Revolutionizing Taxane Production: Advanced Semi-Synthetic Routes for Commercial Scale-Up
The global demand for potent anti-cancer agents continues to surge, placing immense pressure on the supply chains of critical oncology therapeutics. Among these, taxane derivatives such as Paclitaxel and Docetaxel remain cornerstone treatments for various solid tumors, yet their complex molecular architectures have historically posed significant challenges for consistent, large-scale manufacturing. The patent CN101663287B introduces a transformative semi-synthetic process that addresses these bottlenecks by optimizing reaction pathways and enhancing stereochemical control. This innovation represents a pivotal shift from extraction-dependent models to robust chemical synthesis, offering a reliable taxane intermediate supplier pathway for the global pharmaceutical industry. By leveraging advanced protection strategies and single-isomer side chain coupling, this technology not only improves yield but also drastically simplifies the purification landscape, ensuring that high-purity pharmaceutical intermediates can be delivered with greater consistency and economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
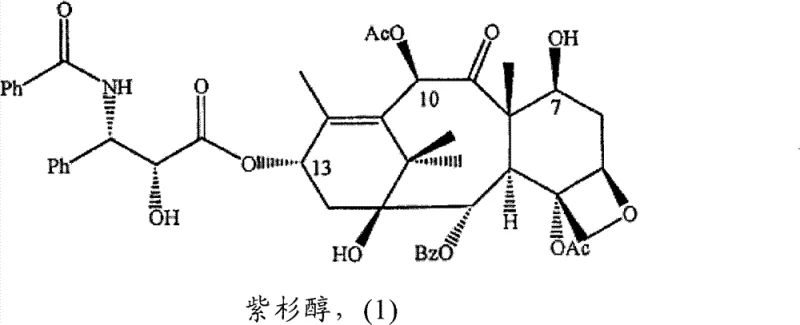
Historically, the semi-synthesis of taxanes has been plagued by inefficient reaction schemes that rely heavily on the availability of specific natural precursors and suffer from poor stereochemical outcomes. Traditional methodologies, such as those disclosed in earlier patents like US 4,924,011, often involve lengthy reaction sequences that require multiple protection and deprotection steps, increasing both the time and cost of production. A critical drawback of these legacy processes is the generation of epimeric mixtures at the C-13 side chain attachment point, typically resulting in a ratio of 60:40 between the desired active isomer and its inactive counterpart. This lack of selectivity necessitates complex and costly chromatographic separations to isolate the therapeutically active component, leading to substantial material loss and an overall yield that rarely exceeds 40%. Furthermore, the reliance on variable natural extracts introduces batch-to-batch inconsistency, complicating regulatory compliance and quality control for procurement teams seeking stable long-term contracts.
The Novel Approach
In stark contrast, the improved process detailed in CN101663287B revolutionizes the synthesis landscape by introducing a streamlined route that prioritizes stereochemical purity and operational simplicity. The core innovation lies in the utilization of a single isomer of the protected phenylisoserine side chain precursor, which ensures that the coupling reaction at the C-13 position proceeds with high specificity. This strategic modification eliminates the formation of unwanted epimers at the source, thereby bypassing the need for arduous separation processes that traditionally erode profit margins. Additionally, the method employs a unified protection strategy where identical hydroxy-protecting groups are utilized for both the C-7 position of the taxane core and the 2'-position of the side chain. This symmetry simplifies the reaction monitoring and allows for a simultaneous deprotection step, significantly shortening the overall production timeline. The result is a robust manufacturing protocol that consistently delivers yields surpassing 65%, providing a compelling case for cost reduction in API manufacturing through enhanced material throughput.
