Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
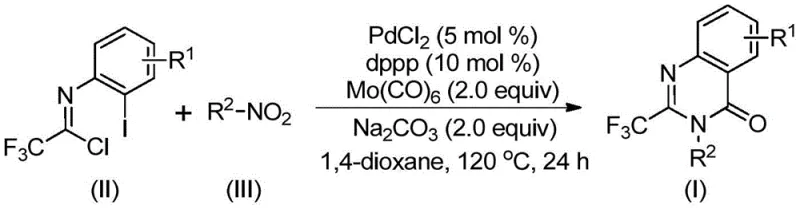
The pharmaceutical industry continuously seeks robust and efficient synthetic pathways to access privileged scaffolds that serve as the backbone for next-generation therapeutics. Among these, quinazolinone derivatives stand out due to their profound biological activities, ranging from antifungal and antiviral properties to potent anticancer effects. A recent technological breakthrough, documented in patent CN112480015B, introduces a highly efficient multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This innovation addresses critical bottlenecks in traditional heterocyclic chemistry by leveraging a palladium-catalyzed carbonylation cascade that utilizes inexpensive nitro compounds and trifluoroethylimidoyl chloride as primary building blocks. The integration of the trifluoromethyl group is particularly strategic, as fluorine atoms significantly enhance the metabolic stability, lipophilicity, and bioavailability of drug candidates, making this methodology invaluable for modern medicinal chemistry programs aiming to optimize lead compounds.
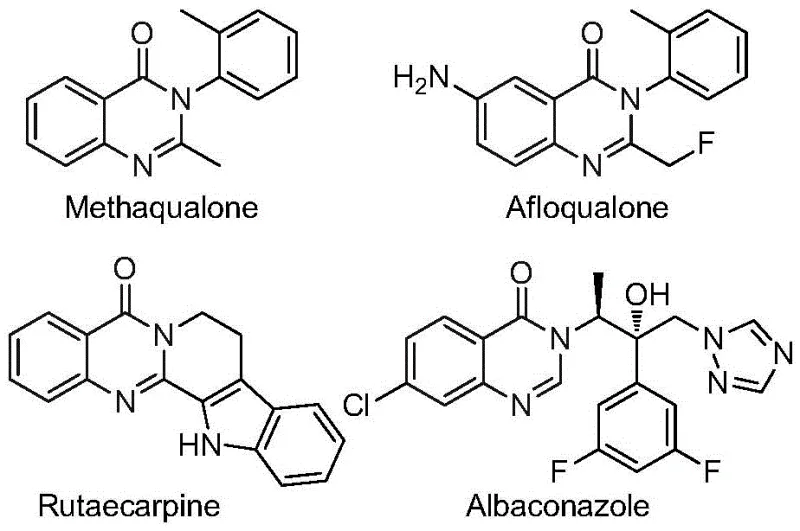
Quinazolinone compounds represent a crucial class of condensed nitrogen-containing six-membered heterocycles that are ubiquitously found in numerous functional molecules and approved pharmaceutical agents. As illustrated by well-known drugs such as methaqualone and various fluoroquinolones, this core structure is essential for eliciting specific biological responses including anti-inflammatory and anticonvulsant activities. The introduction of a trifluoromethyl group onto this scaffold further amplifies its therapeutic potential by modulating electronic properties and improving binding affinity to biological targets. However, accessing these specific fluorinated derivatives has historically been challenging due to the harsh conditions and limited substrate scope associated with conventional synthetic strategies. The new method described in the patent data offers a transformative solution, enabling the rapid assembly of diverse 2-trifluoromethyl quinazolinone libraries with high atom economy and operational simplicity, thereby accelerating the drug discovery timeline for researchers focused on oncology and infectious disease treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazolinone rings has relied on methodologies that are often fraught with significant practical and economic disadvantages for large-scale manufacturing. Traditional approaches frequently involve the use of high-pressure carbon monoxide gas, which necessitates specialized autoclave equipment and rigorous safety protocols, thereby inflating capital expenditure and operational risks. Furthermore, many established routes depend on precious metal catalysts such as ruthenium or platinum, which are not only costly but can also leave trace metal impurities that are difficult to remove to meet stringent pharmaceutical purity standards. Other methods require pre-activated substrates, such as 2-bromoformylaniline or acid anhydrides, which add extra synthetic steps and generate additional waste streams. These conventional processes are often characterized by narrow substrate tolerance, meaning that slight modifications to the molecular structure can lead to drastic drops in yield or complete reaction failure, limiting their utility in the rapid iteration required during lead optimization phases in drug development.
The Novel Approach
In stark contrast to these legacy techniques, the novel multi-component one-pot method disclosed in the patent data represents a paradigm shift towards greener and more cost-effective chemical manufacturing. This approach ingeniously utilizes molybdenum hexacarbonyl as a solid carbon monoxide surrogate, effectively eliminating the hazards and infrastructure costs associated with handling gaseous CO. By employing cheap and readily available nitro compounds as starting materials, the process bypasses the need for expensive pre-functionalized precursors, directly reducing the raw material cost basis. The reaction proceeds under relatively mild thermal conditions at 120°C in common organic solvents like 1,4-dioxane, demonstrating exceptional compatibility with a wide array of functional groups including halogens, alkyls, and alkoxy moieties. This high level of substrate flexibility allows chemists to design and synthesize a broad spectrum of substituted trifluoromethyl quinazolinone derivatives without the need for protecting group strategies, streamlining the overall synthetic sequence and significantly enhancing the throughput of medicinal chemistry campaigns.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The success of this synthetic transformation relies on a sophisticated yet elegant catalytic cycle driven by a palladium system supported by a bidentate phosphine ligand. The mechanism is believed to initiate with the in situ reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which simultaneously serves as the source of carbon monoxide for the subsequent carbonylation step. Following this reduction, a base-promoted intermolecular coupling occurs between the newly formed amine and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. The palladium catalyst then inserts into the carbon-iodine bond of the aromatic ring, forming a reactive divalent palladium species. Crucially, the carbon monoxide released from the molybdenum complex inserts into this carbon-palladium bond to create an acyl-palladium intermediate. This species then undergoes intramolecular cyclization facilitated by the nitrogen lone pair, forming a seven-membered palladacycle before finally undergoing reductive elimination to release the desired 2-trifluoromethyl substituted quinazolinone product and regenerate the active catalyst.
