Advanced One-Step Synthesis of Chiral Oxazoline Ligands for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced One-Step Synthesis of Chiral Oxazoline Ligands for Commercial Scale-Up of Complex Pharmaceutical Intermediates
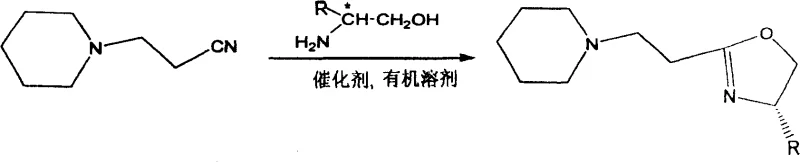
The landscape of asymmetric catalysis relies heavily on the availability of high-purity chiral ligands, yet traditional manufacturing routes often suffer from inefficiencies that hinder cost-effective production. Patent CN100425598C introduces a transformative approach to synthesizing 1-[2-(4S)-4-R-4,5-dihydro-2-oxazoline-ethyl]piperidine derivatives, utilizing a direct cyclization strategy that bypasses multiple intermediate steps. This technology leverages Lewis acid catalysis to facilitate the ring closure between hexahydropyridine propionitrile and various chiral amino alcohols, achieving yields ranging from 65% to 85% under optimized thermal conditions. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this methodology represents a significant leap forward in process intensification. By consolidating the synthesis into a single reaction vessel, the patent addresses critical pain points regarding waste generation and operational complexity, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral oxazoline ligands has been plagued by multi-step sequences that introduce substantial logistical and financial burdens to the supply chain. Traditional pathways often involve the conversion of carboxylic acids into acid chlorides using thionyl chloride or oxalyl chloride, followed by condensation with amino alcohols to form hydroxyamides before final cyclization. These legacy methods not only require stringent moisture-free environments to handle reactive acid chlorides but also generate stoichiometric amounts of corrosive byproducts that necessitate expensive waste treatment protocols. Furthermore, the isolation of unstable hydroxyamide intermediates often leads to material loss and purity degradation, complicating the downstream purification processes required for high-performance catalytic applications. The cumulative effect of these inefficiencies results in prolonged lead times and inflated manufacturing costs, making it difficult for producers to offer competitive pricing in the global market for fine chemical intermediates.
The Novel Approach
In stark contrast, the innovative route disclosed in the patent data streamlines the entire synthetic sequence into a direct, one-pot cyclization reaction driven by Lewis acid catalysts. By utilizing hexahydropyridine propionitrile as the starting nitrile source, the process eliminates the need for hazardous acid chloride activation entirely, thereby enhancing operational safety and environmental compliance. The reaction proceeds smoothly in high-boiling inert solvents such as chlorobenzene or xylene at temperatures between 115°C and 150°C, allowing for the direct formation of the oxazoline ring without isolating intermediate species. This telescoped approach significantly reduces the number of unit operations, minimizing solvent consumption and energy usage while maximizing overall throughput. For stakeholders focused on cost reduction in fine chemical manufacturing, this simplified workflow translates directly into lower capital expenditure and reduced operating expenses, establishing a robust foundation for sustainable production.
Mechanistic Insights into Lewis Acid-Catalyzed Nitrile Activation
The core of this technological breakthrough lies in the precise activation of the nitrile functionality by transition metal or rare earth catalysts, which lowers the energy barrier for nucleophilic attack by the hydroxyl group of the amino alcohol. When catalysts such as anhydrous ZnCl2, NiCl2, or rare earth chlorides are introduced into the reaction matrix, they coordinate with the nitrogen atom of the nitrile group, increasing its electrophilicity and rendering the carbon atom more susceptible to attack. This coordination facilitates the intramolecular cyclization where the hydroxyl oxygen attacks the activated nitrile carbon, followed by proton transfer and dehydration to form the stable oxazoline ring system. The choice of catalyst is critical, as different metal centers offer varying degrees of Lewis acidity and steric environments that can influence both reaction rate and stereoselectivity. Understanding this mechanistic nuance allows process chemists to fine-tune reaction conditions, ensuring that the chiral integrity of the amino alcohol substrate is preserved throughout the transformation.
Beyond the primary cyclization event, the reaction mechanism also incorporates inherent impurity control features that are vital for producing high-purity OLED material or pharmaceutical precursors. The use of specific Lewis acids helps suppress side reactions such as polymerization of the nitrile or over-alkylation, which are common pitfalls in non-catalyzed thermal cyclizations. Additionally, the selection of high-boiling solvents like chlorobenzene ensures that the reaction mixture remains homogeneous at elevated temperatures, preventing localized hot spots that could lead to decomposition or racemization of the chiral center. The subsequent workup procedure, involving aqueous extraction and standard column chromatography, effectively removes residual metal catalysts and unreacted starting materials, yielding a product with the stringent purity specifications required for downstream asymmetric catalysis applications. This level of control over the impurity profile is essential for R&D teams who require consistent batch-to-batch reproducibility for their catalytic screening programs.
