Advanced Palladium-Catalyzed Synthesis of Multi-Substituted Conjugated Dienes for Pharmaceutical Intermediates
Introduction to Next-Generation Diene Synthesis
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient and versatile synthetic routes for complex molecular scaffolds. A significant breakthrough in this domain is detailed in patent CN111018830B, which discloses a novel method for synthesizing multi-substituted conjugated diene compounds. These structures are pivotal in medicinal chemistry, serving as robust building blocks for biologically active agents and advanced materials. The core innovation lies in the utilization of α-nitrodithioacetals as key starting materials, which undergo a transition metal-catalyzed cross-dehydrogenative coupling with acrylates or styrenes. This approach represents a paradigm shift from traditional methods, offering a streamlined pathway to access highly functionalized olefinic systems that were previously difficult to construct with such precision.
The general structural framework of these valuable intermediates is characterized by a conjugated diene system bearing a nitro group and a dithiolane moiety, as illustrated in the generalized formula below. This specific arrangement provides a unique electronic environment that facilitates further downstream transformations, making these compounds highly attractive for R&D teams focused on drug discovery. The ability to introduce diverse ester or aryl groups at the terminal position significantly expands the chemical space available for exploration. For a reliable pharmaceutical intermediate supplier, mastering such versatile chemistries is essential to meeting the dynamic demands of global clients seeking novel scaffolds for their pipeline programs.
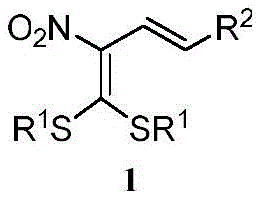
Furthermore, the strategic placement of the nitro group and the dithioacetal functionality allows for orthogonal reactivity, enabling chemists to selectively modify different parts of the molecule without compromising the integrity of the conjugated system. This level of control is critical when optimizing lead compounds where slight structural changes can dramatically alter biological activity. The patent highlights that these compounds are not merely theoretical constructs but are accessible through practical, scalable methodologies that align with modern green chemistry principles. By leveraging air as a terminal oxidant and employing mild reaction temperatures, this technology addresses both economic and environmental concerns inherent in large-scale chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted conjugated dienes has relied on methodologies that often suffer from significant drawbacks regarding operational complexity and substrate scope. Prior art, such as the work by Gaunt and Loh groups, utilized palladium-catalyzed oxidative C-H olefination strategies involving indoles or specific activated alkenes. While scientifically elegant, these conventional routes frequently require harsh reaction conditions, stoichiometric amounts of expensive oxidants like silver salts or benzoquinone, and strictly anhydrous environments. Such requirements impose a heavy burden on process chemistry teams, leading to increased waste generation and higher production costs. Additionally, many traditional methods exhibit poor functional group tolerance, limiting their applicability to simple substrates and failing to deliver the structural diversity required for modern drug development programs.
Another critical limitation of older technologies is the difficulty in controlling stereoselectivity and regioselectivity during the formation of the conjugated system. Unwanted isomers and polymeric byproducts often contaminate the crude reaction mixture, necessitating tedious purification steps that reduce overall yield and throughput. For procurement managers, these inefficiencies translate into longer lead times and unpredictable supply chains. The reliance on specialized reagents that are not commodity chemicals further exacerbates supply chain vulnerabilities. Consequently, there has been a persistent industry demand for a more robust, cost-effective, and operationally simple method to access these high-value intermediates without compromising on purity or scalability.
The Novel Approach
The methodology described in the patent data offers a compelling solution to these longstanding challenges by introducing a direct cross-dehydrogenative coupling between α-nitrodithioacetals and electron-deficient alkenes. As demonstrated in the reaction scheme below, the process utilizes readily available starting materials and operates under remarkably mild conditions. The use of palladium acetate as a catalyst in a mixed solvent system of acetic acid and dimethyl sulfoxide (HOAc/DMSO) enables the reaction to proceed efficiently at just 50°C under an air atmosphere. This eliminates the need for inert gas protection and expensive external oxidants, drastically simplifying the experimental setup and reducing the overall cost of goods sold (COGS).
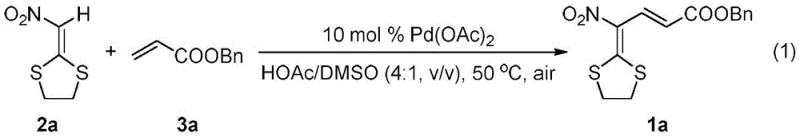
In this novel approach, the synergy between the solvent system and the catalyst is paramount. The specific volume ratio of 4:1 for HOAc to DMSO was found to be critical for maximizing yield, as evidenced by comparative experiments where pure solvents resulted in significantly lower conversion rates. The reaction tolerates a wide array of acrylate esters, including ethyl, n-butyl, and tert-butyl variants, as well as styrene derivatives, showcasing exceptional substrate adaptability. For a cost reduction in fine chemical manufacturing context, this versatility means that a single standardized protocol can be adapted to produce a library of diverse analogues, minimizing the need for extensive process re-optimization for each new target molecule. The high yields reported, such as 85% for the benzyl acrylate derivative, underscore the efficiency and reliability of this new synthetic route.