From a process chemistry perspective, understanding this mechanism is vital for controlling impurity profiles and ensuring consistent batch-to-batch quality. The use of sodium carbonate as a base plays a dual role in neutralizing the hydrochloric acid byproduct generated during the amidine formation and facilitating the final cyclization step. The choice of 1,3-bis(diphenylphosphino)propane (dppp) as the ligand is critical for stabilizing the palladium center throughout the high-temperature reaction, preventing catalyst decomposition and minimizing the formation of homocoupling byproducts. Furthermore, the reaction conditions are optimized to ensure that the reduction of the nitro group and the carbonylation occur in a synchronized manner within the same pot, preventing the accumulation of unstable intermediates. This mechanistic harmony results in high conversion rates and simplifies the downstream purification process, as the reaction mixture typically contains fewer side products compared to stepwise protocols, thus supporting the production of high-purity pharmaceutical intermediates required for clinical applications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The protocol is designed to be operationally simple, allowing for the direct combination of all necessary components in a single vessel, which minimizes manual handling and exposure to potentially hazardous intermediates. The process begins with the precise weighing of the palladium catalyst, ligand, and carbon monoxide source, followed by the addition of the organic solvent and substrates. Maintaining an inert atmosphere is recommended to protect the catalyst system, although the robustness of the method allows for some tolerance. The reaction is then heated to the specified temperature for a defined period, after which standard workup procedures involving filtration and chromatography are employed to isolate the final product. For detailed operational parameters and specific stoichiometric ratios validated through extensive experimentation, please refer to the standardized synthesis guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and the nitro compound substrate in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for a duration of 16 to 30 hours to allow for the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 2-trifluoromethyl substituted quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The shift towards using nitro compounds and solid CO surrogates fundamentally alters the cost structure of producing these valuable heterocyclic intermediates. By removing the dependency on high-pressure gas infrastructure and expensive noble metal catalysts like ruthenium, manufacturers can achieve significant reductions in both capital investment and ongoing operational expenditures. The simplicity of the one-pot procedure also translates to reduced labor costs and shorter cycle times, allowing facilities to increase throughput without expanding their physical footprint. Moreover, the use of commercially available and stable starting materials mitigates supply chain risks associated with sourcing specialized or hazardous reagents, ensuring a more reliable and continuous flow of materials for production schedules.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide equipment removes a major barrier to entry and reduces safety compliance costs significantly. Additionally, replacing expensive pre-activated substrates with cheap nitro compounds drastically lowers the raw material bill of materials. The high atom economy of the one-pot reaction minimizes waste generation, leading to lower disposal costs and improved overall process efficiency, which collectively contribute to a more competitive pricing structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as nitrobenzenes and simple imidoyl chlorides ensures that raw material sourcing is not bottlenecked by limited suppliers or geopolitical instability. These starting materials are produced at a massive global scale for various industries, guaranteeing long-term availability and price stability. This robustness allows supply chain planners to forecast production needs with greater confidence and reduces the likelihood of delays caused by raw material shortages, thereby securing the continuity of supply for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The method has been demonstrated to be effective on a gram scale with clear pathways for expansion to kilogram and tonnage levels, making it suitable for commercial manufacturing. The avoidance of toxic gases and the use of standard organic solvents simplify waste treatment protocols, aiding in compliance with increasingly stringent environmental regulations. The reduced complexity of the process also lowers the risk of operational errors during scale-up, ensuring that the transition from laboratory to pilot plant to full production is smooth and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing pipelines and for procurement specialists assessing the long-term viability of the supply source.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method eliminates the need for hazardous high-pressure carbon monoxide gas by using solid Mo(CO)6 as a safe CO surrogate. It operates as a one-pot multi-component reaction, significantly simplifying the workflow compared to stepwise protocols that require pre-activation of substrates or expensive ruthenium/platinum catalysts.
Q: What is the substrate compatibility for this palladium-catalyzed reaction?
A: The protocol demonstrates excellent functional group tolerance, accommodating various substituents on both the nitro compound and the imidoyl chloride. Electron-withdrawing groups like halogens and trifluoromethyl, as well as electron-donating alkyl and alkoxy groups, are well-tolerated, yielding products with high efficiency.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It utilizes cheap and readily available starting materials like nitro compounds and avoids complex purification steps beyond standard column chromatography. The robust reaction conditions and high yields make it highly viable for commercial scale-up in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate cutting-edge academic research into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from early-stage development to full-scale manufacturing. We are committed to delivering high-purity 2-trifluoromethyl quinazolinone intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in palladium-catalyzed reactions allows us to optimize this specific patent technology further, tailoring the process to meet your specific cost and quality targets while maintaining full regulatory compliance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific drug development program. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments that demonstrate our capability to support your long-term growth and innovation goals in the pharmaceutical sector.
