Advanced Enzymatic Reduction for High-Purity Chiral Alcohol Commercialization
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards greener, more efficient synthetic methodologies, a transition exemplified by the technological breakthroughs detailed in patent CN106497996B. This specific intellectual property discloses a highly sophisticated enzymatic preparation method for chiral alcohols, utilizing a specialized ketoreductase to achieve asymmetric reduction of prochiral ketone compounds with exceptional precision. Unlike traditional chemical synthesis routes that often rely on harsh conditions and toxic reagents, this biocatalytic approach operates under mild physiological conditions, ensuring that the structural integrity of sensitive functional groups is maintained throughout the transformation. The core innovation lies in the specific selection of ketoreductase enzymes that demonstrate superior stereoselectivity, capable of producing chiral alcohol products with an enantiomeric excess (ee) value exceeding 99% and a chemical purity greater than 95.0%. For R&D directors and technical decision-makers, this represents a critical advancement in process chemistry, offering a robust pathway to synthesize complex chiral building blocks that are essential for the development of next-generation active pharmaceutical ingredients (APIs) and high-value agrochemical intermediates without the baggage of heavy metal contamination.
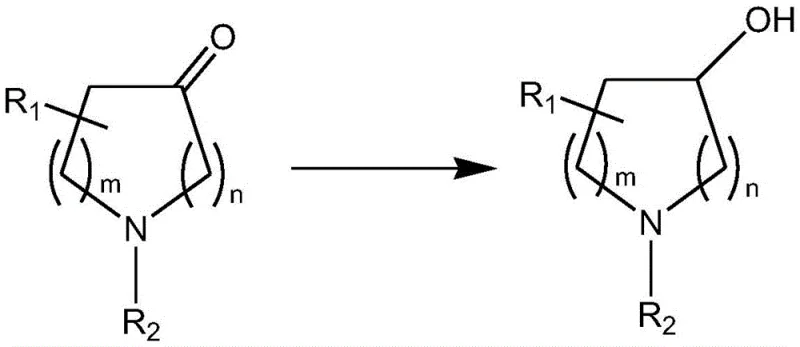
Historically, the manufacturing of optically active chiral alcohols has been plagued by significant technical and economic bottlenecks that hindered efficient commercial scale-up of complex chiral alcohols. The conventional resolution methods, which rely on chiral resolving agents such as tartaric acid, are theoretically limited to a maximum yield of 50% and often suffer from actual resolution yields of less than 10% in practical applications, leading to massive material waste and inflated production costs. Furthermore, the chemical asymmetric reduction methods utilizing chiral metal complexes, while capable of higher yields, necessitate the use of expensive transition metal catalysts like ruthenium or rhodium, which require high-pressure hydrogenation equipment and pose severe risks of heavy metal residue in the final product. These residual metals are notoriously difficult to remove to the stringent parts-per-million levels required by global regulatory agencies, often necessitating additional, costly purification steps that erode profit margins. In stark contrast, the novel enzymatic approach described in the patent data circumvents these limitations by employing a biological catalyst that functions at ambient pressure and moderate temperatures, thereby eliminating the need for specialized high-pressure reactors and removing the risk of toxic metal contamination entirely from the manufacturing workflow.
Mechanistic Insights into Ketoreductase-Catalyzed Asymmetric Reduction
The mechanistic elegance of this biocatalytic system is rooted in the precise hydride transfer facilitated by the ketoreductase enzyme, which selectively reduces the carbonyl group of the prochiral ketone to a hydroxyl group with absolute stereocontrol. The enzyme active site creates a chiral environment that discriminates between the two enantiotopic faces of the ketone substrate, ensuring that the hydride from the reduced cofactor (NADH or NADPH) is delivered exclusively to one face, thus generating a single enantiomer of the chiral alcohol with an ee value that can reach as high as 99.5%. This high level of stereoselectivity is not merely a laboratory curiosity but a critical commercial asset, as it drastically simplifies the downstream purification process by minimizing the formation of the unwanted enantiomeric impurity, which is often the most difficult contaminant to separate via standard chromatography or crystallization techniques. Moreover, the patent specifies that the ketoreductase can be used in conjunction with a coenzyme regeneration system, which is vital for maintaining the catalytic turnover number and ensuring the economic viability of the process on an industrial scale.
