Advanced Semi-Synthesis of Taxol: A Scalable Route for High-Purity API Production
Advanced Semi-Synthesis of Taxol: A Scalable Route for High-Purity API Production
The global demand for Paclitaxel, widely known as Taxol, has surged due to its status as a premier broad-spectrum antineoplastic agent utilized in the treatment of ovarian, breast, and lung cancers. Historically, the reliance on extracting this potent compound directly from the bark of the Pacific Yew tree presented severe ecological and economic bottlenecks, given the extremely low natural abundance of approximately 0.007% to 0.069% in dry bark weight. To address these critical supply chain vulnerabilities, the industry has pivoted towards semi-synthetic methodologies that utilize more abundant precursors. Patent CN103130753A introduces a robust, five-step semi-synthetic protocol that transforms 10-deacetylbaccatin III (10-DAB) into high-purity Taxol. This innovative approach leverages renewable biomass from yew needles, where 10-DAB content exceeds 0.1%, offering a sustainable and commercially viable alternative to destructive harvesting practices while ensuring consistent quality for pharmaceutical manufacturing.
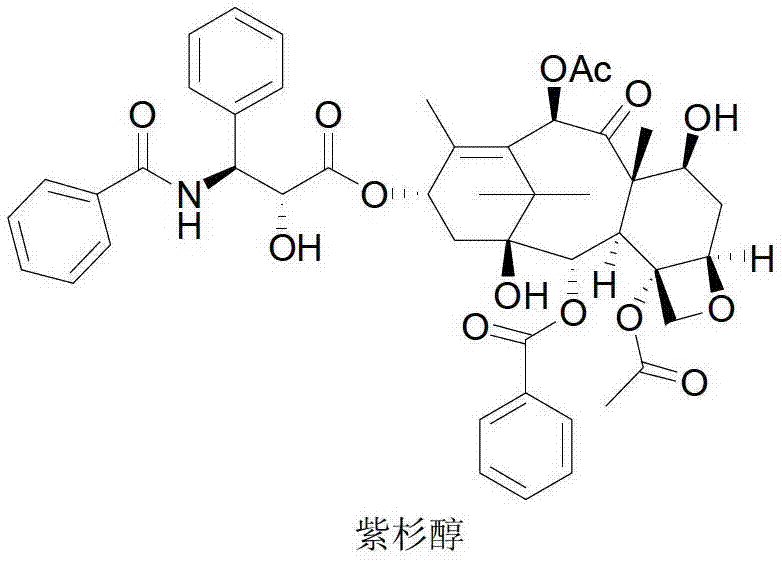
This technological breakthrough represents a significant leap forward for reliable API intermediate suppliers seeking to optimize their production portfolios. By shifting the focus from extraction to chemical synthesis, manufacturers can decouple production volumes from the seasonal and geographical limitations of plant cultivation. The method detailed in the patent not only enhances the sustainability profile of the supply chain but also introduces critical process intensifications that reduce solvent consumption and processing time. For procurement managers and R&D directors alike, understanding the nuances of this semi-synthetic pathway is essential for evaluating potential partners who can deliver cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent purity specifications required for oncology drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional semi-synthetic routes for Taxol have long been plagued by inefficiencies related to regioselectivity and harsh reaction conditions. Conventional protocols often necessitate the protection of the C7 hydroxyl group using chlorotriethylsilane (TES-Cl) prior to acetylation. This step is notoriously cumbersome, requiring a vast excess of the silylating reagent and strict low-temperature control to prevent non-selective reactions at other hydroxyl sites. Furthermore, the subsequent coupling of the C13 side chain frequently employs aggressive bases such as butyllithium or sodium bis(trimethylsilyl)amide (NaHMDS), which demand cryogenic conditions and inert atmospheres, thereby escalating operational costs and safety risks. These legacy methods often result in complex impurity profiles that necessitate extensive purification via column chromatography, a process that is difficult to scale and generates substantial chemical waste, hindering the commercial scale-up of complex polymer additives and pharmaceutical actives.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN103130753A streamlines the synthesis through a highly selective acetylation strategy that bypasses the need for initial C7 protection. By employing cerous chloride heptahydrate as a catalyst, the process achieves exclusive acetylation at the C10 position under mild conditions, preserving the integrity of the C7 and C13 hydroxyl groups for subsequent functionalization. This is followed by a specific protection step using trichloroethyl chloroformate (Troc-Cl), which offers superior stability and ease of removal compared to traditional silyl groups. The culmination of this route involves a gentle condensation reaction and a final deprotection sequence that yields the target molecule with high fidelity. This novel approach drastically simplifies the workflow, enabling a transition from laboratory curiosity to industrial reality.