How to Synthesize 1-[2-(4S)-4-R-4,5-dihydro-2-oxazoline-ethyl]piperidine Efficiently
Implementing this synthesis protocol requires careful attention to reagent quality and thermal management to achieve the reported yields of 65% to 85%. The process begins with the combination of hexahydropyridine propionitrile and the chosen chiral amino alcohol, such as L-isobutylleucinol or L-phenylleucinol, in a dry reaction vessel equipped with a reflux condenser. Anhydrous conditions are paramount during the initial setup to prevent hydrolysis of the nitrile or deactivation of the Lewis acid catalyst, which could otherwise compromise the reaction efficiency. Once the reagents are dissolved in the selected solvent, the catalyst is added at a loading of 1 to 3 wt%, and the mixture is heated to maintain a steady reflux for a duration of 40 to 50 hours. Detailed standardized synthesis steps for optimizing this pathway are provided in the guide below to ensure successful replication in your laboratory.
- Mix hexahydropyridine propionitrile and chiral amino alcohol in an inert high-boiling organic solvent such as chlorobenzene or xylene.
- Add a Lewis acid catalyst such as anhydrous ZnCl2 or rare earth chlorides at a loading of 1-3 wt% relative to raw materials.
- Reflux the mixture at 115°C to 150°C for 40-50 hours, followed by workup involving aqueous extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this direct cyclization technology offers profound strategic benefits that extend beyond simple yield improvements. By collapsing a multi-step synthesis into a single operational unit, manufacturers can drastically simplify their production schedules, reducing the dependency on complex logistics for intermediate storage and transport. This consolidation minimizes the risk of supply disruptions caused by bottlenecks in upstream processing stages, thereby enhancing the overall reliability of the supply chain for critical chiral building blocks. Furthermore, the elimination of hazardous reagents like thionyl chloride reduces the regulatory burden and safety costs associated with handling dangerous chemicals, contributing to a more sustainable and compliant manufacturing footprint. These structural efficiencies create a resilient production model capable of adapting to fluctuating market demands without sacrificing quality or delivery performance.
- Cost Reduction in Manufacturing: The transition from a multi-step acid chloride route to a one-pot nitrile cyclization fundamentally alters the cost structure of producing these valuable ligands. By removing the requirement for separate activation and isolation steps, the process significantly lowers labor costs and reduces the consumption of auxiliary materials such as drying agents and filtration media. The ability to use commodity solvents like xylene and chlorobenzene, rather than specialized anhydrous ethers or dichloromethane for every step, further drives down raw material expenses. Additionally, the higher overall yield achieved through this direct method means less starting material is wasted, maximizing the value extracted from every kilogram of input and resulting in substantial cost savings for the final buyer.
- Enhanced Supply Chain Reliability: Sourcing complex chiral intermediates often involves navigating a fragmented supplier base, but this streamlined process enables more vertical integration and local production capabilities. Since the raw materials, including hexahydropyridine propionitrile and various amino alcohols, are commercially available in bulk quantities, manufacturers are less vulnerable to shortages of exotic reagents. The robustness of the reaction conditions, which tolerate standard industrial heating and stirring equipment, ensures that production can be scaled rapidly across different facilities without extensive requalification. This flexibility allows suppliers to maintain consistent inventory levels and meet tight delivery windows, providing peace of mind to downstream users who depend on just-in-time manufacturing models.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden challenges, yet this methodology is inherently designed for large-scale operation. The absence of gas-evolving steps or highly exothermic events simplifies reactor design and heat management, allowing for safe expansion to hundred-ton scales. From an environmental perspective, the reduction in solvent volume and the avoidance of corrosive waste streams align with modern green chemistry principles, facilitating easier permitting and waste disposal. This alignment with sustainability goals not only mitigates regulatory risks but also enhances the brand reputation of companies adopting these cleaner technologies, appealing to environmentally conscious partners in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral oxazoline synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature to provide accurate guidance for potential adopters. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production lines or research pipelines. We encourage technical teams to review these insights closely to fully appreciate the operational advantages offered by this novel catalytic system.
Q: What are the primary advantages of this nitrile-based cyclization method over traditional acid chloride routes?
A: This method eliminates the need for pre-activating carboxylic acids into acid chlorides, thereby reducing the number of synthetic steps from two or more down to a single pot operation. It avoids the generation of corrosive HCl gas associated with acid chloride formation and simplifies the purification process by removing intermediate isolation requirements.
Q: Which catalysts are most effective for achieving high yields in this oxazoline synthesis?
A: The patent data indicates that transition metal chlorides like anhydrous ZnCl2 and NiCl2, as well as rare earth metal chlorides, provide excellent catalytic activity. Specifically, ZnCl2 has demonstrated yields up to 85% with benzyl-substituted substrates, while maintaining mild reaction conditions compatible with sensitive functional groups.
Q: Is this process suitable for large-scale industrial production of chiral ligands?
A: Yes, the process utilizes commercially available solvents like chlorobenzene and xylene which are standard in industrial settings. The reaction operates at atmospheric pressure with standard reflux conditions, making it highly scalable without requiring specialized high-pressure equipment, thus ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced catalytic ligands requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the enantiomeric excess and chemical purity of every batch we produce. This dedication to technical precision allows us to serve as a trusted partner for multinational corporations seeking high-purity pharmaceutical intermediates that meet the most demanding global standards.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the field of asymmetric catalysis.