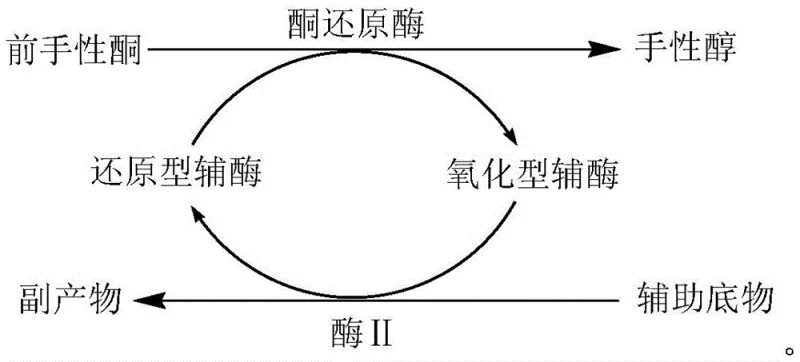
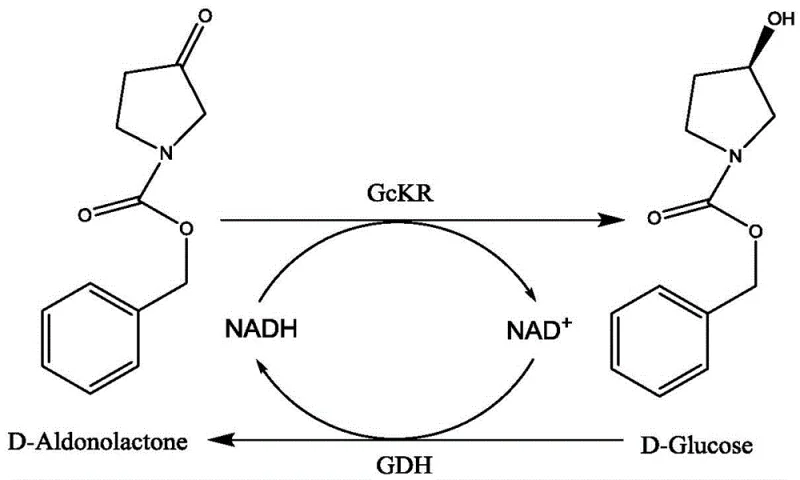
A critical component of this mechanism is the in situ regeneration of the oxidized coenzyme, which prevents the accumulation of inhibitory byproducts and sustains the reaction drive towards completion. The patent details the use of a coupled enzyme system, such as glucose dehydrogenase (GDH) with glucose, or a substrate coupling method using isopropanol, to continuously convert the oxidized NAD+ back into the active reduced NADH form required for the reduction step. This regeneration loop is thermodynamically favorable and kinetically efficient, allowing for the use of catalytic rather than stoichiometric amounts of the expensive nicotinamide cofactors, which significantly lowers the raw material cost per kilogram of product. From an impurity control perspective, the mild aqueous buffer conditions (pH 6.0-8.0) prevent side reactions such as racemization or dehydration that are common in acidic or basic chemical reduction environments, thereby ensuring a cleaner impurity profile and higher overall yield of the target high-purity chiral alcohol. The specific examples in the patent, such as the reduction of N-benzyloxycarbonyl-3-pyrrolidone, demonstrate yields exceeding 80% with purity above 95%, validating the robustness of this mechanistic design for practical application.
How to Synthesize Chiral Alcohol Efficiently
The implementation of this enzymatic route requires careful optimization of reaction parameters to maximize space-time yield and enzyme stability, and the patent provides a clear framework for executing this synthesis in a GMP-compliant environment. The detailed standardized synthesis steps involve dispersing the prochiral ketone substrate in a phosphate buffer, adding the specific ketoreductase and coenzyme regeneration components, and maintaining the system at a controlled temperature between 25°C and 35°C for a defined period to ensure complete conversion. While the specific operational details are critical for reproducibility, the general protocol emphasizes the simplicity of the workup, which typically involves filtration to remove the biocatalyst followed by organic solvent extraction, avoiding the complex quenching procedures associated with reactive metal hydrides.
- Prepare the reaction system by mixing the prochiral ketone compound with a specific ketoreductase and coenzyme in a buffered solution.
- Maintain the pH between 6.0 and 8.0 and control the temperature between 20°C and 50°C to ensure optimal enzyme activity.
