Advanced One-Pot Synthesis of Cinchocaine Hydrochloride for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance safety, efficiency, and purity, particularly for critical local anesthetics like Cinchocaine Hydrochloride. Patent CN110981801B introduces a groundbreaking one-pot production process that fundamentally restructures the synthetic route by utilizing 2-butoxyquinoline-4-carboxylic acid as the primary starting material. This innovation addresses long-standing safety concerns associated with traditional methods while delivering exceptional controllability and yield suitable for large-scale industrial applications. By shifting away from hazardous reagents such as phosphorus pentachloride and metallic sodium, this methodology not only enhances operator safety but also streamlines the downstream purification workflow. The technical breakthrough lies in the seamless integration of acylation and amidation steps within a single reaction vessel, minimizing intermediate handling and reducing the overall environmental footprint of the manufacturing process. For global procurement teams and R&D directors, this patent represents a significant leap forward in reliable pharmaceutical intermediate supplier capabilities, ensuring a stable supply of high-quality anesthetic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cinchocaine Hydrochloride has relied on processes that introduce substantial operational risks and inefficiencies, particularly those documented in earlier patents such as US4839366. These conventional routes frequently employ phosphorus pentachloride for chlorination, a reagent known for its violent reactivity, unpleasant odor, and tendency to sublime at elevated temperatures, creating dangerous working conditions. Furthermore, the decomposition of phosphorus pentachloride generates phosphorus oxychloride, a byproduct that is notoriously difficult to separate from the desired product, often necessitating high-temperature distillation that degrades product quality. Other methods, such as those disclosed in CN106496120A, utilize metallic sodium, which presents extreme fire and explosion hazards during transportation, storage, and reaction phases. The reliance on these hazardous materials not only inflates insurance and safety compliance costs but also complicates the supply chain due to strict regulatory controls on precursor chemicals. Additionally, the need to isolate and dry intermediate compounds in these traditional multi-step processes extends production cycles and increases the likelihood of yield loss through mechanical handling.
The Novel Approach
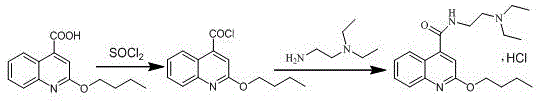
In stark contrast, the novel one-pot method described in the patent data utilizes thionyl chloride and 2-butoxyquinoline-4-carboxylic acid to achieve a safer and more efficient transformation. This approach eliminates the need for metallic sodium entirely, thereby removing the associated risks of fire and explosion while simplifying the raw material procurement strategy for the supply chain head. The reaction conditions are significantly milder, with acylation occurring at manageable temperatures between 60-70°C, avoiding the extreme thermal stress required by older phosphorus-based methods. By conducting the amidation reaction directly in the same vessel without isolating the acid chloride intermediate, the process drastically reduces solvent consumption and labor hours, leading to substantial cost savings in pharmaceutical manufacturing. The streamlined workflow ensures that the production period is shortened, enhancing the overall throughput of the facility without compromising on the quality of the final active pharmaceutical ingredient. This methodological shift exemplifies how modern chemical engineering can resolve legacy safety issues while simultaneously improving economic viability for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Thionyl Chloride-Mediated Acylation and Amidation
The core of this innovative synthesis lies in the precise control of the acylation and amidation mechanisms, which are optimized to maximize yield while minimizing side reactions. The process begins with the activation of 2-butoxyquinoline-4-carboxylic acid using thionyl chloride in a toluene solvent system, generating the reactive 2-butoxyquinoline-4-formyl chloride in situ. This acylation step is carefully monitored at temperatures ranging from 40-70°C to ensure complete conversion while preventing thermal degradation of the quinoline ring structure. Following the removal of excess thionyl chloride, the reaction mixture is cooled to sub-zero temperatures, specifically between -5-0°C, before the introduction of N,N-diethylethylenediamine. This low-temperature amidation is critical for controlling the exothermic nature of the reaction and preventing the formation of di-substituted impurities that could compromise the purity profile. The use of a biphasic system during the workup, involving acid and alkali washing, effectively removes unreacted amines and acidic byproducts, ensuring a clean organic phase prior to crystallization.
Impurity control is further enhanced through a sophisticated pH adjustment strategy during the post-reaction workup, which is essential for meeting the stringent purity specifications required by R&D directors. After the amidation is complete, the solution is treated with hydrochloric acid to adjust the pH to 2.0-3.0, facilitating the separation of the aqueous phase containing the product salt from organic impurities. Subsequent basification to pH 10.0-11.0 allows for the extraction of the free base into an organic solvent, leaving behind water-soluble inorganic salts and polar byproducts. The final crystallization step utilizes isopropanol and hydrochloric acid to precipitate the Cinchocaine Hydrochloride, a process that effectively excludes structurally similar impurities through selective solubility differences. This multi-stage purification within a single workflow ensures that the final product achieves a liquid phase purity of over 99.9%, demonstrating the robustness of the mechanistic design. Such precise control over the chemical environment underscores the feasibility of this route for producing high-purity pharmaceutical intermediates on a multi-ton scale.
