Advanced Purification Technology for High-Purity Torasemide: Scaling Commercial Production
Introduction to Patent CN102079721B: A Breakthrough in Diuretic Purification
The pharmaceutical landscape for loop diuretics has long been dominated by the need for higher purity standards to minimize toxic side effects in patients suffering from congestive heart failure and renal insufficiency. Patent CN102079721B introduces a transformative approach not merely to synthesize, but to rigorously purify Torasemide, a next-generation sulfonylurea pyridine diuretic. Unlike traditional methods that struggle with residual impurities from multi-step synthesis, this invention provides a robust post-synthesis purification protocol that leverages alkaline hydrolysis followed by advanced ion-exchange chromatography. By treating crude Torasemide bullion with specific alkali metal alkoxides under controlled thermal conditions, the process effectively degrades ester-based impurities before utilizing macroporous strong-base resins to strip away heavy metals and endotoxins. This dual-stage purification strategy ensures that the final active pharmaceutical ingredient meets stringent global quality benchmarks, offering a viable pathway for manufacturers aiming to upgrade their product quality without reinventing the synthetic wheel.
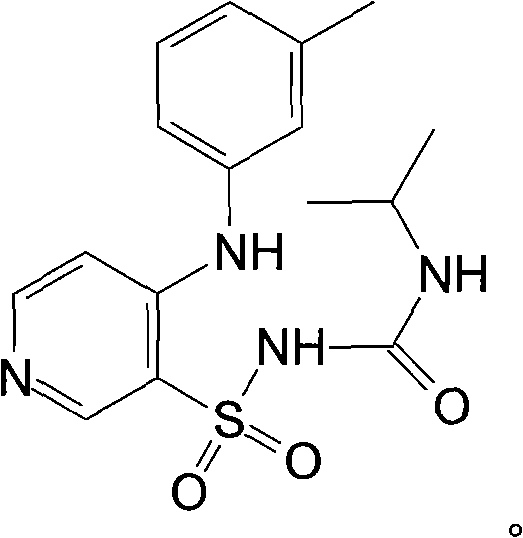
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Torasemide has relied on complex five-step synthetic routes starting from 4-pyridone, involving sulfonation, chlorination, and condensation reactions that inherently accumulate difficult-to-remove byproducts. These conventional pathways often result in crude products laden with unreacted intermediates, inorganic salts from neutralization steps, and trace heavy metals introduced by catalysts, leading to lower overall purity profiles typically below optimal therapeutic standards. Furthermore, the reliance on multiple reaction vessels and intermediate isolation steps drastically increases the operational expenditure and extends the production cycle time, creating bottlenecks for supply chain efficiency. The presence of colored impurities and organic particulates in traditionally synthesized Torasemide not only complicates downstream formulation but also raises significant regulatory concerns regarding patient safety and batch-to-batch consistency. Consequently, manufacturers face substantial challenges in meeting the increasingly rigorous impurity limits set by pharmacopoeias, often requiring expensive and time-consuming re-crystallization attempts that further erode yield.
The Novel Approach
The methodology outlined in CN102079721B circumvents these synthetic bottlenecks by focusing on a high-efficiency purification of existing crude material, thereby streamlining the manufacturing workflow into a more manageable and controllable sequence. Instead of battling impurities at every synthetic junction, this novel approach applies a targeted alkaline treatment using agents like sodium ethylate in alcoholic solvents at temperatures between 80°C and 98°C to selectively hydrolyze unstable ester impurities. Following this chemical refinement, the process employs a sophisticated adsorption step using macroporous strong-base ion exchange resins, which possess a unique physical pore structure capable of trapping non-electrolyte impurities, decolorizing the solution, and removing toxic metallic residues. This shift from exhaustive synthesis optimization to targeted purification allows for a dramatic improvement in the final product's physicochemical properties, including powder flowability and dissolution rate, while maintaining a high recovery rate that preserves economic viability. The result is a streamlined protocol that transforms low-grade bulk drug into a premium-grade API suitable for sensitive parenteral or oral formulations.
Mechanistic Insights into Alkaline Hydrolysis and Ion Exchange Adsorption
The core chemical mechanism driving the initial purification stage relies on the nucleophilic attack of alkoxide ions on ester functionalities present within the impurity profile of the crude Torasemide. Under the strongly basic conditions maintained at a pH of 11 to 13, the alkali metal alkoxide acts as a potent base that facilitates the saponification of ester contaminants, converting them into water-soluble carboxylate salts that remain in the mother liquor upon subsequent acidification. This selective degradation is critical because it targets specific organic impurities that share similar solubility characteristics with Torasemide, which would otherwise co-crystallize and degrade the purity of the final isolate. The use of lower alcohols such as ethanol or methanol as solvents ensures a homogeneous reaction medium that maximizes the contact between the alkoxide and the impurities, while the elevated temperature range of 89°C to 95°C provides the necessary activation energy to drive the hydrolysis to completion without degrading the thermally stable Torasemide backbone. This precise control over reaction kinetics ensures that the target molecule remains intact while the chemical landscape of the mixture is fundamentally altered to favor impurity removal.
