Advanced Manufacturing of High-Purity Torasemide Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Torasemide Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for loop diuretics continues to evolve, driven by the persistent demand for high-quality treatments for congestive heart failure and edema. Central to this therapeutic class is Torasemide (also known as Torsemide), marketed under trademarks such as DEMADEX®, which has established itself as a potent agent for managing hypertension associated with renal and hepatic diseases. The commercial viability of this active pharmaceutical ingredient (API) relies heavily on the efficiency of its synthetic pathway, particularly the preparation of its critical precursors. Patent CN1203059C discloses a transformative methodology for synthesizing the key intermediate (3-sulfamoyl-4-chloro)pyridine, addressing long-standing inefficiencies in yield and purity that have plagued conventional manufacturing. This technical insight explores how shifting from harsh, waste-intensive protocols to a controlled organic-phase ammonolysis can redefine the supply chain economics for diuretic manufacturers.
![Chemical structure of Torasemide (1-Isopropyl-3-[(4-m-tolylamino-3-pyridyl)sulfonyl]urea)](/insights/img/torasemide-intermediate-synthesis-supplier-pharma-20260308141922-01.png)
For R&D directors and process chemists, the structural integrity and purity of the pyridine sulfonamide core are non-negotiable. The molecule functions by inhibiting the Na+/K+/2Cl- cotransporter in the thick ascending limb of the loop of Henle, and any structural deviations in the sulfonyl urea linkage or the pyridine ring can compromise efficacy or introduce toxicity. The patent highlights a specific bottleneck in the historical synthesis of Torasemide: the conversion of (3-sulfonyl chloride-4-chloro)pyridine to (3-sulfamoyl-4-chloro)pyridine. Traditionally, this step was fraught with complications, necessitating a re-evaluation of reaction conditions to meet modern regulatory and economic standards. By adopting the novel approach outlined in the intellectual property, manufacturers can secure a more robust supply of high-purity intermediates, ensuring the final API meets stringent pharmacopeial specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of (3-sulfamoyl-4-chloro)pyridine relied on reaction conditions that were inherently inefficient and environmentally burdensome. Prior art methods typically involved conducting the ammonolysis in polar solvents such as acetone or dioxane, or even more problematically, in molten reagents with a vast excess of ammonium hydroxide. In these legacy processes, the starting material, (3-sulfonyl chloride-4-chloro)pyridine (SCCPY), was often added dropwise to a large volume of aqueous ammonium hydroxide. While this dilution strategy was intended to minimize the condensation reaction between the starting sulfonyl chloride and the newly formed sulfonamide product, it created a cascade of downstream issues. The use of massive excesses of base generated substantial volumes of alkaline waste, requiring complex neutralization and disposal procedures that inflated operational costs and environmental liabilities.
Furthermore, the harsh alkaline conditions inherent to these traditional methods frequently led to significant degradation and side reactions. The yield of the desired intermediate was notoriously low, often hovering around 50%, meaning that half of the valuable starting material was lost to hydrolysis or converted into difficult-to-remove impurities. This low efficiency not only drove up the raw material costs but also necessitated rigorous and costly purification steps to isolate the product from the complex reaction mixture. For a procurement manager evaluating the cost structure of Torasemide manufacturing, these inefficiencies represent a significant leakage of value, making the final API less competitive in the generic marketplace.
The Novel Approach
The innovative method described in the patent fundamentally reengineers this critical transformation by shifting the reaction environment from an aqueous-dominated system to a controlled organic phase. Instead of drowning the reactants in excess base, the new protocol dissolves or suspends the (3-sulfonyl chloride-4-chloro)pyridine in a suitable organic solvent, such as tert-butyl methyl ether (MTBE), toluene, or acetonitrile. Ammonia, preferably in the form of a 25% aqueous ammonium hydroxide solution, is then introduced in a stoichiometrically controlled manner, typically using about 1.75 to 2.25 molar equivalents. This precise dosing prevents the localized high concentrations of base that drive side reactions, while the organic solvent provides a medium that favors the stability of the intermediate.
This strategic shift yields dramatic improvements in process performance. By avoiding the extreme conditions of the past, the novel method achieves isolated yields of approximately 74%, a substantial increase over the historical 50% benchmark. Moreover, the purity of the crude product is remarkably high, ranging from 93% to 97%, significantly reducing the burden on downstream purification units. For supply chain heads, this translates to a more predictable and reliable production schedule, as the reduced formation of by-products minimizes the risk of batch failures and extends the campaign life of purification equipment. The ability to operate at near-room temperature further enhances the safety profile of the process, making it an ideal candidate for large-scale commercialization.
Mechanistic Insights into Controlled Ammonolysis and Crystallization
The success of this novel synthesis lies in the delicate balance of nucleophilic substitution kinetics and solubility dynamics. The reaction involves the nucleophilic attack of ammonia on the sulfonyl chloride group of the pyridine ring. In conventional aqueous systems, the high concentration of hydroxide ions competes with ammonia, leading to hydrolysis of the sulfonyl chloride back to the sulfonic acid, a dead-end by-product. Additionally, the product sulfonamide retains some nucleophilic character and can react with unreacted sulfonyl chloride to form sulfonamide-sulfonamide dimers or oligomers. By utilizing an organic solvent like MTBE, the activity of the hydroxide ion is modulated, and the solubility of the organic reactants is optimized. The addition of approximately 2 molar equivalents of ammonia ensures that there is sufficient nucleophile to drive the reaction to completion without creating the excessive alkalinity that promotes hydrolysis.
