Revolutionizing Ticagrelor Production: A Low-Temperature 6-Step Synthetic Strategy for Commercial Scale
The pharmaceutical landscape for antiplatelet agents continues to evolve, driven by the critical need for efficient, scalable, and cost-effective manufacturing processes for life-saving medications like Ticagrelor. As a potent P2Y12 receptor inhibitor, Ticagrelor (CAS: 274693-27-5) represents a cornerstone therapy for acute coronary syndrome, yet its complex molecular architecture, featuring six chiral centers and a fused triazolopyrimidine core, has historically posed significant synthetic challenges. Recent advancements detailed in patent CN115160320A introduce a groundbreaking preparation method that fundamentally reimagines the synthetic trajectory. This novel approach replaces convoluted, multi-step sequences with a streamlined six-step process that leverages mild reaction conditions and readily available starting materials. By shifting away from the traditional reliance on expensive sugar derivatives and harsh thermal conditions, this technology offers a robust pathway for producing high-purity API intermediates, addressing the persistent pain points of yield loss and impurity management that have long plagued the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ticagrelor has been burdened by excessive step counts and inefficient atom economy. Early methodologies, such as those originating from the original developer, often utilized D-ribose as a chiral pool starting material. While effective in establishing stereochemistry, this route necessitates a staggering 17 distinct reaction steps to reach the final target. Such elongated sequences inherently accumulate yield losses at every stage, drastically inflating the cost of goods sold (COGS) and complicating inventory management. Furthermore, many alternative routes reported in literature rely on high-temperature coupling reactions or the use of hazardous reagents like diethyl azodicarboxylate (DEAD) and triphenylphosphine in Mitsunobu reactions. These conditions not only pose safety risks on a kilogram scale but also generate substantial quantities of phosphine oxide waste, creating a severe environmental burden and requiring complex downstream purification to meet stringent regulatory limits on residual impurities.
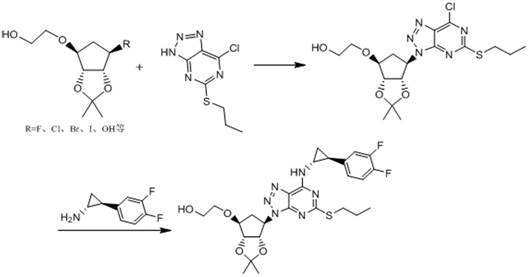
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN115160320A presents a highly convergent strategy that drastically truncates the synthetic timeline. By identifying key disconnection points that allow for the assembly of the core scaffold in just six operational steps, this method eliminates the need for tedious protecting group manipulations common in carbohydrate chemistry. The route strategically employs a condensation reaction between a functionalized cyclopentyl amine and a chlorinated pyrimidine precursor, followed by an intramolecular cyclization to forge the triazole ring. This logical progression avoids the formation of difficult-to-remove regioisomers often seen in direct coupling approaches. Moreover, the entire sequence is designed to proceed under mild thermal conditions, typically ranging from 0°C to 50°C, which preserves the integrity of sensitive functional groups and prevents the thermal degradation that frequently compromises yield in conventional high-temperature protocols.
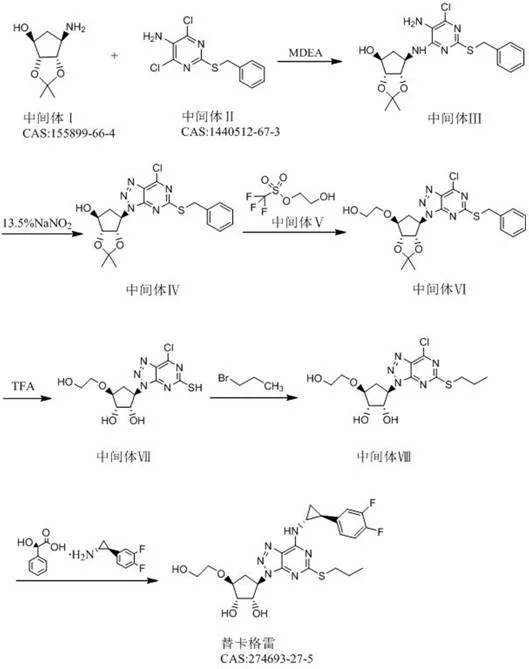
Mechanistic Insights into the 6-Step Convergent Synthesis
The success of this streamlined methodology lies in its precise control over chemoselectivity and stereochemistry during the critical bond-forming events. The process initiates with a nucleophilic aromatic substitution where the amino group of the chiral cyclopentyl intermediate attacks the electron-deficient pyrimidine ring. This step is carefully modulated by the choice of base and solvent, ensuring that only the desired nitrogen atom participates in the coupling, thereby preventing bis-alkylation or hydrolysis of the chloride leaving groups. Following this, the construction of the triazolo[4,5-d]pyrimidine core is achieved through a diazotization-cyclization cascade. Unlike traditional methods that might use unstable diazonium salts, this protocol utilizes controlled nitrosating agents like sodium nitrite or isoamyl nitrite in the presence of acetic acid. This generates the reactive nitroso species in situ, which attacks the ortho-amino group to close the ring with high fidelity, effectively locking in the heterocyclic architecture required for biological activity.
