Advanced Edoxaban Synthesis: Scalable Routes for Global Pharmaceutical Supply Chains
Advanced Edoxaban Synthesis: Scalable Routes for Global Pharmaceutical Supply Chains
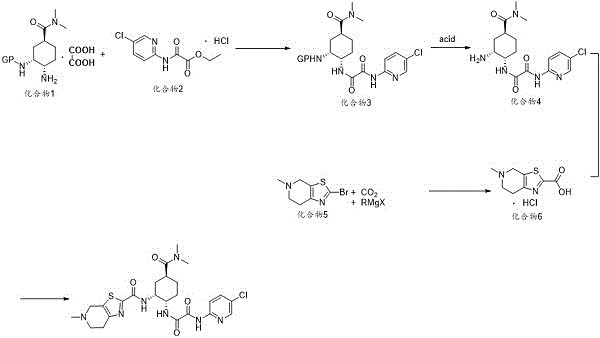
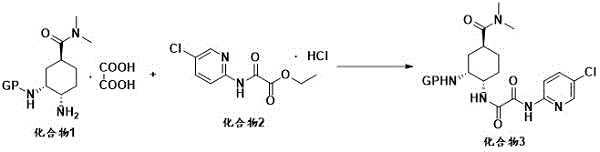
The pharmaceutical landscape for anticoagulant therapies is continuously evolving, with Edoxaban standing out as a critical oral Factor Xa inhibitor for preventing Venous Thromboembolism (VTE). Recent intellectual property developments, specifically patent CN112940012A, have unveiled a transformative preparation method for Edoxaban and its key intermediates. This technical breakthrough addresses long-standing inefficiencies in the supply chain by introducing a route that bypasses the need for extreme low-temperature conditions and expensive noble metal catalysts. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more robust and economically viable manufacturing processes. The method utilizes compound 1, compound 2, and compound 5 as starting materials, orchestrating a sequence of ammonolysis, deprotection, carboxylation, and condensation reactions. This approach not only enhances reaction yields but also significantly simplifies the purification workflow, ensuring a consistent supply of high-purity API intermediates for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Edoxaban intermediates has been plagued by significant technical and economic bottlenecks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often rely on organolithium reagents such as n-butyllithium or tert-butyllithium, which necessitate reaction temperatures below -60°C. Maintaining such cryogenic conditions requires specialized cooling systems involving liquid nitrogen and ethanol, imposing a heavy burden on infrastructure and energy costs. Furthermore, alternative methods utilizing palladium-catalyzed coupling introduce expensive noble metals and ligands into the process, creating challenges in residual metal control and waste management. Another critical failure point in prior art involves the ammonolysis step, where the use of triethylamine frequently causes the reaction system to solidify. This physical change impedes effective stirring, leading to the formation of difficult-to-remove impurities and drastically reducing the overall yield and quality of the intermediate.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent offers a streamlined pathway that eliminates the need for harsh reaction conditions and costly catalysts. By optimizing the carboxylation step with Grignard reagents and carbon dioxide under mild conditions, the process avoids the safety hazards associated with high-pressure autoclaves and toxic carbon monoxide gas. The introduction of a refined ammonolysis protocol using N,N-Diisopropylethylamine (DIEA) with a specific addition sequence prevents system solidification, ensuring homogeneous reaction conditions throughout the process. This methodological shift results in a drastic simplification of the workflow, allowing for easier separation and purification of the target compounds. Consequently, this innovation supports cost reduction in anticoagulant manufacturing by removing expensive reagents and reducing the operational complexity required for industrial production.

Mechanistic Insights into Grignard-Mediated Carboxylation and Ammonolysis
The core of this technical advancement lies in the precise control of the carboxylation and ammonolysis mechanisms, which are critical for achieving high-purity Factor Xa inhibitor intermediates. In the carboxylation step, compound 5 reacts with a Grignard reagent, such as isopropyl magnesium chloride, in an aprotic solvent like tetrahydrofuran. The introduction of carbon dioxide gas into the reaction system, followed by quenching with a metal alkali like lithium hydroxide, facilitates the formation of the carboxylic acid intermediate (compound 6) with exceptional efficiency. The molar ratio of the Grignard reagent is carefully optimized to 1:5 relative to compound 5, ensuring complete conversion while minimizing side reactions. This mechanistic precision allows the reaction to proceed at temperatures of 50°C or less, often at room temperature, which is a significant departure from the cryogenic requirements of organolithium-based routes.
