outlines a superior method for preparing intermediate compound VII of Rivastigmine, a key therapeutic agent for treating presenile dementia. This technology represents a paradigm shift from traditional chiral separation techniques to advanced asymmetric catalytic hydrogenation, offering a reliable pharmaceutical intermediate supplier with a distinct competitive edge. By leveraging a highly efficient Ruthenium-based catalyst system, this process achieves exceptional optical purity and chemical yield while drastically simplifying post-processing operations. For procurement and supply chain leaders, this innovation translates directly into enhanced supply security and reduced manufacturing overheads, addressing the perennial challenges of cost and consistency in complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral intermediates like those required for Rivastigmine has relied heavily on chiral resolution technologies or stoichiometric chiral induction. Classical resolution methods, utilizing agents such as tartrates or camphorsulfonic acids, suffer from an inherent theoretical yield limitation of 50%, as the unwanted enantiomer is typically discarded or requires costly racemization cycles. Furthermore, these processes often involve multiple crystallization steps, leading to significant solvent consumption and extended production cycles. Alternatively, stoichiometric chiral induction employs expensive chiral auxiliaries that must be attached and subsequently removed, adding unnecessary synthetic steps and generating substantial chemical waste. These conventional approaches are not only economically inefficient but also pose significant environmental burdens, making them less attractive for modern, green chemistry-compliant manufacturing facilities seeking a reliable agrochemical intermediate supplier or pharma partner.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes a catalytic asymmetric hydrogenation strategy that constructs the chiral center directly from a prochiral ketone precursor. This method bypasses the need for resolution entirely, theoretically allowing for 100% atom economy regarding the carbon skeleton. The process employs a specialized transition metal catalyst that facilitates the transfer of hydrogen with high stereoselectivity, ensuring that the desired S-configuration is formed predominantly. 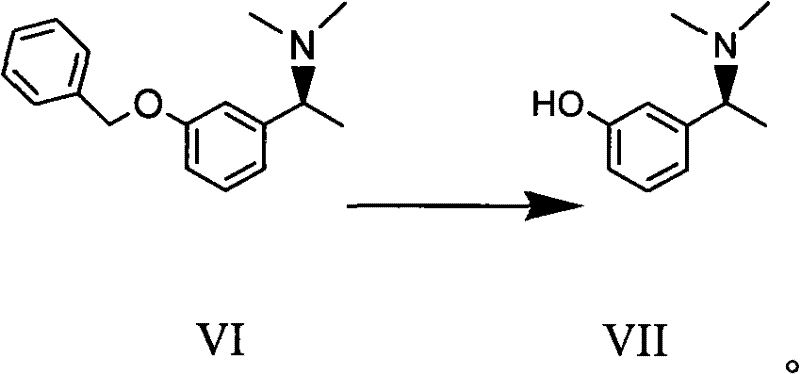 As illustrated in the transformation of compound VI to VII, the final deprotection step is equally streamlined, utilizing standard catalytic hydrogenolysis conditions that are easily scalable. This holistic route minimizes unit operations, reduces solvent usage, and eliminates the generation of unwanted isomeric waste, thereby offering a clear pathway for cost reduction in API manufacturing.
As illustrated in the transformation of compound VI to VII, the final deprotection step is equally streamlined, utilizing standard catalytic hydrogenolysis conditions that are easily scalable. This holistic route minimizes unit operations, reduces solvent usage, and eliminates the generation of unwanted isomeric waste, thereby offering a clear pathway for cost reduction in API manufacturing.
Mechanistic Insights into Ru-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the sophisticated design of the catalyst system, specifically the Ruthenium complex designated as Formula IX in the patent documentation. This catalyst comprises a chiral diphosphine ligand, such as BINAP or its derivatives like xylBinap, coordinated with a chiral diamine ligand like Daipen. The synergy between the atropisomeric diphosphine and the chelating diamine creates a rigid chiral environment around the Ruthenium center. During the reaction, the prochiral ketone substrate coordinates to the metal center, and the hydride transfer occurs through a highly organized transition state that favors the formation of one enantiomer over the other.  This precise stereocontrol is what enables the process to achieve enantiomeric excess (ee) values exceeding 99%, a critical specification for regulatory compliance in pharmaceutical production. The mechanism avoids the formation of racemic mixtures, thereby simplifying the downstream purification burden significantly.
This precise stereocontrol is what enables the process to achieve enantiomeric excess (ee) values exceeding 99%, a critical specification for regulatory compliance in pharmaceutical production. The mechanism avoids the formation of racemic mixtures, thereby simplifying the downstream purification burden significantly.
