Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
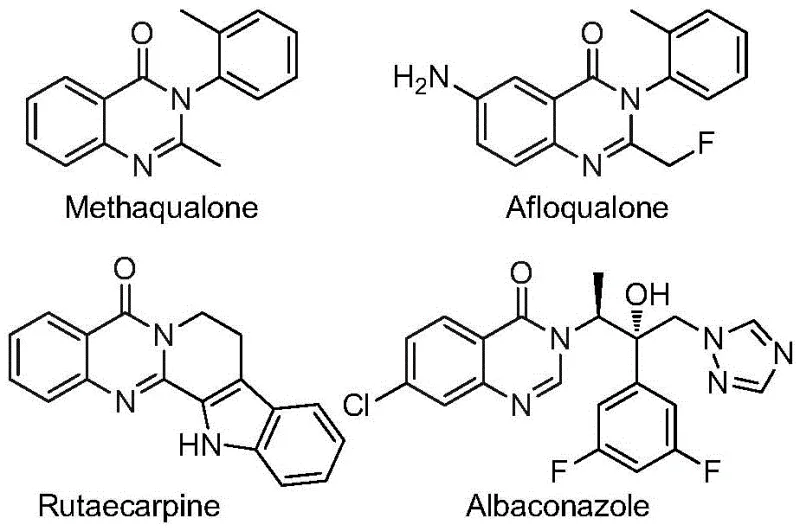
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN112480015B introduces a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a class of compounds renowned for their potent biological activities ranging from antifungal to anticancer properties. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by leveraging a palladium-catalyzed carbonylation cascade that utilizes readily available nitro compounds and trifluoroethylimidoyl chlorides. For R&D directors and procurement specialists, this technology represents a significant leap forward in process efficiency, offering a pathway to high-purity intermediates without the need for hazardous high-pressure carbon monoxide infrastructure. The strategic incorporation of the trifluoromethyl group further enhances the metabolic stability and lipophilicity of the resulting drug candidates, making this synthetic route highly valuable for modern medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that impose severe constraints on scalability and operational safety. Traditional routes often necessitate the use of pre-activated substrates, such as 2-bromoformylaniline or acid anhydrides, which are not only expensive but also require additional synthetic steps to prepare, thereby increasing the overall cost of goods. Furthermore, many established protocols depend on high-pressure carbon monoxide atmospheres to facilitate carbonylation, requiring specialized autoclaves and rigorous safety protocols that are difficult to implement in standard laboratory or pilot plant settings. Other methods utilizing iron or ruthenium catalysts frequently suffer from narrow substrate scope and moderate yields, limiting their utility for generating diverse libraries of analogs required for structure-activity relationship (SAR) studies. These inefficiencies create significant supply chain vulnerabilities and delay the timeline for bringing new active pharmaceutical ingredients (APIs) to market.
The Novel Approach
In stark contrast, the method disclosed in patent CN112480015B revolutionizes the synthesis landscape by employing a transition metal palladium-catalyzed carbonylation cascade that operates under remarkably mild and accessible conditions. By utilizing nitro compounds as inexpensive starting materials and molybdenum hexacarbonyl as a solid carbon monoxide surrogate, this process eliminates the need for handling toxic CO gas directly. The reaction proceeds efficiently in common organic solvents like 1,4-dioxane at 120°C, achieving high conversion rates across a broad spectrum of substrates. This one-pot strategy not only streamlines the workflow by combining multiple transformation steps into a single vessel but also significantly reduces waste generation and solvent consumption. The ability to directly couple trifluoroethylimidoyl chlorides with nitro compounds opens up new avenues for introducing the pharmacologically crucial trifluoromethyl motif with precision and reliability.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
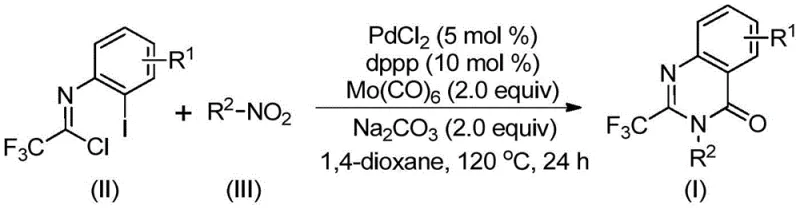
The mechanistic elegance of this transformation lies in its intricate cascade sequence, which is initiated by the in situ reduction of the nitro group. Under the reaction conditions, molybdenum hexacarbonyl serves a dual purpose: it acts as the source of carbon monoxide for the carbonylation step and facilitates the reduction of the nitro compound to the corresponding amine. Once the amine is generated, it undergoes a base-promoted intermolecular coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst, coordinated with the dppp ligand, inserts into the carbon-iodine bond of the imidoyl chloride derivative, forming a reactive divalent palladium species. The carbon monoxide released from the molybdenum complex then inserts into the carbon-palladium bond, generating an acyl-palladium intermediate that is poised for cyclization.