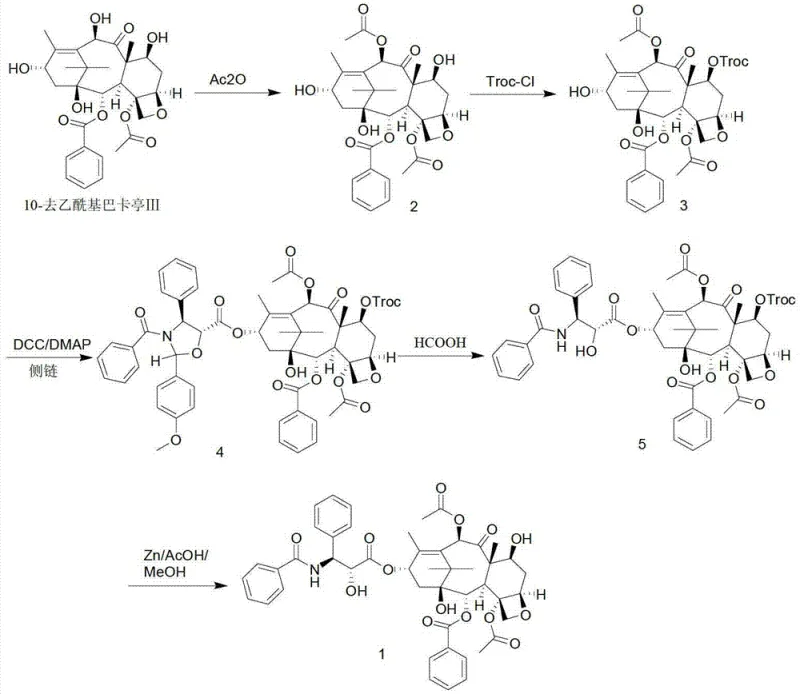
Mechanistic Insights into Cerium-Catalyzed Selective Acetylation
The cornerstone of this improved synthesis lies in the mechanistic behavior of the lanthanide catalyst, cerous chloride heptahydrate, during the initial acylation step. In the absence of a catalyst, acetic anhydride reacts indiscriminately with the multiple hydroxyl groups present on the baccatin III core, leading to a mixture of mono-, di-, and tri-acetylated byproducts that are difficult to separate. However, the presence of the cerium salt coordinates selectively with the oxygen atoms of the substrate, effectively activating the C10 hydroxyl group towards nucleophilic attack by the acetic anhydride. This coordination chemistry creates a transient complex that lowers the activation energy specifically for the C10 position, ensuring that the reaction proceeds with high regioselectivity even at temperatures ranging from 0 to 5 degrees Celsius. This precision eliminates the formation of unwanted isomers at the C7 and C13 positions, which are critical for the subsequent biological activity of the final drug.
Following the selective acetylation, the protection of the C7 hydroxyl group is achieved using Troc-Cl, a reagent chosen for its orthogonal stability. The mechanism involves the nucleophilic attack of the C7 oxygen on the carbonyl carbon of the chloroformate, displacing the chloride ion. The resulting trichloroethoxycarbonyl group is robust enough to withstand the subsequent acidic and basic conditions of the side-chain coupling and ring-opening steps but can be cleanly removed later using zinc dust in acetic acid. This specific choice of protecting group is pivotal for impurity control, as it prevents the formation of elimination byproducts that often occur with acid-labile groups. The final deprotection utilizes a single-electron transfer mechanism where zinc reduces the trichloroethyl moiety, triggering a fragmentation that releases the free hydroxyl group and volatile byproducts, leaving the sensitive taxane core intact and ready for final crystallization.
How to Synthesize Taxol Efficiently
The execution of this semi-synthetic pathway requires precise control over stoichiometry and temperature to maximize yield and minimize impurity generation. The process begins with the dissolution of 10-DAB in tetrahydrofuran, followed by the addition of the cerium catalyst and acetic anhydride at controlled low temperatures to ensure selectivity. Subsequent steps involve careful pH management during the workup phases to prevent hydrolysis of the sensitive ester linkages. The condensation with the oxazoline side chain precursor is facilitated by DCC and DMAP, which activate the carboxylic acid for amide bond formation without racemization. Finally, the removal of the Troc group and the opening of the oxazoline ring are performed in a sequential manner to reveal the active pharmacophore. For a detailed breakdown of the specific reaction parameters, stoichiometric ratios, and workup procedures, please refer to the standardized synthesis guide below.