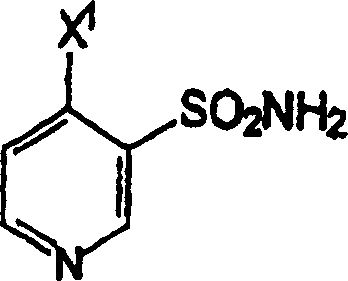
A critical aspect of the mechanism is the isolation strategy, which leverages pH-dependent crystallization. Once the reaction is complete, indicated by the stabilization of the pH, the mixture is carefully adjusted to a pH of approximately 8±0.1 using additional ammonium hydroxide. At this specific pH, the (3-sulfamoyl-4-chloro)pyridine product reaches its minimum solubility in the organic-aqueous biphasic system and precipitates out as crystals. This in-situ crystallization acts as a primary purification step, excluding soluble impurities and unreacted starting materials from the solid lattice. The result is a high-purity solid that can be easily separated by filtration. This mechanistic understanding allows process engineers to fine-tune the crystallization parameters, such as cooling rates and agitation speeds, to control particle size distribution, which is vital for efficient filtration and drying on an industrial scale.
How to Synthesize (3-Sulfamoyl-4-chloro)pyridine Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters regarding solvent selection, reagent stoichiometry, and thermal control. The process begins by suspending the chlorosulfonyl precursor in a water-miscible or partially miscible organic solvent, with MTBE being the preferred choice due to its favorable boiling point and extraction properties. The addition of the ammoniating agent must be controlled to manage the exotherm, keeping the reaction temperature within the 22°C to 26°C range to prevent thermal degradation. Following the reaction period, the precise adjustment of pH to induce precipitation is the key operational step that determines the final quality of the intermediate. Detailed standardized operating procedures for this synthesis are essential for technology transfer and scale-up.
- Suspend (3-sulfonyl chloride-4-chloro)pyridine in an organic solvent such as tert-butyl methyl ether (MTBE) at room temperature.
- Add approximately 2.15 molar equivalents of 25% aqueous ammonium hydroxide dropwise, maintaining the temperature between 22°C and 26°C.
- Adjust the suspension pH to 8±0.1 to induce crystal precipitation, then filter and dry to isolate the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this patented methodology offers compelling economic and operational benefits. The transition from a low-yield, waste-heavy process to a high-efficiency organic-phase reaction directly addresses the primary cost drivers in intermediate manufacturing. By eliminating the need for massive excesses of reagents and reducing the volume of hazardous waste generated, the process aligns with modern green chemistry principles while simultaneously lowering disposal costs. This efficiency gain is not merely theoretical; it is rooted in the tangible improvement of mass balance, where more input material is successfully converted into saleable product.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the significant increase in reaction yield. Moving from a baseline of roughly 50% to over 74% effectively reduces the consumption of the expensive (3-sulfonyl chloride-4-chloro)pyridine starting material by nearly one-third per unit of output. Furthermore, the high crude purity (93-97%) minimizes the requirement for resource-intensive recrystallization or chromatographic purification steps. This reduction in processing time and solvent usage for purification translates into substantial cost savings in utilities and labor, allowing for a more competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by complex purification bottlenecks and inconsistent batch quality. The robustness of this new method, characterized by its tolerance for standard industrial solvents like acetonitrile and MTBE, ensures that raw material sourcing is flexible and secure. The simplified work-up procedure, relying on straightforward filtration rather than complex extractions or distillations, reduces the cycle time per batch. This acceleration in throughput enables manufacturers to respond more agilely to market demand fluctuations, ensuring a steady flow of high-purity intermediates to downstream API synthesis facilities.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, particularly when dealing with strong bases and exothermic reactions. This novel approach mitigates these risks by operating at mild temperatures and avoiding the handling of molten reagents or concentrated alkaline sludges. The reduction in alkaline waste volume simplifies effluent treatment, lowering the environmental compliance burden. Additionally, the use of common, recyclable solvents facilitates the implementation of closed-loop solvent recovery systems, further enhancing the sustainability profile and long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating technology transfer and procurement officers assessing supplier capabilities.
Q: How does the new solvent system improve purity compared to traditional methods?
A: Traditional methods often utilize molten reagents or excessive ammonium hydroxide, leading to significant condensation by-products between the starting material and the product. The novel approach employs organic solvents like MTBE or acetonitrile with stoichiometric control of ammonia (approx. 2 equivalents), which drastically minimizes these side reactions, resulting in purities of 93-97% without extensive recrystallization.
Q: What are the scalability advantages of this synthesis route?
A: The process operates at mild temperatures (room temperature to 40°C) and avoids the handling of large volumes of concentrated alkaline waste associated with older protocols. The use of common industrial solvents like MTBE and acetonitrile, combined with a simple pH-triggered crystallization step, makes the reaction highly amenable to multi-kilogram and metric-ton scale production.
Q: Why is yield optimization critical for torasemide intermediates?
A: Conventional routes often suffer from yields around 50% due to competitive hydrolysis and condensation pathways. Improving the yield to over 74% effectively nearly doubles the output per batch of expensive sulfonyl chloride starting material, directly impacting the cost of goods sold (COGS) and reducing the environmental footprint of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Torasemide Intermediate Supplier
The technical potential of this improved synthesis route is immense, offering a clear pathway to higher quality and lower costs in the production of loop diuretics. At NINGBO INNO PHARMCHEM, we specialize in translating such complex laboratory innovations into robust, commercial-scale realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1203059C are fully realized in the final product. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every critical process parameter, from reaction pH to crystal morphology, guaranteeing consistency across every batch.
We invite global pharmaceutical partners to collaborate with us to leverage these advanced manufacturing capabilities. By optimizing the supply chain for critical intermediates like (3-sulfamoyl-4-chloro)pyridine, we can jointly achieve significant efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance your supply chain security and profitability.