Furthermore, the impurity profile of this reaction is exceptionally clean due to the high specificity of the catalyst. Unlike non-catalytic reductions which might reduce other functional groups or lead to over-reduction, this system is tuned to selectively reduce the ketone moiety while leaving the benzyl ether protection group intact. The use of mild reaction conditions, typically involving hydrogen pressures between 150 to 500 psi and temperatures ranging from 25 to 100 degrees Celsius, further mitigates the risk of thermal degradation or side reactions. The result is a crude product with high HPLC purity, often above 98%, which reduces the need for extensive chromatographic purification. This level of control over the reaction trajectory is essential for maintaining batch-to-batch consistency, a key metric for any high-purity OLED material or pharmaceutical intermediate manufacturer.
How to Synthesize Rivastigmine Intermediate Efficiently
The synthesis of the target intermediate involves a logical sequence of transformations starting from readily available commodity chemicals. The process begins with the protection of the phenolic hydroxyl group, followed by the critical asymmetric hydrogenation step to install chirality. Subsequent functionalization involves activating the alcohol and displacing it with dimethylamine, finally concluding with the removal of the protecting group. This modular approach allows for flexibility in process optimization and ensures that each step can be monitored and controlled effectively. The detailed standardized synthesis steps see the guide below.
- Perform nucleophilic substitution between 3-hydroxyacetophenone and benzyl chloride to form the protected ketone intermediate.
- Execute asymmetric catalytic hydrogenation using a Ruthenium-BINAP-Daipen complex to establish chirality with high enantiomeric excess.
- Activate the alcohol group followed by nucleophilic substitution with dimethylamine to install the amine functionality.
- Remove the benzyl protecting group via catalytic hydrogenolysis to yield the final phenolic intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By avoiding exotic chiral pool starting materials or expensive resolving agents, the process relies on bulk commodities that are widely available from multiple global sources. This diversification of the supply base mitigates the risk of single-source bottlenecks and price volatility, ensuring a more stable and predictable procurement landscape. Additionally, the reduction in the number of synthetic steps directly correlates to shorter lead times for high-purity pharmaceutical intermediates, allowing for faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of the 50% yield loss associated with classical resolution. By converting nearly all starting material into the desired chiral product, the effective cost per kilogram of the active intermediate is significantly lowered. Furthermore, the catalyst system operates with extremely low loading levels, with turnover numbers reaching up to 50,000 or higher, which minimizes the cost contribution of the precious metal Ruthenium. The simplified workup procedures, often requiring only filtration and evaporation rather than complex chromatography, also reduce labor and utility costs, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the catalytic hydrogenation step ensures high reproducibility across different batches and scales. The process tolerates minor variations in reaction parameters without compromising optical purity, which reduces the rate of batch failures and reworks. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers. Moreover, the use of common solvents like alcohols and ethers, rather than specialized or hazardous chlorinated solvents in every step, simplifies logistics and storage requirements, further enhancing the resilience of the supply chain against regulatory or transportation disruptions.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns perfectly with green chemistry principles. The high atom economy means less waste generation per unit of product, reducing the burden on waste treatment facilities. The absence of stoichiometric chiral auxiliaries eliminates a major source of solid waste. The process is designed for commercial scale-up of complex pharmaceutical intermediates, capable of transitioning smoothly from kilogram-scale laboratory trials to multi-ton annual production without losing efficiency. This scalability ensures that as demand for Rivastigmine grows, the supply of its intermediates can expand concurrently without requiring disproportionate increases in capital expenditure or environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What is the enantiomeric purity achievable with this Ru-catalyzed route?
A: The patented process utilizing the RuCl2(s)xylBinap(s)-Daipen catalyst system consistently achieves enantiomeric excess (ee) values exceeding 99%, often reaching up to 99.9% under optimized conditions.
Q: How does this method improve upon traditional chiral resolution?
A: Unlike classical resolution which theoretically caps yield at 50% due to isomer discarding, this asymmetric synthesis builds chirality directly, offering near-quantitative theoretical yields and eliminating the need for expensive resolving agents.
Q: Is the catalyst loading cost-effective for industrial scale-up?
A: Yes, the process operates with extremely low catalyst loadings, with turnover numbers (TON) ranging from 5,000 to 50,000 or higher, significantly reducing the cost contribution of the precious metal catalyst per kilogram of product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivastigmine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the route described in CN102381988A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to ensure ee values meet or exceed 99%. Our commitment to quality ensures that every batch of Rivastigmine intermediate delivered meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic advantages tailored to your volume requirements. We encourage you to contact us for specific COA data and route feasibility assessments to validate the compatibility of this process with your downstream operations. Let us collaborate to drive efficiency and innovation in your API supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