Mechanistic Insights into Pd-Catalyzed Cross-Dehydrogenative Coupling
The success of this transformation hinges on a sophisticated catalytic cycle mediated by palladium species. The mechanism likely initiates with the coordination of the palladium catalyst to the electron-rich double bond of the α-nitrodithioacetal, followed by C-H activation to generate a key organopalladium intermediate. The presence of the nitro group plays a crucial electronic role, activating the adjacent carbon center for nucleophilic attack or facilitating the stabilization of the transition state during the coupling event. Subsequent insertion of the acrylate or styrene alkene into the palladium-carbon bond forms a new C-C bond, extending the carbon skeleton. The final step involves beta-hydride elimination to restore the conjugated diene system and regenerate the active palladium species, with molecular oxygen from the air serving as the terminal oxidant to close the catalytic loop.
From an impurity control perspective, the choice of the dithiolane protecting group is strategic. It not only stabilizes the reactive enamine-like intermediate but also prevents unwanted polymerization of the nitro-alkene species, which is a common side reaction in similar systems. The high E-selectivity observed in the products suggests that the steric bulk of the catalyst-ligand complex and the thermodynamic stability of the trans-alkene geometry drive the stereochemical outcome. This level of mechanistic understanding allows process chemists to fine-tune reaction parameters, such as temperature and concentration, to suppress minor byproducts. For R&D directors, knowing that the reaction proceeds through a well-defined catalytic cycle provides confidence in the reproducibility of the process when scaling from milligram to kilogram quantities.
How to Synthesize Multi-Substituted Conjugated Dienes Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal results. The protocol is designed to be user-friendly, utilizing standard glassware and commercially available reagents. The key to success lies in maintaining the correct solvent ratio and ensuring adequate oxygen exposure, as the aerobic oxidation step is integral to the catalyst turnover. While the reaction time can vary depending on the specific substrate, a standard duration of approximately 23 hours at 50°C has been proven effective for a broad range of examples. Detailed standard operating procedures regarding workup and purification are essential to isolate the product in high purity, typically involving silica gel chromatography.
- Combine alpha-nitrodithioacetal and acrylate/styrene substrates with 10 mol% Pd(OAc)2 catalyst in a Schlenk flask.
- Add a mixed solvent system of acetic acid and DMSO (4: 1 v/v) and stir under air atmosphere at 50°C for 23 hours.
- Remove volatiles under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target diene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the supply chain for raw materials. Unlike methods requiring exotic or custom-synthesized reagents, this process relies on α-nitrodithioacetals and common acrylates, which are commodity chemicals available from multiple global vendors. This redundancy in sourcing mitigates the risk of supply disruptions and provides leverage in price negotiations. Furthermore, the elimination of stoichiometric oxidants like silver acetate or benzoquinone removes a major cost driver and reduces the burden of hazardous waste disposal, aligning with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the drastic reduction in reagent costs and waste treatment expenses. By utilizing air as the oxidant instead of expensive chemical oxidants, the material cost per kilogram of product is significantly lowered. Additionally, the mild reaction temperature of 50°C reduces energy consumption compared to high-temperature processes, contributing to lower utility costs. The high atom economy and excellent yields minimize the loss of valuable starting materials, ensuring that the maximum amount of input is converted into saleable product. These factors combined result in a highly competitive cost structure for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly into improved supply chain reliability. The tolerance for air and the use of stable solvents mean that the process is less sensitive to minor fluctuations in operational conditions, reducing the likelihood of batch failures. This consistency ensures predictable delivery schedules for downstream customers. Moreover, the simplicity of the workup procedure, which involves standard extraction and chromatography, allows for faster turnaround times between batches. For supply chain planners, this predictability is invaluable for maintaining inventory levels and meeting Just-In-Time delivery commitments without the need for excessive safety stock.
- Scalability and Environmental Compliance: Scaling this chemistry from the bench to commercial production is facilitated by the absence of hazardous reagents and the use of common solvents. The process generates minimal hazardous waste, primarily consisting of aqueous acidic waste and spent silica, which are easier to treat than heavy metal-contaminated streams from other catalytic methods. This environmental profile simplifies the permitting process for new manufacturing lines and reduces long-term liability. The ability to run the reaction under air rather than pure oxygen also enhances process safety, removing the risk of flammability associated with enriched oxygen atmospheres in large-scale reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing workflows. The answers reflect the practical realities of running this reaction, from substrate compatibility to downstream processing capabilities.
Q: What are the optimal reaction conditions for this palladium-catalyzed coupling?
A: The optimal conditions involve using 10 mol% Pd(OAc)2 as the catalyst in a mixed solvent of HOAc and DMSO (4:1) at 50°C under air.
Q: Can this method tolerate diverse functional groups on the alkene substrate?
A: Yes, the method demonstrates broad substrate adaptability, successfully reacting with various acrylates (ethyl, butyl, tert-butyl) and styrenes.
Q: Is the nitro group in the product stable for further derivatization?
A: Yes, the nitro group remains intact during the coupling and can be subsequently reduced to an amine using SnCl2 for further functionalization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-Substituted Conjugated Diene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed cross-dehydrogenative coupling described in CN111018830B. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from discovery to market. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of multi-substituted conjugated diene meets the highest industry standards. Our commitment to quality and technical excellence makes us the ideal partner for developing complex pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By engaging our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments to understand how this efficient synthesis can optimize your supply chain. Let us help you accelerate your development timeline with our reliable manufacturing capabilities and deep technical expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
