Advanced Atosiban Purification Technology for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust methodologies to ensure the highest quality of peptide-based therapeutics, particularly for critical applications in obstetrics and gynecology. Patent CN102584953B discloses a groundbreaking purification method for Atosiban, a potent oxytocin antagonist used to prevent premature labor. This technology addresses the persistent challenges of impurity removal in complex peptide synthesis, offering a pathway to achieve purity levels exceeding 99.5%. By integrating a specialized redox treatment using trivalent iron ion salts followed by high-performance liquid chromatography (HPLC), this process effectively eliminates unoxidized impurities that traditional methods often fail to remove. For R&D directors and procurement specialists, understanding this patent is crucial as it represents a significant leap in process reliability and product consistency. The method not only ensures the structural integrity of the cyclic polypeptide but also optimizes the yield to over 85%, making it a viable candidate for large-scale commercial production. This technical advancement underscores the importance of adopting innovative purification strategies to meet stringent regulatory standards while maintaining economic feasibility in the competitive landscape of API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis and purification routes for Atosiban have historically struggled with achieving the purity levels required for direct pharmaceutical application without extensive reprocessing. Conventional solid-phase synthesis often results in crude peptide mixtures containing significant amounts of unoxidized byproducts and deletion sequences that are structurally similar to the target molecule. These impurities are notoriously difficult to separate using standard chromatographic techniques alone, often leading to substantial product loss during the purification phase. Furthermore, the reliance on iodine cyclization in older methods can introduce variability in the disulfide bond formation, which is critical for the biological activity of the peptide. The inability to consistently remove these specific oxidative impurities means that manufacturers often face batch-to-batch variability, complicating the regulatory approval process and increasing the cost of goods sold. Additionally, the use of harsh conditions or non-selective reagents in conventional workflows can degrade the sensitive amino acid residues, further compromising the overall yield and quality of the final active pharmaceutical ingredient.
The Novel Approach
The innovative process described in the patent introduces a targeted chemical treatment step that fundamentally changes the impurity profile prior to chromatographic separation. By treating the crude Atosiban peptide solution with a trivalent iron ion salt, such as ferric chloride or ferric sulfate, the method selectively reduces unoxidized impurities, rendering them more distinguishable or removable in subsequent steps. This redox pre-treatment is conducted in an acetic acid solution, which maintains the solubility of the peptide while facilitating the reaction kinetics necessary for impurity modification. Following this critical step, the process employs preparative HPLC with a perchlorate-based mobile phase, which offers superior resolution for peptide separation compared to standard buffers. The combination of chemical pre-treatment and advanced chromatographic separation allows for the isolation of Atosiban perchlorate with exceptional purity. Finally, a salt conversion step ensures the final product is in the pharmaceutically acceptable acetate form, bridging the gap between high-efficiency purification and clinical safety requirements.
Mechanistic Insights into Iron-Mediated Purification and HPLC Separation
The core of this purification strategy lies in the specific interaction between the trivalent iron ions and the sulfur-containing impurities present in the crude peptide mixture. Atosiban contains a disulfide bridge between the Mpa and Cys residues, which is essential for its cyclic structure and receptor binding affinity. During synthesis, incomplete oxidation can lead to free thiol groups or incorrect disulfide pairings that act as persistent impurities. The addition of ferric ions acts as a mild oxidizing or complexing agent that targets these sulfur species, effectively cleaning the mixture before it enters the chromatographic column. This mechanism prevents the co-elution of impurities with the main product peak, a common issue in peptide purification that drastically reduces recovery rates. By controlling the concentration of the iron salt to be not less than 2% of the crude peptide weight, the process ensures complete reaction without introducing excessive metal contamination that would require difficult downstream removal. This precise chemical manipulation demonstrates a deep understanding of peptide chemistry, allowing for a cleaner feed into the HPLC system and maximizing the efficiency of the separation media.
Following the redox treatment, the separation mechanism relies on the differential partitioning of the peptide and its impurities between the stationary and mobile phases under acidic conditions. The use of perchloric acid in the mobile phase, adjusted to a pH between 2.5 and 3.2, protonates the peptide residues in a way that enhances their interaction with the octadecylsilane (C18) stationary phase. This specific pH range is critical for maintaining the peptide's stability while achieving sharp peak resolution. The linear gradient elution with acetonitrile allows for the precise elution of the Atosiban perchlorate, separating it from any remaining hydrophobic or hydrophilic contaminants. 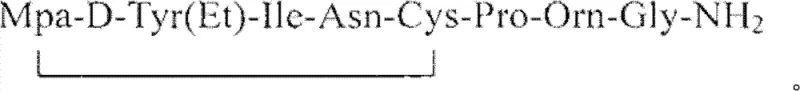 The subsequent conversion to the acetate salt involves a second chromatographic step using ammonium acetate buffers, which displaces the perchlorate ions through ion exchange mechanisms on the column. This two-stage chromatographic approach ensures that the final product is not only chemically pure but also possesses the correct counter-ion profile required for stability and bioavailability in the final drug formulation.
The subsequent conversion to the acetate salt involves a second chromatographic step using ammonium acetate buffers, which displaces the perchlorate ions through ion exchange mechanisms on the column. This two-stage chromatographic approach ensures that the final product is not only chemically pure but also possesses the correct counter-ion profile required for stability and bioavailability in the final drug formulation.
