Advanced Manufacturing of Paliperidone: A Breakthrough in Yield and Purity Control
Advanced Manufacturing of Paliperidone: A Breakthrough in Yield and Purity Control
The pharmaceutical industry constantly seeks robust, scalable, and high-purity synthetic routes for critical antipsychotic agents. Patent CN115974870A, published in April 2023, introduces a significant technological advancement in the preparation of Paliperidone (CAS: 144598-75-4), a widely used atypical antipsychotic medication. This innovation addresses long-standing challenges in the synthesis of this complex molecule, specifically focusing on the final coupling step to form the target compound (1). The core breakthrough lies in the strategic utilization of EDTA.2Na.2H2O (Disodium ethylenediaminetetraacetic acid dihydrate) as a reaction additive. By incorporating this chelating agent into the nucleophilic substitution reaction between the chloroethyl-pyridopyrimidinone derivative and the fluorobenzisoxazole-piperidine moiety, the inventors have achieved a dramatic improvement in both reaction yield and product aesthetics. This development is particularly relevant for reliable pharmaceutical intermediates suppliers aiming to optimize their production lines for cost efficiency and regulatory compliance.
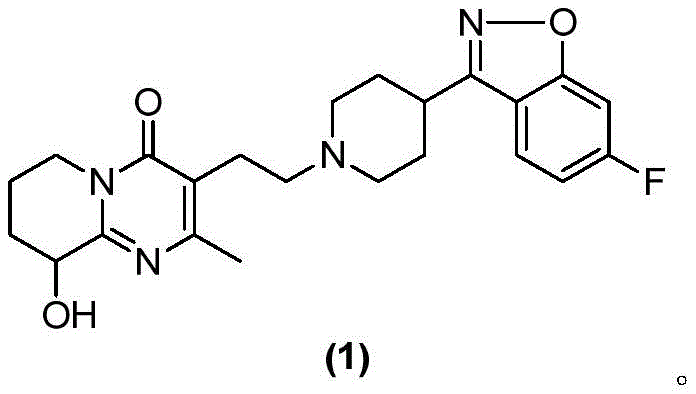
The structural complexity of Paliperidone demands precise control over impurities, especially colored by-products that often necessitate expensive purification steps. The traditional synthesis pathways often struggle with discoloration and moderate yields, creating bottlenecks in the supply chain for high-purity API intermediates. The method disclosed in CN115974870A offers a compelling solution by modifying the reaction environment at a molecular level. Instead of relying solely on thermal energy or excessive base loading, the process leverages the metal-chelating properties of EDTA to sequester trace metal ions that typically catalyze oxidative degradation or polymerization side reactions. This results in a cleaner reaction profile, allowing for the isolation of the target compound as a white or off-white solid directly from the reaction mixture, thereby streamlining the entire manufacturing workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies, including those documented in patents such as CN101641353B, WO2012134445A1, and WO2008024415A2, have historically defined the landscape of Paliperidone synthesis. However, these established routes suffer from significant operational drawbacks that hinder large-scale commercial viability. A primary deficiency in these conventional methods is the consistently low reaction yield, which directly impacts the overall cost of goods sold (COGS) and material throughput. Furthermore, the crude products obtained from these older processes are frequently contaminated with deeply colored impurities, often appearing as yellow solids. To meet stringent pharmaceutical quality standards, manufacturers are forced to employ labor-intensive and solvent-heavy purification techniques, most notably column chromatography. This reliance on chromatographic separation is not only economically inefficient due to high solvent consumption and silica gel costs but also poses significant challenges for commercial scale-up of complex pharmaceutical intermediates, as column loading capacities are limited and batch times are extended.
The Novel Approach
In stark contrast to the limitations of the prior art, the novel approach detailed in CN115974870A revolutionizes the synthesis by introducing a simple yet highly effective modification: the addition of EDTA.2Na.2H2O. This new methodology transforms the reaction outcome, shifting the product appearance from an undesirable yellow to a commercially acceptable white or off-white solid. Experimental data within the patent demonstrates that this additive facilitates a substantial increase in yield, with specific embodiments reporting yields exceeding 90%, compared to roughly 64-74% in comparative examples lacking the additive. The process operates under mild conditions, typically utilizing methanol as a solvent and DIPEA as a base at temperatures around 65°C. By eliminating the absolute necessity for column chromatography and relying instead on straightforward crystallization and filtration, this route significantly reduces the environmental footprint and operational complexity, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into EDTA-Assisted Nucleophilic Substitution
The efficacy of this synthetic route can be attributed to the specific role of EDTA as a chelating agent within the reaction matrix. During the nucleophilic substitution between the chloroethyl group of Compound (2) and the secondary amine of Compound (3), trace transition metal ions—often present as impurities in reagents or leached from reactor walls—can act as unintended catalysts for oxidative side reactions. These metal-catalyzed pathways often lead to the formation of conjugated impurities that manifest as yellow discoloration in the final product. By introducing EDTA.2Na.2H2O at a molar ratio of approximately 1:0.01 relative to the substrate, these free metal ions are effectively sequestered into stable, non-reactive complexes. This "cleaning" of the reaction medium prevents the degradation of the sensitive benzisoxazole and pyrimidinone rings, preserving the integrity of the molecule and ensuring high chemical purity.