Simultaneously, the ammonolysis mechanism has been re-engineered to overcome the physical limitations of previous methods. By employing DIEA as the base and adding it in two distinct stages, the process maintains a fluid reaction system that prevents the precipitation of solids which typically trap impurities. The first addition of DIEA activates compound 2 under heating, while the second addition coincides with the introduction of compound 1. This controlled environment ensures that the nucleophilic attack proceeds smoothly without the interference of heterogeneous mixing issues. The result is a significant reduction in Impurity 1, which is a common byproduct in conventional triethylamine-mediated reactions. This level of impurity control is vital for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical ingredients.
How to Synthesize Edoxaban Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters that define its efficiency and reproducibility. The process begins with the preparation of the carboxylic acid intermediate, followed by the coupling of the amine components under controlled basic conditions. The final steps involve deprotection and condensation to yield the target molecule. Each stage is designed to maximize yield while minimizing the generation of hazardous waste, aligning with modern green chemistry principles. For technical teams looking to adopt this methodology, the detailed standardized synthesis steps are crucial for ensuring batch-to-batch consistency and regulatory compliance.
- Perform carboxylation of compound 5 using Grignard reagent and CO2 with metal alkali quenching to obtain compound 6.
- Conduct ammonolysis reaction between compound 1 and compound 2 using DIEA in acetonitrile to prevent solidification.
- Execute deprotection and condensation reactions to finalize the edoxaban structure with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this novel synthesis route offers substantial cost savings and supply chain resilience by fundamentally altering the input material profile. The elimination of palladium catalysts and ligands removes a major variable cost driver, as noble metals are subject to significant market volatility and supply constraints. Furthermore, the avoidance of ultra-low temperature equipment reduces capital expenditure requirements for manufacturing facilities, allowing for production in a wider range of industrial settings. The mild reaction conditions also translate to lower energy consumption, contributing to a more sustainable and cost-effective production model. These factors combined create a robust economic argument for shifting to this new methodology, particularly for long-term supply agreements where cost stability is paramount.
- Cost Reduction in Manufacturing: The removal of expensive noble metal catalysts and the simplification of the purification process lead to significant cost optimization. By avoiding the need for specialized low-temperature infrastructure and high-pressure reactors, the overall operational expenditure is drastically reduced. The use of common, commercially available solvents and reagents further stabilizes the raw material costs, ensuring that the manufacturing process remains economically viable even at large scales. This efficiency allows for competitive pricing strategies without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and standard reaction conditions enhances the reliability of the supply chain. Unlike processes that depend on specialized organolithium reagents or toxic gases, this method utilizes stable Grignard reagents and carbon dioxide, which are easier to source and handle. The improved yield and purity reduce the need for re-processing or batch rejection, ensuring a consistent flow of materials to downstream customers. This reliability is critical for reducing lead time for high-purity API intermediates and maintaining uninterrupted production schedules for finished dosage forms.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The absence of heavy metal catalysts simplifies waste treatment and reduces the environmental footprint of the manufacturing process. This alignment with environmental compliance standards minimizes regulatory risks and facilitates faster approval for new production lines. The ability to scale up complex pharmaceutical intermediates efficiently ensures that the supply can meet growing global demand for anticoagulant therapies without bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the patent data to provide accurate guidance for stakeholders evaluating this technology. Understanding these details is essential for making informed decisions about process adoption and supply chain integration.
Q: How does the new method improve impurity control compared to traditional routes?
A: The novel ammonolysis process utilizes a specific addition sequence of DIEA and controlled heating, which prevents the reaction system from solidifying. This significantly reduces the formation of Impurity 1, ensuring higher HPLC purity compared to methods using triethylamine.
Q: Does this synthesis route require expensive palladium catalysts?
A: No, this method eliminates the need for expensive palladium catalysts and ligands required in previous coupling methods. It relies on Grignard reagents and standard condensing agents, drastically lowering raw material costs and removing heavy metal removal steps.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild, avoiding the need for ultra-low temperatures below -60°C or high-pressure autoclaves. The use of common solvents like acetonitrile and THF, along with ambient pressure carboxylation, makes it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis routes to maintain competitiveness in the global pharmaceutical market. Our CDMO expertise allows us to translate complex patent methodologies like CN112940012A into reliable commercial production processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Edoxaban intermediate meets the highest industry standards. We are committed to supporting your R&D and commercial goals through technical excellence and operational reliability.
We invite you to collaborate with us to optimize your supply chain and leverage the benefits of this innovative synthesis method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a reliable edoxaban intermediate supplier dedicated to driving value and efficiency in your anticoagulant manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
