Advanced One-Pot Conversion of Paclitaxel to High-Purity Docetaxel Intermediates
Introduction to Next-Generation Taxane Conversion Technologies
The pharmaceutical industry continuously seeks more efficient pathways for producing high-value anticancer agents, and the semi-synthetic conversion of paclitaxel to docetaxel represents a pivotal advancement in this domain. Patent CN101048394A discloses a revolutionary methodology that streamlines the production of docetaxel intermediates by utilizing paclitaxel or paclitaxel-containing materials as the primary starting feedstock. This approach fundamentally shifts the paradigm from traditional total synthesis or 10-DAB coupling methods towards a more direct modification of the naturally abundant paclitaxel scaffold. By implementing a sophisticated one-pot reaction sequence, this technology enables the simultaneous protection of critical hydroxyl groups and the introduction of the essential tert-butoxycarbonyl (t-Boc) functionality at the C-3' position. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenges of step economy and impurity control in taxane manufacturing.
Furthermore, the versatility of this synthetic route extends beyond pure paclitaxel, accommodating crude extracts and waste streams that are often discarded in conventional processing facilities. This capability transforms potential environmental liabilities into valuable chemical assets, aligning perfectly with modern green chemistry principles and sustainable manufacturing goals. The technical depth of this invention lies in its ability to manage the complex stereochemistry and reactivity of the taxane core while minimizing harsh reaction conditions that typically degrade these sensitive molecules. As we delve deeper into the mechanistic details and commercial implications, it becomes evident that this technology provides a robust framework for scaling up the production of high-purity docetaxel intermediates to meet global demand.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional semi-synthetic routes for docetaxel production have historically relied heavily on 10-deacetylbaccatin III (10-DAB) as the starting material, necessitating a complex coupling reaction with a chiral beta-lactam side chain. This conventional approach presents significant logistical and chemical hurdles, including the requirement for extremely low temperatures, often below -40°C, to maintain stereochemical integrity during the coupling step. Additionally, the use of expensive chiral auxiliaries and the generation of difficult-to-remove diastereomeric impurities create substantial bottlenecks in purification workflows. These factors collectively contribute to high production costs and extended lead times, making the supply chain vulnerable to fluctuations in the availability of high-purity 10-DAB. Moreover, the multi-step nature of the traditional synthesis involves numerous isolation and purification stages, each contributing to cumulative yield losses and increased solvent waste generation.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the inherent structural similarity between paclitaxel and docetaxel to bypass the need for side-chain construction entirely. Instead of building the molecule up from a smaller core, this method strategically modifies the existing C-3' N-benzoyl phenylisoserine side chain of paclitaxel directly. The core innovation is a one-pot reaction sequence where hydroxyl groups at the C-2', C-7, and C-10 positions are protected, followed immediately by the introduction of the t-Boc group on the amide nitrogen without isolating the intermediate. 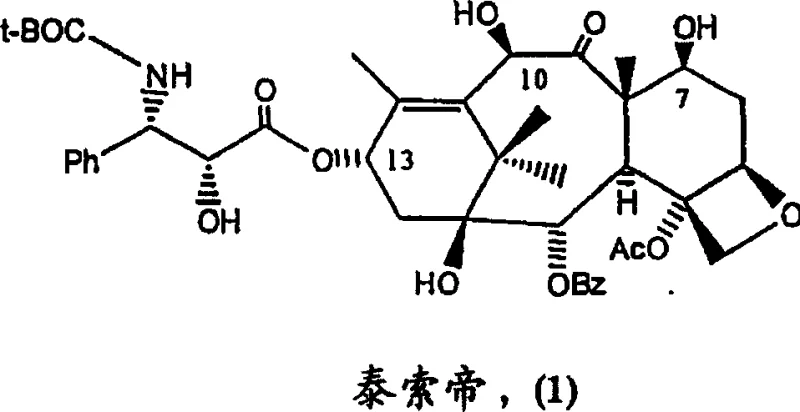 This consolidation of steps drastically reduces the operational complexity and solvent usage associated with the synthesis. By utilizing paclitaxel, which is often more readily available from natural sources or as a byproduct of other extraction processes, manufacturers can secure a more stable and cost-effective raw material supply. The ability to process mixed taxane streams further enhances the economic viability of this route, allowing for the valorization of materials that would otherwise be considered waste in standard paclitaxel purification protocols.
This consolidation of steps drastically reduces the operational complexity and solvent usage associated with the synthesis. By utilizing paclitaxel, which is often more readily available from natural sources or as a byproduct of other extraction processes, manufacturers can secure a more stable and cost-effective raw material supply. The ability to process mixed taxane streams further enhances the economic viability of this route, allowing for the valorization of materials that would otherwise be considered waste in standard paclitaxel purification protocols.