Mechanistic Insights into C-7 Protection and C-13 Esterification
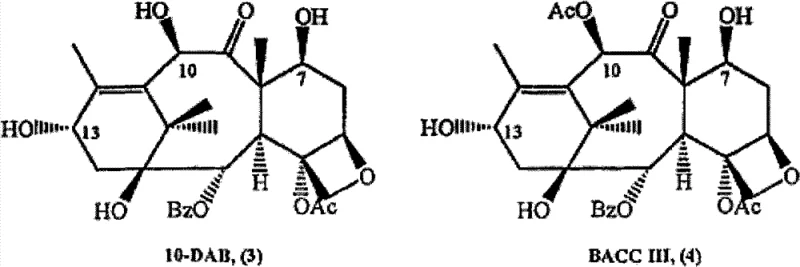
The mechanistic elegance of this semi-synthetic route is rooted in the precise manipulation of hydroxyl reactivity within the highly functionalized taxane core. The process initiates with the selective protection of the free hydroxyl group at the C-7 position of the starting material, such as 9-dihydro-13-acetyl Baccatine III (9-DHB) or 10-deacetylation baccatin III. This protection step is critical because the C-7 hydroxyl is chemically similar to the C-13 hydroxyl, and without masking, it would compete during the subsequent esterification, leading to regioisomeric impurities that are notoriously difficult to remove. By employing protecting groups such as tert-butoxycarbonyl (t-BOC) or silyl ethers, the synthesis ensures that the coupling reagent reacts exclusively at the C-13 position. This regioselectivity is further enhanced by the use of specific condensing agents like DCC or DPC in the presence of catalytic DMAP, which activates the carboxylic acid of the side chain precursor for nucleophilic attack by the C-13 alcohol. The outcome is a clean formation of the ester bond with minimal byproduct formation, setting the stage for a high-purity final product.
Following the coupling reaction, the integrity of the stereochemistry is maintained through the use of optically active or chiral single isomers of the phenylisoserine side chain. In conventional syntheses, the use of racemic mixtures inevitably leads to a 1:1 mixture of diastereomers, requiring resolution. However, by introducing a pre-configured single isomer, the reaction trajectory is locked into the formation of the biologically active configuration. The subsequent deprotection step is equally sophisticated, utilizing mild acidic conditions, such as formic acid at temperatures between 30°C to 50°C, to simultaneously cleave the protecting groups at C-7 and the 2'-position of the side chain. This concurrent deprotection not only reduces the number of unit operations but also minimizes the exposure of the sensitive taxane core to harsh conditions that could induce degradation or rearrangement. The mechanistic control exerted at every stage ensures that the impurity profile remains exceptionally low, meeting the stringent purity specifications required for clinical-grade Active Pharmaceutical Ingredients.
How to Synthesize Protected Taxane Intermediates Efficiently
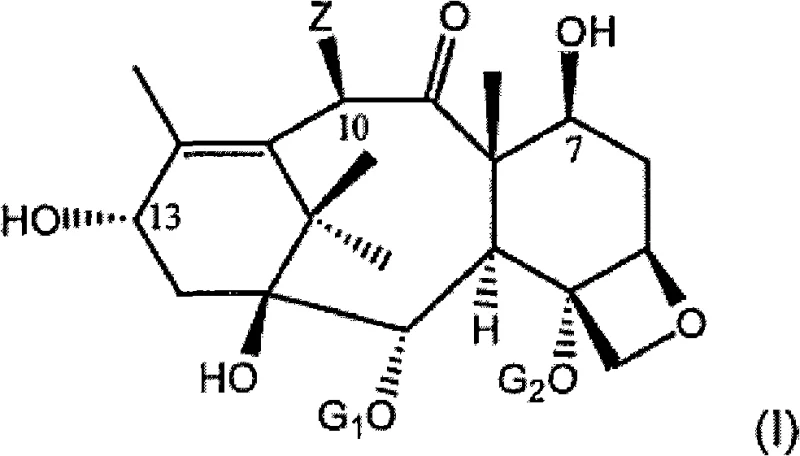
The practical implementation of this synthesis requires careful attention to reaction conditions and reagent quality to maximize the benefits of the patented methodology. The process begins with the dissolution of the taxane precursor in an anhydrous organic solvent such as tetrahydrofuran (THF) or toluene, followed by the addition of the protecting agent under an inert atmosphere to prevent moisture interference. Once the C-7 protection is confirmed via TLC, the activated side chain precursor is introduced, and the mixture is heated to facilitate the esterification. The reaction progress is closely monitored to ensure complete conversion before proceeding to the workup phase, which involves standard aqueous washes and drying procedures. The resulting protected intermediate can be purified via crystallization or column chromatography, although the high selectivity of the reaction often renders extensive purification unnecessary. For the final conversion to the active drug substance, the protected intermediate is treated with acid to remove the blocking groups, yielding the target taxane derivative in high purity. Detailed standardized synthetic steps see the guide below.
