Advanced Purification Strategies For Paliperidone Ensuring Commercial Scalability And Regulatory Compliance
Advanced Purification Strategies For Paliperidone Ensuring Commercial Scalability And Regulatory Compliance
The pharmaceutical industry continuously demands higher standards of purity for active pharmaceutical ingredients to ensure patient safety and regulatory compliance. Patent CN101679419A introduces a groundbreaking methodology for the purification of Paliperidone, a critical antipsychotic agent, by effectively removing specific genotoxic and process-related impurities. This technical insight report analyzes the novel crystallization and slurry techniques disclosed in the patent, highlighting their potential to revolutionize the manufacturing landscape for this high-value compound. By leveraging specific solvent systems and temperature controls, manufacturers can achieve purity levels that exceed current international pharmacopoeia standards. The strategic implementation of these purification protocols offers a significant competitive advantage for suppliers aiming to dominate the global market for psychiatric medications. Furthermore, the robustness of these methods ensures consistent quality across large-scale production batches, addressing a major pain point for procurement teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
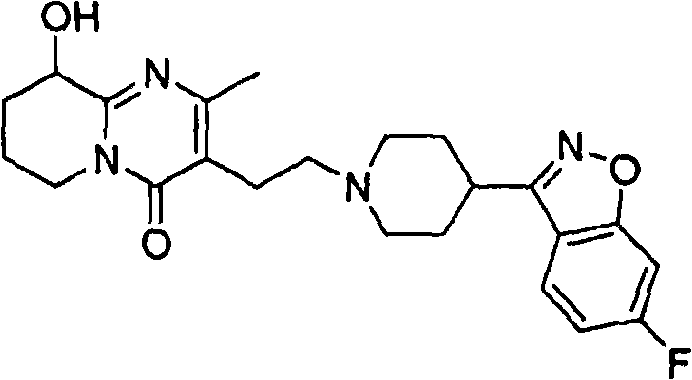
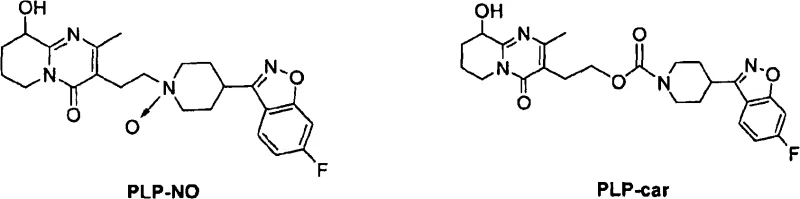
Traditional synthesis routes for Paliperidone often struggle with the persistent presence of structurally related impurities that are difficult to separate using standard workup procedures. Conventional recrystallization techniques frequently fail to adequately reduce levels of Impurity X, which possesses a relative retention time of approximately 1.27, to acceptable limits without significant yield loss. Many existing processes rely on complex chromatographic separations that are economically unviable for commercial scale manufacturing due to high solvent consumption and low throughput. Additionally, standard methods may inadvertently introduce new degradation products during prolonged heating or harsh pH adjustments required for impurity removal. The inability to consistently control the levels of PLP-car and PLP-NO impurities poses a significant regulatory risk for drug manufacturers seeking approval in stringent markets. These limitations result in extended development timelines and increased costs, creating bottlenecks in the supply chain for finished dosage forms.
The Novel Approach
The innovative approach detailed in the patent utilizes a sophisticated combination of solvent selection and thermal cycling to selectively crystallize pure Paliperidone while leaving impurities in the mother liquor. By employing specific solvent mixtures, such as acetone and water in precise volumetric ratios, the process exploits subtle differences in solubility profiles between the API and its contaminants. This method allows for the reduction of Impurity X to levels below 0.02% without the need for expensive chromatographic resin columns or excessive solvent volumes. The protocol also incorporates optional activated carbon treatment steps that effectively adsorb colored impurities and trace organic by-products prior to the final crystallization event. Such a streamlined workflow significantly simplifies the manufacturing process, making it more amenable to automation and continuous processing technologies. Ultimately, this novel approach provides a scalable and cost-effective solution that aligns perfectly with modern green chemistry principles and regulatory expectations.
Mechanistic Insights into Crystallization-Driven Purification
The core mechanism driving the success of this purification strategy lies in the precise manipulation of supersaturation levels within the chosen solvent system. When Paliperidone is dissolved in a heated solvent mixture and subsequently cooled under controlled conditions, the thermodynamic stability of the crystal lattice favors the incorporation of pure API molecules over impurity species. Impurities such as PLP-NO and PLP-car, which possess different polarity and steric configurations, remain solvated in the liquid phase due to their incompatibility with the growing crystal face of the Paliperidone. This selective exclusion is further enhanced by the use of anti-solvents that rapidly decrease the solubility of the target compound while maintaining the solubility of the contaminants. The kinetic control of nucleation and crystal growth ensures that the resulting solid form is highly ordered and free from occluded solvent or impurity pockets. Understanding these mechanistic nuances is crucial for R&D directors aiming to replicate and optimize this process for their specific manufacturing environments.