Following the alkaline treatment, the second mechanistic pillar involves the physical and chemical adsorption capabilities of macroporous strong-base anion exchange resins, such as the styrene-divinylbenzene copolymer types mentioned in the patent. These resins function through a dual mechanism: ion exchange, where quaternary ammonium groups swap hydroxide ions for anionic impurities, and physical adsorption, where the vast surface area and macro-reticular pore structure trap neutral organic molecules, bacterial endotoxins, and colored bodies. Unlike traditional activated carbon which can sometimes adsorb the product itself, these specialized resins are tuned to retain impurities while allowing the Torasemide to pass through or be selectively eluted, effectively acting as a molecular sieve. The regeneration of the resin using alkali metal hydroxides allows for the recovery of the adsorbed product in a highly concentrated form, which is then subjected to a final crystallization step where pH adjustment to 6.0 induces the precipitation of ultra-pure crystals. This comprehensive removal of both ionic and non-ionic contaminants is what enables the process to consistently achieve purity levels exceeding 99.8%, addressing the critical need for high-quality raw materials in modern pharmaceutical manufacturing.
How to Synthesize Torasemide Efficiently
The implementation of this purification protocol requires precise adherence to the four-step sequence detailed in the patent to ensure reproducibility and maximum yield. Operators must begin by dispersing the crude Torasemide bullion in water and carefully adjusting the pH to the alkaline range before introducing the alkoxide solution, ensuring that the hydrolysis reaction proceeds uniformly throughout the batch.
- Treat crude Torasemide with alkali metal alkoxide (e.g., sodium ethylate) in alcohol at 80-98°C under basic conditions (pH 11-13) to hydrolyze ester impurities.
- Adjust pH to 6.0-8.0 using acid to precipitate elementary purified Torasemide, then separate via filtration.
- Dissolve the precipitate and pass through a strong-base macroporous ion exchange resin (e.g., D201) to adsorb heavy metals and colored impurities.
- Elute with alkali, concentrate, adjust pH to 6.0 for final crystallization, and dry to obtain >99.8% pure Torasemide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology represents a strategic opportunity to optimize cost structures and enhance supply reliability without compromising on quality standards. By shifting the focus from a lengthy multi-step synthesis to a streamlined purification of crude bulk, manufacturers can significantly reduce the consumption of expensive starting materials and reagents associated with the earlier stages of production. This reduction in chemical intensity directly translates to lower raw material costs and decreased waste disposal expenses, as the process eliminates the need for multiple intermediate isolation and washing steps that typically generate large volumes of solvent waste. Furthermore, the use of commercially available ion exchange resins and common solvents like ethanol ensures that the supply chain is not vulnerable to the shortages of exotic catalysts or specialized reagents, thereby stabilizing the production schedule and reducing lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of complex synthetic steps and the ability to utilize lower-grade crude Torasemide as a starting material drastically simplifies the production workflow, leading to substantial operational savings. By avoiding the capital expenditure associated with maintaining multiple reaction lines for sulfonation and chlorination, facilities can allocate resources more efficiently towards high-value purification units. Additionally, the high recovery yield of over 90% ensures that minimal product is lost during the upgrading process, maximizing the return on investment for every kilogram of crude material processed. The qualitative reduction in solvent usage and waste generation further contributes to a leaner manufacturing model that aligns with modern cost-containment strategies in the generic pharmaceutical sector.
- Enhanced Supply Chain Reliability: The reliance on standard industrial chemicals such as sodium hydroxide, hydrochloric acid, and ethanol mitigates the risk of supply disruptions that often plague specialized chemical markets. Since the ion exchange resins used, such as D201 or Amberlite IRA-900, are widely produced commodities, procurement teams can secure long-term contracts with multiple vendors to ensure uninterrupted production flows. This robustness in the supply chain is critical for meeting the continuous demand for essential diuretics in global markets, where any interruption can have significant clinical consequences. The simplicity of the process also allows for easier technology transfer between manufacturing sites, providing flexibility in sourcing and production location to mitigate geopolitical or logistical risks.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production, utilizing unit operations like fixed-bed adsorption and crystallization that are easily scalable from pilot plants to multi-ton reactors. The removal of heavy metals and toxic impurities at the source reduces the burden on downstream wastewater treatment facilities, facilitating compliance with increasingly strict environmental regulations regarding effluent discharge. By producing a higher purity product with fewer processing steps, the overall environmental footprint of the manufacturing operation is reduced, supporting corporate sustainability goals and enhancing the company's reputation as a responsible manufacturer. This scalability ensures that the technology can grow with market demand, supporting the commercial scale-up of complex diuretics without the need for prohibitive infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology, derived directly from the patent's experimental data and claims.
Q: How does this purification method improve Torasemide safety compared to standard synthesis?
A: The process utilizes strong-base macroporous ion exchange resins which effectively adsorb and remove toxic inorganic salts, heavy metals, and bacterial endotoxins that are common in crude bulk drugs, significantly reducing potential side effects.
Q: What is the expected yield and purity using this ion-exchange technique?
A: Experimental data from the patent indicates that this method consistently achieves a chemical purity of over 99.8% (up to 99.97%) with a recovery yield surpassing 90%, making it highly efficient for industrial application.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the method is explicitly designed for large-scale industrial production. It uses standard solvents like ethanol and commercially available resins (e.g., Amberlite IRA-900, D201), avoiding complex catalytic systems that hinder scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Torasemide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably replicated on an industrial scale. We are committed to delivering high-purity Torasemide that adheres to stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the >99.8% purity benchmark established by advanced purification protocols. Our facility is equipped to handle the specific requirements of ion-exchange purification and alkaline treatment, guaranteeing a supply of API that is free from heavy metals and critical impurities.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this advanced purification route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain insights into the specific economic benefits of switching to this high-yield purification method for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and supply security in the competitive diuretic market.
