Advanced Phosphine Ligand Technology for Scalable Aldehyde Production and Cost Reduction
Advanced Phosphine Ligand Technology for Scalable Aldehyde Production and Cost Reduction
The global demand for high-purity aldehydes as critical building blocks in the pharmaceutical and agrochemical sectors continues to surge, driving the need for more efficient catalytic systems. Patent CN111333680A introduces a groundbreaking class of isophosphinoline ligands that address the longstanding stability and activity limitations of traditional catalysts in olefin hydroformylation. This technology leverages a novel fluoride-promoted synthesis route to create phosphine ligands capable of coordinating effectively with cobalt and rhodium metals. For R&D directors and procurement specialists, this represents a significant opportunity to enhance process reliability while reducing the dependency on expensive, degradation-prone ligands. The ability to synthesize these ligands in a single step from readily available isochroman salts marks a pivotal shift towards more sustainable and cost-effective fine chemical manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional homogeneous hydroformylation processes predominantly rely on rhodium-triphenylphosphine catalytic systems, which, while effective for simple terminal olefins, exhibit significant drawbacks when applied to complex substrates. The primary issue lies in the chemical instability of triphenylphosphine under prolonged reaction conditions, leading to gradual ligand degradation and subsequent loss of catalytic activity over time. Furthermore, conventional systems struggle with sterically hindered substrates such as internal olefins, trisubstituted, or tetrasubstituted alkenes, often resulting in unacceptably slow reaction rates and poor selectivity for linear aldehyde products. These inefficiencies necessitate higher catalyst loadings and more rigorous purification steps to remove metal residues, thereby inflating operational costs and extending production lead times for high-value intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a robust isophosphinoline scaffold that demonstrates superior thermal stability and coordination strength. The synthesis involves a direct, one-step reaction between isochroman salts and trisilyl phosphine, facilitated by the presence of fluoride salts which act as powerful promoters for the phosphination process. This method eliminates the need for multi-step ligand construction, significantly simplifying the supply chain for catalyst precursors. The resulting ligands maintain integrity at elevated temperatures up to 120°C, allowing for more flexible process windows and higher turnover frequencies. By enabling the efficient conversion of challenging internal olefins like 2-butene and cyclohexene, this technology opens new pathways for synthesizing complex aldehyde derivatives that were previously economically unviable.
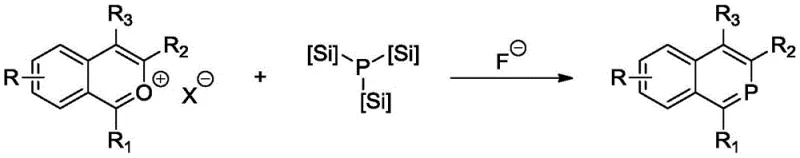
Mechanistic Insights into Fluoride-Promoted Phosphination
The core innovation lies in the unique electronic and steric properties of the isophosphinoline structure, which features a rigid fused ring system that locks the phosphorus atom in an optimal geometry for metal coordination. This structural rigidity prevents the ligand from undergoing the conformational changes that typically lead to decomposition in flexible phosphine ligands. The fluoride ion plays a critical mechanistic role by activating the silicon-phosphorus bond in the trisilyl phosphine reagent, facilitating a nucleophilic attack on the isochroman salt intermediate. This mechanism ensures high atom economy and minimizes the formation of phosphine oxide by-products, which are common impurities in traditional phosphine synthesis. The resulting metal complexes exhibit enhanced electron density at the metal center, promoting the oxidative addition of hydrogen and the subsequent insertion of olefins during the hydroformylation cycle.
Impurity control is another critical advantage provided by this mechanistic design, as the high selectivity of the catalyst reduces the formation of branched aldehyde isomers and hydrogenation by-products. The patent data indicates that the catalyst system achieves high conversion rates exceeding 99% for various substrates while maintaining selectivity for the desired linear or specific branched aldehydes. This precision reduces the burden on downstream purification units, such as distillation columns, which often consume significant energy when separating close-boiling isomers. For quality control teams, this translates to a more consistent impurity profile in the final aldehyde product, ensuring compliance with stringent pharmaceutical intermediate specifications. The stability of the ligand also means that catalyst life is extended, reducing the frequency of catalyst recharge and the associated waste generation.