Controlling the impurity profile is not merely about removal but also about preventing the formation of new degradation products during the purification itself. The patent emphasizes the importance of temperature management, noting that excessive heat can lead to the decomposition of sensitive functional groups within the Paliperidone structure. By maintaining temperatures within a specific range, typically between 20°C and 70°C depending on the solvent, the process minimizes thermal stress on the molecule. Furthermore, the use of activated carbon acts as a scavenger for reactive species that might otherwise catalyze degradation pathways during storage or processing. This dual approach of selective crystallization and adsorptive purification ensures a comprehensive cleanup of the crude material. For quality assurance teams, this means a more robust specification sheet with tighter controls on known and unknown impurities, facilitating smoother regulatory filings and inspections.
How to Synthesize Paliperidone Efficiently
Implementing this purification protocol requires a systematic approach to solvent handling and temperature control to ensure reproducible results across different batch sizes. The process begins with the preparation of a saturated solution of crude Paliperidone in a primary solvent, followed by the careful addition of an anti-solvent to induce precipitation. Operators must adhere strictly to the specified cooling rates and stirring speeds to prevent the formation of amorphous solids or oiling out, which can trap impurities. Detailed standard operating procedures should be established to monitor critical process parameters such as pH, turbidity, and endpoint determination via HPLC analysis. The following guide outlines the fundamental steps required to execute this purification method effectively in a pilot or production setting.
- Dissolve crude Paliperidone in a selected solvent system such as acetone and water mixture at elevated temperatures.
- Cool the solution gradually to induce crystallization while maintaining specific temperature gradients to exclude impurities.
- Filter and wash the resulting crystals with cold solvent to remove surface impurities and dry under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this advanced purification technology offers substantial benefits that extend beyond mere technical compliance. Procurement managers and supply chain heads are increasingly pressured to reduce costs while ensuring uninterrupted supply of high-quality raw materials. This patented method addresses these challenges by simplifying the manufacturing workflow, which directly translates to lower operational expenditures and reduced dependency on specialized separation equipment. The ability to source Paliperidone with consistently high purity reduces the risk of batch rejections and downstream processing failures, thereby enhancing overall supply chain reliability. Moreover, the scalability of the crystallization process ensures that suppliers can meet surging market demand without compromising on quality standards. These factors collectively contribute to a more resilient and cost-efficient supply network for psychiatric pharmaceutical products.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic steps significantly lowers the cost of goods sold by reducing solvent consumption and waste disposal fees. By relying on standard crystallization equipment rather than specialized resin columns, manufacturers can capitalize on existing infrastructure without heavy capital investment. The high yield associated with this purification method ensures that raw material costs are amortized over a larger quantity of saleable product. Additionally, the reduced processing time allows for faster turnover of production batches, optimizing facility utilization rates. These efficiencies culminate in a more competitive pricing structure for the final API, providing tangible value to downstream formulation partners.
- Enhanced Supply Chain Reliability: Consistent purity profiles minimize the variability in downstream tablet compression and coating processes, leading to fewer production stoppages. Suppliers utilizing this method can guarantee tighter specifications, which reduces the need for extensive incoming quality control testing by customers. The robustness of the solvent systems used ensures that raw material availability is not a bottleneck, as these chemicals are commodity items with stable global supply chains. This reliability fosters stronger long-term partnerships between API manufacturers and pharmaceutical companies. Consequently, the risk of supply disruptions due to quality disputes is drastically mitigated, ensuring a steady flow of medication to patients.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production without significant re-engineering of the core chemistry. The use of common organic solvents facilitates easier recovery and recycling, aligning with increasingly strict environmental regulations regarding volatile organic compound emissions. Reduced waste generation lowers the environmental footprint of the manufacturing site, enhancing the corporate social responsibility profile of the supplier. Furthermore, the simplified workflow reduces the potential for operator error, ensuring consistent compliance with Good Manufacturing Practices. This alignment with sustainability goals makes the supply chain more attractive to environmentally conscious investors and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Paliperidone using the methods described in patent CN101679419A. These answers are derived directly from the experimental data and claims presented in the intellectual property documentation. They serve to clarify the capabilities and limitations of the technology for potential partners and technical evaluators. Understanding these details is essential for making informed decisions regarding sourcing and process adoption.
Q: What is the primary impurity targeted in this purification method?
A: The process specifically targets Impurity X, which has a relative retention time of approximately 1.27, along with PLP-car and PLP-NO derivatives.
Q: Can this method achieve pharmaceutical grade purity?
A: Yes, the described crystallization and slurry techniques can reduce total impurities to below 0.1%, achieving overall purity levels exceeding 99.9%.
Q: Is activated carbon filtration effective for this API?
A: Activated carbon filtration is highly effective as a supplementary step to adsorb colored impurities and trace organic contaminants before final crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paliperidone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of Paliperidone meets the highest international standards. We understand the critical nature of supply continuity in the pharmaceutical sector and have optimized our operations to deliver consistent results. By partnering with us, you gain access to a team of experts dedicated to solving complex synthesis challenges and ensuring regulatory compliance. Our facility is equipped to handle the specific solvent systems and temperature controls required for this advanced purification method.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to our optimized supply chain. Our team is ready to provide specific COA data and route feasibility assessments to support your internal review processes. Let us help you secure a reliable source of high-purity Paliperidone that drives your product success. Contact us today to initiate a conversation about your supply chain optimization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