How to Synthesize Atosiban Efficiently
Implementing this purification protocol requires precise control over solvent composition, reagent stoichiometry, and chromatographic parameters to ensure reproducible results. The process begins with the dissolution of the crude peptide in acetic acid concentrations ranging from 50% to 100%, followed by the controlled addition of the iron salt reagent. Operators must monitor the dilution of the acetic acid to below 50% after the reaction to prepare the solution for HPLC loading, ensuring compatibility with the aqueous mobile phase. The chromatographic separation is performed using columns with dimensions scalable from 5cm to 45cm in diameter, allowing for flexibility from pilot to production scale. Detailed standard operating procedures regarding flow rates, gradient times, and fraction collection thresholds are essential to maintain the high purity specifications of over 99.5%. For a comprehensive guide on the exact operational parameters and equipment setup required for this synthesis, please refer to the standardized technical steps provided below.
- Dissolve crude Atosiban peptide in 50-100% acetic acid and treat with trivalent iron ion salt (e.g., ferric chloride) to reduce unoxidized impurities.
- Separate the treated solution using preparative HPLC with a perchlorate and acetonitrile mobile phase system to isolate Atosiban perchlorate.
- Convert the purified Atosiban perchlorate into the final acetate salt form using reversed-phase chromatography with ammonium acetate buffers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates into tangible improvements in supply reliability and cost structure. The ability to achieve high yields in a single batch significantly reduces the raw material consumption per kilogram of final product, directly impacting the cost of goods. By eliminating the need for multiple recrystallization steps or extensive reprocessing to meet purity standards, the manufacturing cycle time is drastically simplified, allowing for faster turnaround on orders. The scalability of the method, demonstrated by its capacity to handle over 1000g per batch, ensures that suppliers can meet large-volume demands without compromising on quality or consistency. This robustness reduces the risk of supply interruptions caused by batch failures, a critical factor for pharmaceutical companies managing tight production schedules. Furthermore, the use of standard chromatographic resins and common reagents like acetic acid and iron salts ensures that the supply chain for manufacturing inputs remains stable and cost-effective, avoiding reliance on exotic or expensive proprietary catalysts.
- Cost Reduction in Manufacturing: The elimination of complex and costly purification steps traditionally required to remove oxidative impurities leads to substantial cost savings in the overall production process. By integrating the iron salt treatment, the process reduces the load on the HPLC columns, extending their lifespan and reducing the frequency of expensive resin replacement. The high recovery rate of over 85% means that less starting material is wasted, optimizing the utilization of expensive amino acid building blocks. Additionally, the streamlined workflow reduces labor hours and utility consumption associated with extended processing times, contributing to a more lean and efficient manufacturing operation. These efficiencies collectively lower the barrier to entry for high-quality Atosiban production, making it more accessible for generic and branded drug manufacturers alike.
- Enhanced Supply Chain Reliability: The robustness of this purification method ensures a consistent supply of high-purity Atosiban, mitigating the risks associated with batch-to-batch variability. The ability to scale the chromatographic columns from small pilot sizes to large production diameters allows suppliers to flexibly adjust output based on market demand without revalidating entirely new processes. This scalability is crucial for maintaining continuity of supply during periods of high demand or raw material fluctuations. Moreover, the use of stable and widely available reagents reduces the dependency on single-source suppliers for critical manufacturing inputs, further strengthening the resilience of the supply chain. For procurement teams, this means a lower risk of delays and a more predictable delivery schedule for this critical obstetric medication.
- Scalability and Environmental Compliance: The process is designed with environmental sustainability in mind, utilizing aqueous acetic acid and acetonitrile systems that are well-understood in terms of waste management and solvent recovery. The high efficiency of the purification reduces the volume of waste generated per unit of product, aligning with green chemistry principles and regulatory expectations for pharmaceutical manufacturing. The ability to process large batches efficiently means fewer runs are needed to meet production targets, thereby reducing the overall energy footprint and solvent consumption of the facility. This compliance with environmental standards not only avoids potential regulatory fines but also enhances the corporate social responsibility profile of the manufacturing partner. For supply chain leaders, partnering with a facility that employs such efficient and compliant technologies ensures long-term viability and reduces the risk of production shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this Atosiban purification technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential manufacturing partners. Understanding these details is essential for R&D teams evaluating process transfer and procurement officers assessing supplier capabilities. The answers provided reflect the specific technical parameters and performance metrics outlined in the intellectual property, ensuring accuracy and relevance for decision-making.
Q: How does the iron salt treatment improve Atosiban purity?
A: The process utilizes trivalent iron ions as a specific reducing agent to target and remove unoxidized impurities inherent in the crude peptide solution before chromatographic separation, significantly enhancing the final purity profile.
Q: What is the scalability of this purification method?
A: The method is designed for industrial application, capable of processing over 1000g of pure peptide in a single batch, meeting the rigorous demands of commercial pharmaceutical manufacturing.
Q: Why is the conversion to acetate salt necessary?
A: While perchlorate salts are effective for HPLC separation, the final conversion to the acetate form ensures the product meets standard pharmaceutical safety and stability specifications required for clinical use.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atosiban Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity peptides in the development of safe and effective pharmaceutical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN102584953B can be seamlessly transferred to industrial manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Atosiban meets the highest international standards. Our facility is equipped with state-of-the-art preparative HPLC systems and lyophilization capabilities, allowing us to handle complex peptide purifications with precision and efficiency. By leveraging our expertise in peptide chemistry and process optimization, we provide a reliable source for high-quality Atosiban intermediates and APIs that support your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced process technologies. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements, demonstrating how our purification methods can enhance your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of technical knowledge and manufacturing capacity dedicated to delivering excellence in fine chemical production. Let us help you secure a stable and cost-effective supply of Atosiban for your pharmaceutical needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