How to Synthesize 1,3-Diphenylisophosphinoline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-performance ligands using standard laboratory equipment and commercially available reagents. The process begins with the preparation of the isochroman salt precursor, which is reacted with trisilyl phosphine in a polar aprotic solvent such as tetrahydrofuran under an inert nitrogen atmosphere. The addition of a fluoride salt source, such as tetramethylammonium fluoride, is the key trigger that initiates the phosphination reaction at moderate temperatures between 70°C and 100°C. Detailed standardized synthesis steps see the guide below.
- Prepare isochroman salt and trisilyl phosphine in a polar aprotic solvent such as tetrahydrofuran under nitrogen atmosphere.
- Heat the mixture to 70-100°C and slowly add a fluoride salt solution like tetramethylammonium fluoride to promote the reaction.
- Maintain the reaction temperature for 2-4 hours, then isolate the solid product through crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ligand technology offers substantial strategic benefits for procurement managers and supply chain heads looking to optimize their manufacturing footprint. The simplified one-step synthesis of the ligand itself reduces the number of unit operations required, directly lowering capital expenditure and operational complexity for in-house catalyst production. Furthermore, the enhanced stability of the catalyst system means that less precious metal is lost to degradation or difficult-to-remove residues, effectively stretching the value of every gram of rhodium or cobalt purchased. This efficiency gain is crucial in a market where precious metal prices are volatile and supply security is a top priority for long-term planning.
- Cost Reduction in Manufacturing: The elimination of complex multi-step ligand synthesis procedures significantly lowers the raw material and labor costs associated with catalyst preparation. By using readily available isochroman salts and trisilyl phosphine, manufacturers can avoid the supply chain bottlenecks often associated with specialized phosphine ligands. Additionally, the high thermal stability of the catalyst allows for operation at higher temperatures without decomposition, which can accelerate reaction rates and increase throughput without requiring expensive high-pressure equipment upgrades. The reduction in catalyst loading required to achieve high conversion further contributes to overall cost savings in the production of fine chemical intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including isochroman salts and fluoride promoters, are chemically stable and can be sourced from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the ligand under storage and reaction conditions ensures that inventory shelf-life is extended, allowing for larger batch production runs and better alignment with demand forecasting. This reliability is particularly valuable for just-in-time manufacturing models where interruption in catalyst supply can halt entire production lines. The ability to synthesize the ligand on demand also provides flexibility to scale production up or down based on market needs without long lead times.
- Scalability and Environmental Compliance: The reaction conditions described in the patent operate at atmospheric pressure and moderate temperatures, making the process inherently safer and easier to scale from kilogram to multi-ton production volumes. The use of common solvents like tetrahydrofuran and the generation of minimal hazardous by-products simplify waste treatment protocols and reduce the environmental footprint of the manufacturing process. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly strict environmental regulations in key manufacturing regions. The high selectivity of the reaction also minimizes the generation of waste streams associated with separating unwanted isomers, further enhancing the environmental profile of the process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphine ligand technology in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this catalyst system into existing hydroformylation processes.
Q: What are the stability advantages of these new phosphine ligands compared to triphenylphosphine?
A: Unlike triphenylphosphine which degrades slowly under hydroformylation conditions, these isophosphinoline ligands maintain structural stability even at temperatures up to 120°C without decomposition, ensuring consistent catalytic activity.
Q: Can this catalyst system handle sterically hindered internal olefins?
A: Yes, the patent data demonstrates high conversion rates for internal olefins such as 2-butene and cyclohexene, which are typically challenging substrates for conventional rhodium-triphenylphosphine systems.
Q: What are the typical reaction conditions for the ligand synthesis?
A: The synthesis operates under mild conditions using polar aprotic solvents at atmospheric pressure, with reaction temperatures ranging between 70°C and 100°C for a duration of 2 to 4 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphine Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN111333680A into commercial reality for our global partners. As a specialized CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of phosphine ligand or aldehyde intermediate meets the exacting standards required by the pharmaceutical and fine chemical industries. We understand that consistency is key to your downstream success, and our processes are designed to deliver that reliability batch after batch.
We invite you to collaborate with our technical procurement team to explore how this novel ligand technology can optimize your specific production challenges. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this more stable and efficient catalytic system. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you secure a competitive advantage through superior chemistry and supply chain excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