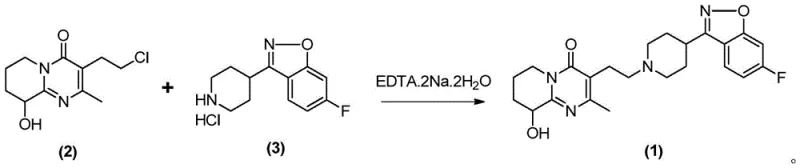
Furthermore, the presence of EDTA appears to stabilize the transition state or prevent the accumulation of basic impurities that could otherwise promote elimination reactions or polymerization. The patent data indicates that without EDTA, even under identical temperature and base conditions (e.g., using DIPEA at 65°C), the reaction yields drop significantly, and the product color deteriorates. This suggests that the chelating agent plays a dual role: protecting the product from oxidative discoloration and maintaining a reaction environment conducive to the desired SN2 substitution mechanism. The result is a highly selective transformation that minimizes the formation of closely related structural impurities, thereby simplifying the downstream purification burden and ensuring that the final high-purity Paliperidone meets rigorous pharmacopeial specifications with minimal processing.
How to Synthesize Paliperidone Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for manufacturing teams to adopt this superior technology. The process is designed to be operationally simple, requiring standard reactor equipment and commonly available reagents, which facilitates rapid technology transfer from the laboratory to pilot and production scales. The key to success lies in the precise control of stoichiometry, particularly the molar ratio of the reactants and the catalytic loading of the EDTA additive. While the reaction is robust across a range of conditions, optimizing the solvent-to-substrate ratio and the crystallization temperature is critical for maximizing recovery and ensuring the desired physical form of the product. For detailed operational parameters, please refer to the standardized guide below.
- Charge Compound (3) and Compound (2) into a reactor with methanol solvent and DIPEA base.
- Add a catalytic amount of EDTA.2Na.2H2O (approx. 0.01 eq) to the reaction mixture.
- Heat the mixture to 65°C, monitor by HPLC, then cool to crystallize the off-white product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this EDTA-mediated synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of complex purification steps translates directly into reduced manufacturing cycle times and lower utility consumption. By removing the dependency on column chromatography, facilities can repurpose valuable floor space and reduce the inventory of hazardous solvents required for elution, leading to a safer and more compliant working environment. This process intensification allows for a more agile response to market demand fluctuations, as batch turnover rates can be significantly accelerated without compromising quality.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic simplification of the workup procedure. Traditional methods requiring column chromatography incur high costs associated with silica gel, massive volumes of organic solvents, and extended labor hours for fraction collection and analysis. By achieving high purity through simple crystallization, this new method effectively removes these variable costs. Additionally, the improved yield means that less raw material is required to produce the same amount of finished API intermediate, optimizing the utilization of expensive starting materials like the fluorobenzisoxazole derivative. This efficiency gain creates a buffer against raw material price volatility, ensuring more stable pricing for downstream customers.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that are sensitive to minor variations or rely on specialized, hard-to-source consumables. The robustness of the EDTA-assisted method, which tolerates a range of molar ratios and uses commodity chemicals like methanol and DIPEA, reduces the risk of batch failures. The ability to consistently produce white or off-white product without rework minimizes the likelihood of supply disruptions caused by out-of-specification batches. This reliability is crucial for maintaining the uninterrupted production of the final antipsychotic medication, safeguarding the patient supply chain against potential shortages.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant step forward. The reduction in solvent usage aligns with green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The mild reaction conditions (65°C) reduce energy consumption compared to high-temperature reflux processes. Furthermore, the simplicity of the isolation step (filtration and drying) makes the process inherently easier to scale from kilogram to multi-ton quantities, ensuring that commercial scale-up of complex pharmaceutical intermediates can be achieved with predictable engineering outcomes and minimal capital expenditure on specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative studies presented in CN115974870A, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific manufacturing contexts.
Q: What is the primary advantage of using EDTA in Paliperidone synthesis?
A: The addition of trace EDTA.2Na.2H2O significantly improves the reaction yield (up to 93%) and crucially controls product color, shifting it from yellow to off-white, which simplifies downstream purification.
Q: Does this method require column chromatography?
A: No, unlike prior art methods such as CN101641353B, this novel process eliminates the need for column chromatography, relying instead on simple crystallization and filtration.
Q: What are the optimal reaction conditions described in the patent?
A: The optimal conditions involve using methanol as a solvent, DIPEA as the base, a molar ratio of reactants near 1:1, and a reaction temperature of approximately 65°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paliperidone Supplier
The technological advancements described in CN115974870A highlight the evolving landscape of antipsychotic drug manufacturing, where efficiency and purity are paramount. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovative synthetic strategies to maintain competitiveness in the global pharmaceutical market. As a premier CDMO partner, we possess the technical expertise to rapidly integrate this EDTA-catalyzed protocol into our production lines. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of Paliperidone intermediate we deliver meets the highest international standards.
We invite you to leverage our technical capabilities to optimize your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this improved synthesis route can lower your total landed cost. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your upcoming projects. Let us collaborate to bring high-quality, cost-effective mental health medications to patients worldwide.