Mechanistic Insights into One-Pot Protection and N-Acylation
The chemical elegance of this process is rooted in the precise control of reactivity across multiple functional groups within the taxane skeleton. The initial phase involves the selective protection of hydroxyl groups, where the inherent difference in nucleophilicity between the C-2', C-7, and C-10 positions is exploited. Typically, the C-2' hydroxyl is the most reactive, followed by C-7 and then C-10, allowing for sequential or simultaneous protection strategies depending on the specific reagents employed. Common protecting agents include silylating agents like triethylsilyl chloride or acylating agents such as di-tert-butyl dicarbonate (Boc2O), which serve a dual purpose in this specific protocol. The reaction is conducted in the presence of a base, such as DMAP or lithium tert-butoxide, which facilitates the deprotonation of the hydroxyl groups and drives the formation of the protected esters or ethers under mild conditions ranging from low temperature to ambient room temperature.
Following the protection phase, the reaction mixture proceeds directly to the N-acylation step without any workup or isolation, which is critical for maintaining high throughput and minimizing material loss. A t-Boc agent, frequently the same reagent used for hydroxyl protection like Boc2O, reacts with the amide nitrogen at the C-3' position to form a urea linkage. 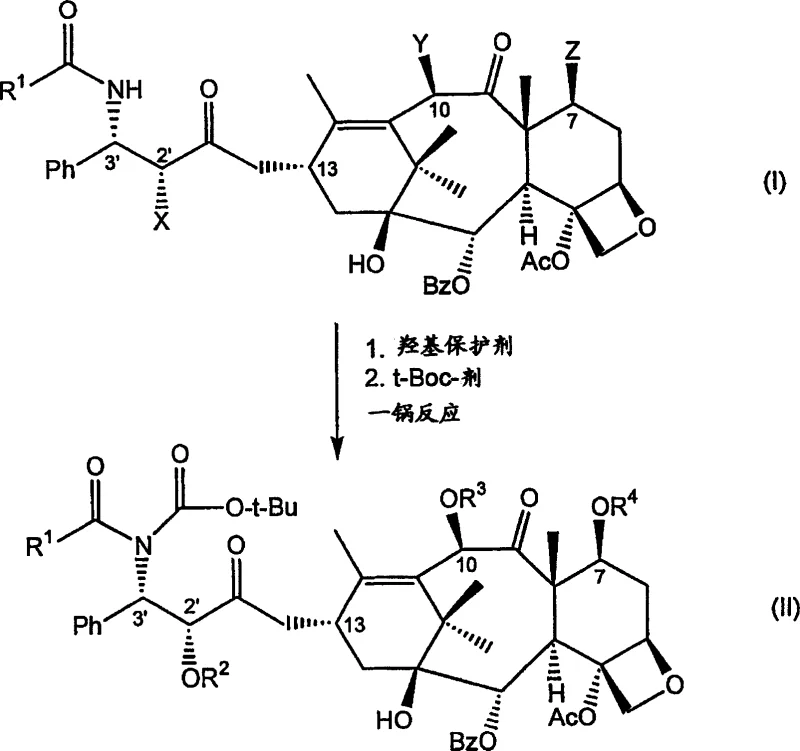 This transformation is pivotal as it installs the key structural motif that distinguishes docetaxel from paclitaxel. The subsequent N-deacylation step involves the selective removal of the benzoyl group from the urea intermediate using a strong base, such as lithium hydroxide, potentially in the presence of peroxides to enhance selectivity. This sequence effectively converts the paclitaxel side chain into the docetaxel side chain while the core remains safeguarded by the protecting groups. The final deprotection yields the target docetaxel molecule with high fidelity, demonstrating a sophisticated understanding of orthogonal protection strategies tailored for complex natural product derivatives.
This transformation is pivotal as it installs the key structural motif that distinguishes docetaxel from paclitaxel. The subsequent N-deacylation step involves the selective removal of the benzoyl group from the urea intermediate using a strong base, such as lithium hydroxide, potentially in the presence of peroxides to enhance selectivity. This sequence effectively converts the paclitaxel side chain into the docetaxel side chain while the core remains safeguarded by the protecting groups. The final deprotection yields the target docetaxel molecule with high fidelity, demonstrating a sophisticated understanding of orthogonal protection strategies tailored for complex natural product derivatives.
