Advanced Zinc-Catalyzed Synthesis Of Chiral Oxazoline Intermediates For Commercial Scale Production
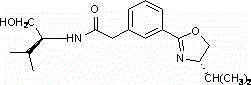
The landscape of modern pharmaceutical manufacturing is increasingly defined by the demand for high-purity chiral intermediates that serve as the foundational building blocks for complex active pharmaceutical ingredients. Patent CN102702124B introduces a significant technological advancement in this domain by detailing a robust preparation method for chiral oxazoline derivatives, specifically N-(1(S)-hydroxymethyl-1'-isopropyl)-2-[3(S)-(4-isopropyl-4,5-dihydro-2-oxazolinyl)-phenyl] acetamide. This compound represents a critical structural motif utilized extensively in the synthesis of cancer therapy drugs and other high-value therapeutic agents where stereochemical integrity is paramount for biological activity. The disclosed methodology leverages a zinc chloride-catalyzed system to achieve efficient cyclization, addressing long-standing challenges related to reaction complexity and catalyst cost that have historically plagued the production of such fine chemical intermediates. By streamlining the synthetic pathway into a more direct condensation process, this innovation offers a compelling value proposition for R&D directors seeking to optimize their supply chains for chiral building blocks while maintaining rigorous quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing chiral oxazoline rings often rely on multi-step sequences that involve the use of sensitive reagents and harsh reaction conditions which can compromise overall yield and operational safety. Many conventional protocols necessitate the utilization of precious metal catalysts or stoichiometric amounts of activating agents that generate substantial quantities of hazardous waste, thereby increasing the environmental footprint and disposal costs associated with the manufacturing process. Furthermore, older methodologies frequently require strict temperature control and extended reaction times that do not scale linearly from laboratory benchtop to industrial reactor vessels, leading to inconsistent batch quality and prolonged lead times for procurement teams. The reliance on complex protection group strategies in prior art also introduces additional unit operations such as extra purification steps, which not only dilute the overall material throughput but also increase the risk of racemization, potentially rendering the final intermediate unsuitable for enantioselective drug synthesis applications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent data utilizes a streamlined one-step synthesis that capitalizes on the Lewis acidity of zinc chloride to drive the condensation reaction between 7,7,8,8-tetracyanoquinodimethane and L-valinol. This innovative strategy eliminates the need for multiple protection and deprotection stages, thereby drastically reducing the number of processing units required and minimizing the exposure of the chiral center to conditions that could induce epimerization. The use of chlorobenzene as a solvent provides a high-boiling medium that facilitates the reflux conditions necessary for complete conversion without requiring specialized high-pressure equipment, making the process inherently safer and more adaptable to existing infrastructure. By achieving the target molecular architecture through a direct cyclization mechanism, this method significantly enhances the atom economy of the transformation and simplifies the downstream isolation procedures, allowing for a more efficient recovery of the valuable chiral oxazoline product with improved consistency.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization
The core of this technological breakthrough lies in the specific interaction between the zinc chloride catalyst and the nitrile functionalities of the starting materials, which activates the electrophilic centers for nucleophilic attack by the amino alcohol moiety. Under the influence of the Lewis acid, the cyano groups undergo a coordinated activation that lowers the energy barrier for the ring-closing step, facilitating the formation of the oxazoline heterocycle with high stereoselectivity derived from the chiral pool starting material. The reaction mechanism presumably involves the initial coordination of zinc to the nitrogen atoms, followed by an intramolecular cyclization that locks the stereochemistry in place, ensuring that the optical purity of the L-valinol precursor is faithfully transferred to the final oxazoline structure. This mechanistic pathway avoids the formation of unstable intermediates that are prone to decomposition, thereby stabilizing the reaction trajectory and allowing for the accumulation of the desired product over the extended reflux period without significant degradation.
Control of impurity profiles is critically managed through the selection of reaction parameters that favor the thermodynamic product while suppressing side reactions such as polymerization or hydrolysis of the nitrile groups. The use of a substantial excess of the amino alcohol reactant helps to drive the equilibrium towards the formation of the target amide-oxazoline hybrid structure, effectively outcompeting potential bimolecular side reactions that could generate difficult-to-remove byproducts. Additionally, the choice of chlorobenzene as the reaction medium provides a non-protic environment that prevents premature quenching of the reactive intermediates, ensuring that the cyclization proceeds to completion before workup begins. The subsequent purification via column chromatography using a petroleum ether and dichloromethane gradient allows for the precise separation of the target compound from any residual starting materials or minor isomeric impurities, guaranteeing a final product that meets the stringent specifications required for downstream pharmaceutical applications.