- Implement a coenzyme regeneration system using glucose dehydrogenase or isopropanol to sustain the catalytic cycle and isolate the product via extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology offers profound strategic advantages that extend beyond simple technical metrics, fundamentally altering the cost structure and risk profile of the supply chain. The elimination of heavy metal catalysts and high-pressure hydrogenation equipment translates directly into a significantly reduced capital expenditure (CAPEX) for manufacturing facilities, as standard glass-lined or stainless steel reactors can be utilized without the need for specialized pressure-rated vessels or extensive safety systems for handling explosive gases. Furthermore, the removal of toxic metals from the process flow drastically simplifies the environmental compliance landscape, reducing the burden of hazardous waste disposal and minimizing the risk of regulatory delays associated with heavy metal residue testing in the final API. This green chemistry approach aligns perfectly with the increasing global demand for sustainable manufacturing practices, potentially unlocking premium market segments and improving the corporate sustainability profile of the end-product.
- Cost Reduction in Manufacturing: The economic model of this enzymatic process is superior to traditional methods primarily due to the drastic simplification of the purification train and the efficient use of cofactors. By avoiding the use of expensive chiral resolving agents that are consumed in stoichiometric quantities and discarded, and by eliminating the need for costly transition metal catalysts, the direct material cost is substantially lowered. Additionally, the high stereoselectivity means that less material is lost to the wrong enantiomer, effectively doubling the theoretical yield compared to resolution methods and maximizing the return on raw material investment. The mild reaction conditions also result in lower energy consumption for heating and cooling, contributing to a leaner operational expenditure profile that enhances overall margin potential for high-volume production.
- Enhanced Supply Chain Reliability: The reliance on biocatalysts and common organic substrates creates a more resilient supply chain that is less susceptible to the geopolitical and logistical volatility often associated with rare earth metals and specialized chemical reagents. Enzymes can be produced via fermentation using renewable feedstocks, ensuring a stable and scalable source of the critical catalyst that is not subject to the same mining constraints as precious metals. The robustness of the reaction conditions, which tolerate a wide range of pH and temperature without catastrophic failure, ensures consistent batch-to-batch quality and reduces the risk of production stoppages due to process deviations. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical customers who require strict adherence to delivery schedules and quality specifications.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is inherently safer and more straightforward due to the absence of exothermic risks associated with chemical hydride reductions and the lack of high-pressure hazards. The aqueous nature of the reaction medium reduces the volume of organic solvents required, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing site. Waste streams are primarily biological and organic in nature, which are easier to treat and dispose of compared to heavy metal-containing sludge, facilitating smoother environmental permitting and ongoing regulatory compliance. This scalability ensures that the technology can meet the demands of large-scale commercial production without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic chiral alcohol synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-making processes. Understanding these nuances is essential for assessing the feasibility of integrating this biocatalytic route into existing manufacturing portfolios.
Q: What are the stereoselectivity advantages of this enzymatic method?
A: The patented ketoreductase achieves an ee value higher than 99%, significantly outperforming traditional chemical resolution methods which often struggle to exceed 95% purity without multiple recrystallization steps.
Q: How does the coenzyme regeneration system impact cost?
A: By utilizing a glucose dehydrogenase coupled system or isopropanol coupling, the expensive NADH/NADPH cofactors are recycled in situ, eliminating the need for stoichiometric amounts of reduced cofactors and drastically lowering raw material costs.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction operates under mild conditions (20-50°C) and atmospheric pressure without heavy metal catalysts, making it inherently safer and easier to scale from laboratory to multi-ton commercial production compared to high-pressure hydrogenation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in patent CN106497996B and have positioned ourselves as a leader in translating such advanced laboratory technologies into robust commercial realities. Our CDMO capabilities are specifically engineered to handle the complexities of biocatalytic processes, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining the highest standards of quality and safety. We understand that the transition from bench-scale enzymatic reactions to multi-ton manufacturing requires precise control over fermentation, enzyme immobilization, and downstream processing, and our state-of-the-art facilities are equipped with the rigorous QC labs and stringent purity specifications necessary to deliver high-purity chiral alcohol intermediates that meet global regulatory requirements. Our team of expert chemists and engineers works collaboratively with clients to optimize reaction parameters, ensuring that the theoretical benefits of high ee values and yields are fully realized in the final commercial product.
We invite pharmaceutical and fine chemical companies to leverage our expertise to optimize their supply chains and reduce their manufacturing costs through the adoption of this superior enzymatic technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the specific economic benefits of switching from your current chemical synthesis route to this biocatalytic alternative, taking into account your unique volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and comprehensive route feasibility assessments that will demonstrate the viability of this process for your specific target molecules. Let us help you secure a sustainable, cost-effective, and high-quality supply of critical chiral intermediates for your future projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