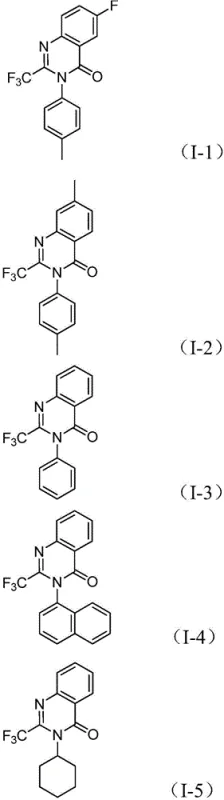
Following the carbonyl insertion, the basic environment promotes the formation of a palladium-nitrogen bond, leading to the assembly of a seven-membered cyclic palladium intermediate. This key structural arrangement undergoes reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product while regenerating the active palladium catalyst for the next cycle. This mechanism ensures high atom economy and minimizes the formation of side products, which is critical for maintaining a clean impurity profile in pharmaceutical manufacturing. The tolerance of various functional groups, including halogens and alkyl chains, suggests that the catalytic cycle is robust against steric and electronic variations, allowing for the synthesis of a wide array of derivatives such as those depicted in the patent examples. This deep mechanistic understanding provides confidence in the reproducibility and scalability of the process for industrial applications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity, leveraging the optimized conditions detailed in the patent documentation. The process is designed to be operationally simple, involving the mixing of commercially available reagents in a standard Schlenk tube or reactor, followed by heating and a straightforward workup procedure. This accessibility makes it an ideal candidate for both small-scale medicinal chemistry campaigns and larger production runs. For detailed procedural specifics regarding reagent ratios, temperature profiles, and purification techniques, please refer to the standardized synthesis guide below which outlines the critical steps for successful execution.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow for the carbonylation cascade and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers profound advantages that directly address the pain points of cost management and supply chain resilience in the fine chemical sector. By shifting away from expensive, pre-functionalized starting materials to commodity chemicals like nitro compounds, the raw material costs are drastically reduced, enhancing the overall economic viability of the production process. The elimination of high-pressure gas equipment not only lowers capital expenditure requirements but also simplifies regulatory compliance and safety audits, thereby reducing the administrative burden on manufacturing sites. Furthermore, the use of a solid CO source like Mo(CO)6 mitigates the risks associated with transporting and storing hazardous gases, ensuring a safer working environment and more stable supply continuity.
- Cost Reduction in Manufacturing: The substitution of costly activated substrates with cheap nitro compounds and the use of a one-pot protocol significantly lowers the cost of goods sold (COGS). By consolidating multiple reaction steps into a single operation, the process reduces labor hours, energy consumption, and solvent usage, leading to substantial operational savings. The high reaction efficiency and yields reported in the patent minimize material waste, further contributing to a leaner and more cost-effective manufacturing model that improves profit margins for downstream API production.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade starting materials ensures that the supply chain is less susceptible to disruptions caused by the scarcity of specialized reagents. Nitro compounds and trifluoroethylimidoyl chlorides are produced by numerous global suppliers, providing procurement teams with multiple sourcing options to mitigate risk. Additionally, the robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to diversify their production footprint and secure a steady flow of high-quality intermediates regardless of regional market fluctuations.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable, with the potential for expansion from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial production. The simplified workup procedure, involving filtration and column chromatography, is easily adaptable to industrial purification standards. Moreover, the avoidance of toxic carbon monoxide gas and the use of recyclable solvents align with green chemistry principles, helping organizations meet increasingly stringent environmental regulations and sustainability goals without compromising on output quality or volume.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the experimental data and specifications provided in the patent literature. Understanding these nuances is essential for process chemists aiming to adapt this methodology for specific project needs or for scaling up operations. The answers reflect the proven capabilities of the system as validated by the inventors, ensuring that stakeholders have accurate information for decision-making.
Q: What are the optimal reaction conditions for this synthesis?
A: The patent specifies using PdCl2 (5 mol %) and dppp (10 mol %) with Na2CO3 (2.0 equiv) and Mo(CO)6 (2.0 equiv) in 1,4-dioxane at 120°C for 24 hours.
Q: Can this method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent substrate compatibility, tolerating substituents such as halogens (F, Cl, Br), alkyl groups, and trifluoromethyl groups on both the nitro compound and the imidoyl chloride.
Q: Why is Mo(CO)6 used instead of CO gas?
A: Mo(CO)6 serves as a solid carbon monoxide substitute, releasing CO in situ under heating. This eliminates the need for high-pressure CO gas equipment, significantly enhancing operational safety and simplicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in patent CN112480015B for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to marketplace is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee the highest quality standards for every batch of 2-trifluoromethyl quinazolinone intermediates we produce.
We invite you to collaborate with us to leverage this cutting-edge technology for your next pharmaceutical project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for maximum efficiency and reliability.