How to Synthesize Cinchocaine Hydrochloride Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters and sequential addition of reagents to ensure optimal performance and safety. The process is designed to be scalable, as evidenced by successful trials in 500L reaction kettles, making it suitable for both pilot and commercial production environments. Operators must maintain strict temperature control during the addition of thionyl chloride and the subsequent amidation step to prevent runaway reactions and ensure consistent batch quality. The detailed standardized synthetic steps involve specific molar ratios and solvent volumes that have been optimized to balance reaction kinetics with downstream processing ease. For a comprehensive understanding of the operational parameters and safety protocols required for execution, the detailed standardized synthesis steps are provided in the guide below.
- Perform acylation by reacting 2-butoxyquinoline-4-carboxylic acid with thionyl chloride in toluene at 60-70°C to form the acid chloride intermediate.
- Conduct amidation by adding N,N-diethylethylenediamine to the reaction mixture at -5-0°C, followed by acid-base washing to remove impurities.
- Finalize the process by removing the solvent, adding isopropanol and hydrochloric acid, and crystallizing at -5-0°C to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this one-pot manufacturing process offers transformative advantages that directly address the pain points of procurement managers and supply chain heads regarding cost and continuity. The elimination of hazardous reagents like metallic sodium and phosphorus pentachloride significantly reduces the regulatory burden and safety compliance costs associated with raw material handling and storage. By simplifying the process flow and removing intermediate isolation steps, the facility can achieve faster turnaround times, effectively reducing lead time for high-purity pharmaceutical intermediates and improving responsiveness to market demand. The use of common solvents like toluene and isopropanol, which are readily available in the global chemical market, ensures supply chain reliability and mitigates the risk of production stoppages due to raw material shortages. Furthermore, the high yield and purity achieved reduce the need for reprocessing or scrapping batches, leading to substantial cost savings in API intermediate manufacturing through improved material efficiency.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents such as metallic sodium directly lowers the raw material cost base, while the one-pot design reduces energy consumption and solvent usage. By avoiding the isolation and drying of intermediate acid chlorides, the process saves significant labor hours and equipment time, which translates into lower overhead costs per kilogram of product. The high molar yield exceeding 82% ensures that raw material input is efficiently converted to saleable product, minimizing waste disposal costs and maximizing return on investment. These cumulative efficiencies result in a more competitive pricing structure without compromising on the quality standards required by global pharmaceutical clients.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available starting materials like 2-butoxyquinoline-4-carboxylic acid ensures a consistent supply flow, reducing the volatility often associated with specialized or controlled reagents. The simplified process flow reduces the number of potential failure points in the production line, enhancing the overall reliability of the manufacturing schedule and ensuring on-time delivery. Safety improvements mean fewer regulatory inspections and shutdowns, providing a more predictable production timeline for supply chain planners. This stability is crucial for maintaining long-term contracts with multinational pharmaceutical companies that require guaranteed continuity of supply for their drug formulations.
- Scalability and Environmental Compliance: The process has been validated at the 500L scale, demonstrating clear pathways for commercial scale-up of complex pharmaceutical intermediates to multi-ton production levels. The reduction in hazardous waste generation, particularly the avoidance of phosphorus-containing byproducts, simplifies wastewater treatment and aligns with increasingly strict environmental regulations. The use of recyclable solvents like toluene further supports sustainability goals, making the manufacturing process more attractive to environmentally conscious partners. This scalability ensures that the production capacity can grow in tandem with market demand, securing the long-term viability of the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel Cinchocaine Hydrochloride production process. These answers are derived directly from the patent data and technical analysis to provide clarity on safety, yield, and operational feasibility. Understanding these details is essential for stakeholders evaluating the potential integration of this technology into their supply chain. The responses highlight the specific advantages over legacy methods and confirm the readiness of the process for industrial adoption.
Q: Why is the one-pot method safer than conventional Cinchocaine synthesis?
A: Conventional methods often utilize phosphorus pentachloride and metallic sodium, which pose significant safety risks due to high reactivity and difficult byproduct separation. The novel one-pot process replaces these with thionyl chloride and eliminates the need for metallic sodium, drastically reducing operational hazards and simplifying waste treatment.
Q: What purity levels can be achieved with this new production process?
A: The optimized crystallization steps involving isopropanol and hydrochloric acid adjustment allow for the removal of trace impurities effectively. Industrial trials have demonstrated liquid phase purity exceeding 99.9%, meeting stringent requirements for pharmaceutical intermediates without requiring complex additional purification stages.
Q: How does this process impact production costs and scalability?
A: By combining acylation and amidation into a single pot without isolating the intermediate acid chloride, the process reduces solvent usage, energy consumption, and labor time. The elimination of expensive and hazardous reagents further lowers raw material costs, while the demonstrated 500L scale-up confirms robust commercial scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinchocaine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes that ensure both safety and economic efficiency for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like this one-pot Cinchocaine process are seamlessly transferred to full-scale manufacturing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that transitioning to a new process requires confidence in technical execution, and our team is dedicated to providing the stability and expertise needed to support your long-term production goals.
We invite you to collaborate with us to optimize your supply chain and leverage these technical advancements for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how this improved synthesis can benefit your operations. By partnering with us, you gain access to a reliable supply of high-quality intermediates backed by a commitment to continuous process improvement and customer success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