- Perform highly selective acetylation of 10-deacetylbaccatin III at the C10 position using acetic anhydride and cerous chloride heptahydrate catalyst.
- Protect the C7 hydroxyl group using trichloroethyl chloroformate (Troc-Cl) to form the 7-Troc protected intermediate.
- Condense the protected baccatin core with the oxazoline side chain precursor using DCC and DMAP coupling agents.
- Execute acid-mediated ring opening of the oxazoline moiety followed by zinc-mediated deprotection to yield the final Taxol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented semi-synthetic route offers transformative benefits that extend beyond mere chemical efficiency. The primary value proposition lies in the drastic simplification of the downstream processing requirements. Unlike traditional methods that rely heavily on silica gel column chromatography for purification—a technique that is notoriously solvent-intensive and difficult to automate—this process allows for the isolation of pharmaceutical-grade Taxol through simple recrystallization. This shift significantly reduces the volume of organic solvents required, lowering both the direct cost of materials and the environmental burden associated with solvent disposal and recovery. Furthermore, the ability to purify via crystallization enhances the scalability of the process, allowing manufacturers to move seamlessly from pilot batches to multi-ton production runs without the bottleneck of chromatographic separation.
- Cost Reduction in Manufacturing: The elimination of cryogenic reagents and the reduction in reaction steps directly translate to lower operational expenditures. By avoiding the use of expensive and hazardous reagents like butyllithium and minimizing the need for excessive protecting group manipulations, the overall cost of goods sold is substantially decreased. Additionally, the high selectivity of the cerium-catalyzed step reduces the formation of byproducts, which in turn improves the overall mass balance and yield of the process. This efficiency means that less raw material is wasted, and the throughput of the manufacturing facility is maximized, providing a competitive edge in pricing for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing 10-DAB from yew needles rather than bark ensures a more stable and sustainable supply of starting materials. Since the needles are a renewable resource that can be harvested without killing the tree, the risk of raw material shortages due to ecological restrictions is mitigated. This stability is crucial for long-term supply contracts with major pharmaceutical companies that require guaranteed continuity of supply. Moreover, the robustness of the synthetic route means that production is less susceptible to variations in raw material quality, as the chemical steps are designed to tolerate minor fluctuations, ensuring consistent delivery schedules and reducing lead time for high-purity APIs.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing reagents and conditions that are compatible with standard stainless steel reactor infrastructure. The avoidance of column chromatography not only saves costs but also aligns with green chemistry principles by reducing hazardous waste generation. The use of zinc-mediated deprotection generates manageable inorganic waste streams that are easier to treat compared to the complex organic waste from silica columns. This environmental compatibility facilitates regulatory approval and supports the corporate sustainability goals of modern pharmaceutical manufacturers, making it an attractive option for companies looking to reduce their carbon footprint while maintaining high production standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic Taxol pathway. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing frameworks.
Q: What is the primary advantage of using Cerous Chloride in Taxol semi-synthesis?
A: Cerous chloride heptahydrate acts as a highly effective Lewis acid catalyst that enables regioselective acetylation at the C10 hydroxyl group without the need for excessive protecting groups on other positions, significantly simplifying the workflow.
Q: How does this process improve supply chain reliability compared to plant extraction?
A: By utilizing 10-deacetylbaccatin III (10-DAB) extracted from renewable yew needles rather than bark, the process ensures a sustainable raw material supply that is over ten times more abundant than direct taxol extraction, mitigating ecological risks.
Q: Does this synthetic route require complex purification methods like column chromatography?
A: No, a key commercial advantage of this patented method is that the crude product can be purified to pharmaceutical grade standards through simple recrystallization, eliminating the need for expensive and time-consuming column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taxol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global oncology market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering Taxol and its key intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. By leveraging the efficiencies of the cerium-catalyzed semi-synthetic route, we can offer our partners a supply solution that balances cost-effectiveness with the highest standards of quality and safety.
We invite you to collaborate with us to optimize your supply chain and secure a reliable source of this vital anticancer agent. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive value and stability in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