How to Synthesize Protected Docetaxel Intermediates Efficiently
Implementing this synthesis requires careful attention to reaction conditions and reagent stoichiometry to ensure optimal conversion and purity. The process begins by dissolving the paclitaxel starting material, which can range from pure API grade to crude extracts, in a suitable anhydrous organic solvent such as tetrahydrofuran (THF) or dichloromethane (DCM) under an inert argon atmosphere. To this solution, a catalytic or stoichiometric amount of base is added to activate the hydroxyl groups, followed by the slow addition of the protecting agent. The reaction mixture is stirred until thin-layer chromatography (TLC) confirms the complete consumption of the starting material, indicating successful protection of the C-2', C-7, and C-10 positions. Without isolating this intermediate, the t-Boc agent is introduced to the same vessel to effect the N-acylation, creating the protected docetaxel precursor in a seamless continuous flow of chemical transformations.
- Dissolve paclitaxel or taxane-containing mixtures in an organic solvent such as THF or DCM under inert atmosphere.
- Add a base and hydroxyl protecting agent to selectively protect C-2', C-7, and C-10 positions without isolating intermediates.
- Introduce a t-Boc agent in the same reaction vessel to acylate the C-3' amide nitrogen, forming the protected urea-linked intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this semi-synthetic pathway offers transformative benefits regarding cost structure and material security. The primary advantage lies in the flexibility of raw material sourcing; by enabling the use of crude paclitaxel extracts or spent mother liquors from existing purification lines, companies can significantly reduce their dependency on expensive, highly purified 10-DAB. This shift not only lowers the direct cost of goods sold but also mitigates the risk of supply disruptions associated with single-source natural product intermediates. Furthermore, the one-pot nature of the reaction drastically simplifies the manufacturing infrastructure required, as fewer reactors and less downstream processing equipment are needed to achieve the same output volume compared to multi-step coupling syntheses.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps translates directly into substantial savings in solvent consumption, energy usage, and labor hours. By avoiding the need for cryogenic conditions often required in beta-lactam coupling, the process operates under more energy-efficient thermal profiles, reducing the utility burden on the production facility. Additionally, the higher atom economy of modifying an existing side chain versus constructing a new one minimizes the purchase of costly chiral reagents and coupling agents. These cumulative efficiencies result in a leaner cost base that enhances competitiveness in the generic oncology market without compromising on the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Diversifying the feedstock to include various taxane-containing materials, such as waste streams from paclitaxel production, creates a more resilient supply chain. This approach allows manufacturers to capitalize on available inventory that might otherwise be discarded, effectively turning a waste management cost into a revenue-generating opportunity. The robustness of the chemistry against impurities found in crude mixes means that upstream purification requirements can be relaxed, shortening the overall lead time from biomass to finished intermediate. This flexibility ensures a more consistent flow of materials to the formulation stage, safeguarding against the volatility of agricultural harvests or extraction yields.
- Scalability and Environmental Compliance: The streamlined process design is inherently more scalable, as the reduction in unit operations simplifies technology transfer from pilot plant to commercial scale. Fewer solvent exchanges and workup steps mean a reduced environmental footprint, aligning with increasingly stringent regulatory standards for pharmaceutical manufacturing emissions and waste disposal. The ability to process larger batches in a single reactor vessel improves throughput capacity, allowing suppliers to respond more agilely to surges in market demand. This scalability, combined with the greener profile of the synthesis, positions the technology as a sustainable long-term solution for high-volume docetaxel production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented semi-synthetic route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers are derived directly from the technical specifications and experimental data provided within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of using paclitaxel over 10-DAB for docetaxel synthesis?
A: Using paclitaxel allows manufacturers to utilize abundant natural sources or waste streams from paclitaxel purification, bypassing the need for complex side-chain coupling reactions required when starting from 10-deacetylbaccatin III.
Q: How does the one-pot protection strategy improve process efficiency?
A: The one-pot strategy eliminates the isolation and purification of intermediate protected taxanes, significantly reducing solvent consumption, processing time, and overall material loss during the synthesis sequence.
Q: Can this method handle crude taxane mixtures?
A: Yes, the patented method is robust enough to process crude extracts or spent mother liquors containing various taxanes, converting them simultaneously into valuable docetaxel intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Docetaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the semi-synthetic conversion of paclitaxel to docetaxel and possesses the technical capability to implement these one-pot protection and N-acylation protocols at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every stage of the synthesis to guarantee the highest standards of safety and efficacy for the final API.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific supply chain needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this paclitaxel-based feedstock strategy. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our intermediates against your internal benchmarks. Together, we can optimize your docetaxel supply chain, reducing costs and enhancing reliability while adhering to the highest standards of pharmaceutical manufacturing excellence.