Subsequent steps focus on the installation of the side chains and the final stereochemical refinement. A key innovation is the use of a Grignard-mediated nucleophilic substitution to extend the carbon chain, introducing the hydroxyethyl moiety with precision. This is followed by a gentle acid-mediated deprotection that removes the acetonide protecting groups without epimerizing the sensitive chiral centers on the cyclopentane ring. The final stages involve a thio-alkylation to install the propylthio group and a stereoselective amination using a chiral resolving agent or chiral amine source. This late-stage introduction of chirality ensures that the final product possesses the correct (1S, 2S, 3R, 5S) configuration essential for P2Y12 inhibition. By deferring the most complex stereochemical adjustments to the end of the synthesis, the route maximizes the efficiency of earlier steps and simplifies the purification of intermediates, resulting in a final product with exceptional optical purity.
How to Synthesize Ticagrelor Efficiently
The implementation of this six-step protocol requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to maximize the benefits of the novel pathway. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates and reducing solvent consumption. For R&D teams looking to adopt this methodology, the key lies in the optimization of the cyclization and deprotection steps, which serve as the pivot points for the entire synthesis. Detailed standard operating procedures regarding reagent addition rates and quenching protocols are essential to maintain the high yields reported in the patent examples. The following guide outlines the critical operational phases derived from the intellectual property, providing a roadmap for laboratory and pilot-scale execution.
- Condense Intermediate I (aminocyclopentene derivative) with Intermediate II (dichloro-pyrimidinamine) under alkaline conditions at 0-30°C to form Intermediate III.
- Perform cyclization on Intermediate III using a nitrosating agent (e.g., sodium nitrite or isoamyl nitrite) to construct the triazole ring, yielding Intermediate IV.
- Execute nucleophilic substitution using a Grignard reagent and Intermediate V (hydroxyethyl triflate) to extend the carbon chain, followed by acid deprotection to reveal the diol system.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthetic route offers profound implications for cost structure and supply security. The shift from a 17-step carbohydrate-based synthesis to a 6-step convergent route fundamentally alters the economic model of Ticagrelor production. By drastically reducing the number of unit operations, manufacturers can achieve significant reductions in labor costs, equipment occupancy time, and utility consumption. The elimination of high-temperature steps further contributes to energy savings, aligning production with modern sustainability goals. Additionally, the use of commodity chemicals as starting materials, rather than specialized chiral sugars, mitigates the risk of raw material shortages and price volatility, ensuring a more stable and predictable supply chain for downstream formulation partners.
- Cost Reduction in Manufacturing: The primary driver of cost efficiency in this process is the dramatic reduction in step count. Each chemical transformation in a synthetic route incurs costs related to reagents, solvents, workup materials, and waste disposal. By cutting the number of steps by more than half compared to traditional routes, the cumulative yield loss is minimized, and the overall material throughput is optimized. Furthermore, the avoidance of expensive transition metal catalysts, which often require specialized scavenging resins to meet residual metal specifications, removes a significant line item from the production budget. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for exotic high-pressure or high-temperature equipment, lowering capital expenditure requirements.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on readily available bulk chemicals. Traditional routes dependent on specific chiral pool materials can be vulnerable to agricultural fluctuations or limited supplier bases. In contrast, the intermediates used in this novel method, such as the dichloro-pyrimidine derivatives and simple cyclopentyl amines, are produced by a wide network of fine chemical manufacturers. This diversification of the supplier base reduces the risk of single-source bottlenecks. Moreover, the simplified purification profile means that production campaigns can be completed faster, reducing lead times and allowing for more responsive inventory management to meet market demand spikes.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but this route is inherently designed for robustness. The absence of hazardous reagents like azodicarboxylates and the reduction of heavy metal usage simplify the environmental, health, and safety (EHS) profile of the manufacturing site. Waste streams are less toxic and easier to treat, facilitating compliance with increasingly stringent environmental regulations. The high selectivity of the reactions minimizes the formation of complex byproduct mixtures, which reduces the volume of organic solvents required for chromatographic purification. This 'green chemistry' aspect not only lowers disposal costs but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Ticagrelor synthesis. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, offering clarity on how this method outperforms legacy technologies in terms of purity, yield, and operational simplicity. Understanding these nuances is critical for technical decision-makers evaluating potential technology transfers or licensing opportunities.
Q: How does the new 6-step route compare to traditional D-ribose based synthesis?
A: Traditional routes often rely on D-ribose and require up to 17 steps with complex protection/deprotection sequences. The novel method described in CN115160320A shortens this to just 6 steps, utilizing cheaper starting materials and avoiding harsh high-temperature conditions, significantly simplifying purification.
Q: What are the key advantages regarding reaction conditions and impurity control?
A: The process operates primarily at low temperatures (0-30°C) rather than the high temperatures required by prior art. This mild environment minimizes side reactions and byproduct formation, ensuring higher crude purity and reducing the need for extensive column chromatography.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for scalability. It avoids expensive transition metal catalysts that require difficult removal steps and uses readily available raw materials. The simplified workflow and milder conditions reduce energy consumption and solid waste generation, making it ideal for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Supplier
The technological potential of this six-step synthesis represents a significant leap forward in the manufacturing of antiplatelet therapeutics, offering a blueprint for efficiency that few competitors can match. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Ticagrelor intermediate meets the exacting standards required for global regulatory submissions. We understand that consistency is key in pharmaceutical supply, and our process engineering team is dedicated to maintaining the tight control parameters necessary for this sensitive chemistry.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized route can enhance your supply chain resilience. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than projections. Let us partner with you to deliver high-quality Ticagrelor intermediates that drive your commercial success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