- Protect the free hydroxyl group at the C-7 position of the taxane precursor using a suitable hydroxy-protecting agent.
- Perform direct esterification at the C-13 position using a single isomer of protected phenylisoserine side chain precursor.
- Execute deprotection under acidic conditions to yield the final taxane derivative such as Paclitaxel or Docetaxel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced semi-synthetic process offers profound strategic advantages that extend beyond mere technical metrics. The primary value proposition lies in the significant optimization of the cost structure associated with taxane production. By eliminating the need for complex separation of epimeric mixtures, the process removes a major cost center related to preparative chromatography and solvent consumption. Furthermore, the increase in overall yield from approximately 40% to over 65% means that less raw material is required to produce the same amount of finished goods, directly translating to substantial cost savings in raw material procurement. This efficiency gain is compounded by the simplified workflow, which reduces labor hours and equipment occupancy time, allowing for higher throughput within existing manufacturing facilities. These factors combine to create a more resilient economic model that can better withstand fluctuations in the pricing of natural precursors.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of purification steps significantly lower the operational expenditure per kilogram of product. The ability to use identical protecting groups for multiple positions streamlines the reagent inventory, reducing the complexity and cost of chemical sourcing. Additionally, the higher yield ensures that the fixed costs of production are amortized over a larger volume of saleable product, enhancing the overall margin profile. This economic efficiency makes the final API more competitive in the global market, allowing for more aggressive pricing strategies without compromising quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route mitigates the risks associated with the variability of natural plant extracts. By relying on a defined chemical process rather than biological extraction alone, manufacturers can guarantee consistent batch quality and availability. The use of stable, commercially available precursors like 9-DHB ensures that the supply chain is not bottlenecked by the seasonal or geographical limitations of yew tree harvesting. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplified reaction sequence facilitates easier scale-up from laboratory to commercial production, as there are fewer unit operations that could introduce variability or failure points. The reduction in solvent usage and waste generation aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. The process avoids the use of hazardous heavy metals, simplifying the waste treatment process and reducing the environmental footprint of the manufacturing site. This compliance advantage is increasingly important for multinational corporations seeking to partner with suppliers who demonstrate a commitment to sustainability and regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the specific advantages in terms of yield, purity, and operational simplicity that distinguish this method from legacy technologies.
Q: How does this semi-synthetic process improve yield compared to conventional methods?
A: Conventional methods often produce a mixture of epimers with yields around 40%. This patented process utilizes a single isomer of the phenylisoserine side chain, achieving yields exceeding 65% and eliminating the need for difficult separation of stereoisomers.
Q: What are the primary precursors used in this synthesis route?
A: The process is designed to utilize readily available natural precursors such as 10-deacetylation baccatin III (10-DAB), Baccatine III, and 9-dihydro-13-acetyl Baccatine III (9-DHB), ensuring a robust and sustainable supply chain foundation.
Q: Why is the protection of the C-7 hydroxyl group critical in this mechanism?
A: Protecting the C-7 hydroxyl group prevents unwanted side reactions during the esterification at the C-13 position. This selectivity is crucial for maintaining high purity and simplifying the downstream purification process, which is vital for GMP compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs depends on a partnership grounded in technical excellence and supply chain integrity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering high-purity Paclitaxel and related taxane derivatives that meet the most rigorous international standards. Our state-of-the-art rigorous QC labs and stringent purity specifications guarantee that every batch released conforms to the exacting requirements of global regulatory bodies, providing our partners with the confidence needed to advance their clinical and commercial programs.
We invite you to collaborate with us to leverage this advanced semi-synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our optimized processes can enhance your supply chain resilience. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable source of critical cancer therapeutics that combines scientific innovation with commercial reliability.