How to Synthesize Chiral Oxazoline Efficiently
The practical implementation of this synthesis route requires careful attention to reagent quality and moisture control to ensure optimal catalyst performance and reproducibility across different batches. Operators must prepare the reaction mixture by combining anhydrous zinc chloride with the tetracyanoquinodimethane derivative and L-valinol in dry chlorobenzene, ensuring that all components are fully dissolved before initiating the heating cycle to prevent localized hot spots. The detailed standardized synthesis steps see the guide below which outlines the precise molar ratios and thermal profiles necessary to achieve the reported conversion efficiencies while maintaining safety protocols appropriate for handling cyanide-containing precursors. Adherence to these procedural guidelines is essential for maximizing yield and ensuring that the physical properties of the isolated crystals match the characterization data provided in the intellectual property documentation.
- Prepare the reaction mixture by combining anhydrous zinc chloride, 7,7,8,8-tetracyanoquinodimethane, and L-valinol in chlorobenzene solvent under inert atmosphere.
- Heat the mixture to reflux temperature and maintain stirring for approximately 72 hours to ensure complete cyclization and conversion.
- Perform workup by removing solvent under reduced pressure, extracting with dichloromethane, drying over sodium sulfate, and purifying via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing process offers profound benefits by shifting the dependency away from scarce and volatile precious metal markets towards abundant and cost-stable commodity chemicals like zinc salts. The elimination of expensive transition metal catalysts translates directly into a significantly reduced raw material cost base, allowing procurement managers to secure more favorable pricing structures for long-term supply agreements without compromising on the quality of the intermediate. Furthermore, the simplified workup procedure involving standard extraction and distillation techniques reduces the consumption of specialized solvents and consumables, contributing to substantial cost savings in the overall operational expenditure of the production facility. This economic efficiency is compounded by the robustness of the reaction conditions, which tolerate minor variations in process parameters better than sensitive enzymatic or organocatalytic alternatives, thereby reducing the risk of batch failures and ensuring a more reliable supply continuity for downstream customers.
- Cost Reduction in Manufacturing: The substitution of noble metal catalysts with zinc chloride represents a fundamental shift in cost structure that removes the volatility associated with rhodium or palladium pricing from the bill of materials. By utilizing a catalyst that is readily available in bulk quantities at a fraction of the cost of traditional alternatives, manufacturers can achieve a drastic simplification of their cost models and pass these savings on to clients through more competitive pricing tiers. Additionally, the high atom economy of the one-step condensation reduces the volume of waste generated per kilogram of product, lowering the environmental compliance costs and waste disposal fees that often burden fine chemical production budgets significantly.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as L-valinol and chlorobenzene ensures that the supply chain is not vulnerable to the geopolitical or logistical bottlenecks that frequently affect specialized reagents. This accessibility allows for the maintenance of healthy inventory levels and rapid replenishment cycles, effectively reducing lead time for high-purity intermediates and enabling just-in-time delivery models that align with the lean manufacturing principles of modern pharmaceutical companies. The stability of the reagents also permits longer storage periods without degradation, providing supply chain heads with greater flexibility in planning and logistics management during periods of market fluctuation.
- Scalability and Environmental Compliance: The process design inherently supports commercial scale-up of complex chiral intermediates due to the absence of exothermic hazards or pressure requirements that typically limit reactor size in batch processing. The use of chlorobenzene allows for efficient solvent recovery and recycling systems that minimize volatile organic compound emissions, aligning the production process with increasingly stringent global environmental regulations and sustainability goals. This scalability ensures that production volumes can be ramped up from pilot plant quantities to multi-ton annual capacities without the need for extensive process re-engineering, securing the long-term viability of the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific chiral oxazoline synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality assurances associated with this manufacturing route for potential industry partners. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own drug substance production workflows.
Q: What are the primary advantages of using zinc chloride in this oxazoline synthesis?
A: Zinc chloride acts as an effective Lewis acid catalyst that facilitates the cyclization process under relatively mild reflux conditions, eliminating the need for expensive transition metal catalysts and simplifying the purification workflow significantly.
Q: How does this method impact the purity profile of the final chiral intermediate?
A: The one-step condensation strategy minimizes the formation of side products associated with multi-step protection-deprotection sequences, resulting in a cleaner crude reaction mixture that allows for higher final purity after standard chromatographic separation.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the use of commodity chemicals like zinc chloride and chlorobenzene, combined with a straightforward workup procedure involving extraction and distillation, makes this process highly scalable and economically viable for ton-level production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazoline Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to translate complex patent methodologies like CN102702124B into robust commercial realities. Our engineering teams have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the stereochemical integrity and chemical purity of every batch, providing our partners with the confidence needed to advance their clinical and commercial programs without delay.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your supply chain configuration. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to delivering high-quality chiral intermediates that drive innovation in the pharmaceutical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
