Advanced One-Pot Semi-Synthesis of Taxane Derivatives for Commercial API Production
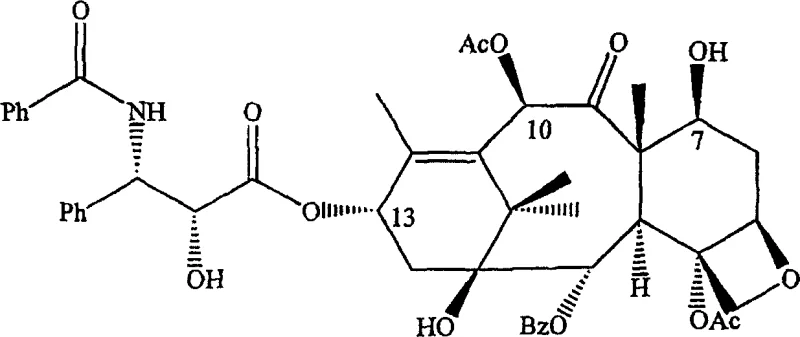
The pharmaceutical industry continuously seeks robust methodologies to enhance the production efficiency of critical oncology therapeutics, and patent CN1942460A presents a transformative approach to the semi-synthesis of taxane derivatives. This intellectual property details a sophisticated one-pot reaction strategy that streamlines the conversion of naturally occurring taxanes into high-value intermediates for paclitaxel and docetaxel. By integrating the protection of hydroxyl groups at the C-7 and C-10 positions with the simultaneous attachment of a side chain at the C-13 position, this technology fundamentally alters the traditional manufacturing landscape. The ability to execute these complex transformations within a single reaction vessel minimizes unit operations and significantly reduces the potential for yield loss associated with intermediate isolation. For a reliable taxane intermediate supplier, adopting such integrated processes is essential to maintaining competitiveness in the global API market. The structural complexity of taxanes, as exemplified by the intricate ring system and multiple stereocenters visible in the core molecule, demands precise chemical control which this patent addresses comprehensively.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional semi-synthetic routes for producing paclitaxel and docetaxel typically involve discrete, sequential steps that require the isolation and purification of intermediates after each chemical transformation. This multi-step methodology inherently introduces significant inefficiencies, including excessive solvent consumption, prolonged processing times, and cumulative yield losses at every isolation stage. Furthermore, the handling of sensitive taxane intermediates often necessitates stringent environmental controls to prevent degradation, thereby increasing operational costs and complexity. The reliance on multiple reactors and purification columns also expands the physical footprint required for manufacturing, which can be a bottleneck for facilities aiming to scale production rapidly. In many conventional processes, the protection of specific hydroxyl groups is performed as a standalone operation, followed by a separate reaction for side-chain attachment, creating opportunities for impurity generation and batch variability. These logistical and chemical challenges underscore the urgent need for process intensification strategies that can consolidate operations without compromising product quality or regulatory compliance.
The Novel Approach
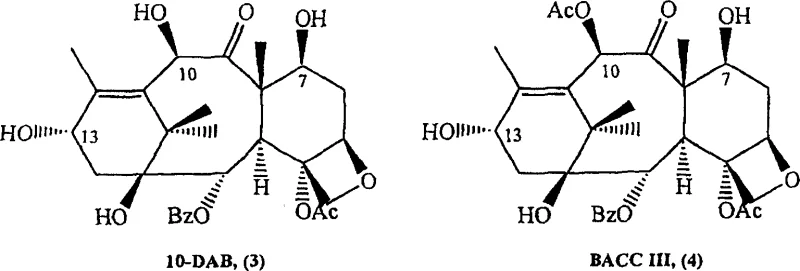
In stark contrast to legacy methods, the novel one-pot synthesis described in the patent data allows for the concurrent protection and functionalization of the taxane core within a unified reaction environment. This approach leverages the chemical compatibility of protecting agents and side-chain precursors to proceed sequentially in the same vessel, effectively eliminating the need for intermediate workup procedures. By utilizing starting materials such as 10-deacetylbaccatin III and baccatin III, which are readily accessible from natural sources or waste streams, the process ensures a stable supply of raw materials for continuous manufacturing. The integration of these steps not only accelerates the overall production timeline but also enhances the atom economy of the synthesis by reducing waste generation. This methodological shift represents a significant advancement in cost reduction in API manufacturing, as it directly addresses the primary drivers of production expenses related to labor, solvents, and equipment utilization. The ability to process complex mixtures of taxanes further amplifies the economic benefits by allowing manufacturers to utilize lower-grade starting materials that would otherwise be discarded.
Mechanistic Insights into One-Pot Protection and Side-Chain Attachment
The core chemical innovation lies in the precise orchestration of reaction conditions that enable selective protection of the C-7 and C-10 hydroxyl groups followed immediately by nucleophilic attack at the C-13 position. The process initiates with the introduction of a base and a hydroxyl protecting agent, such as a silylating agent or an acylating agent like di-tert-butyl dicarbonate, to mask the reactive hydroxyls that could interfere with subsequent steps. Once the protection is established in situ, the reaction mixture is treated with a side-chain precursor, typically a beta-lactam, oxazolidine, or oxazoline derivative, in the presence of additional base to facilitate the esterification at C-13. This tandem sequence requires careful control of temperature and stoichiometry to ensure that the protecting groups remain stable while the side-chain attachment proceeds with high regioselectivity. The mechanistic pathway avoids the formation of stable intermediate salts or complexes that would require filtration, thereby maintaining a homogeneous or manageable heterogeneous system throughout the transformation. Understanding this catalytic and stoichiometric balance is crucial for R&D teams aiming to replicate the process at a commercial scale while maintaining stringent purity specifications.
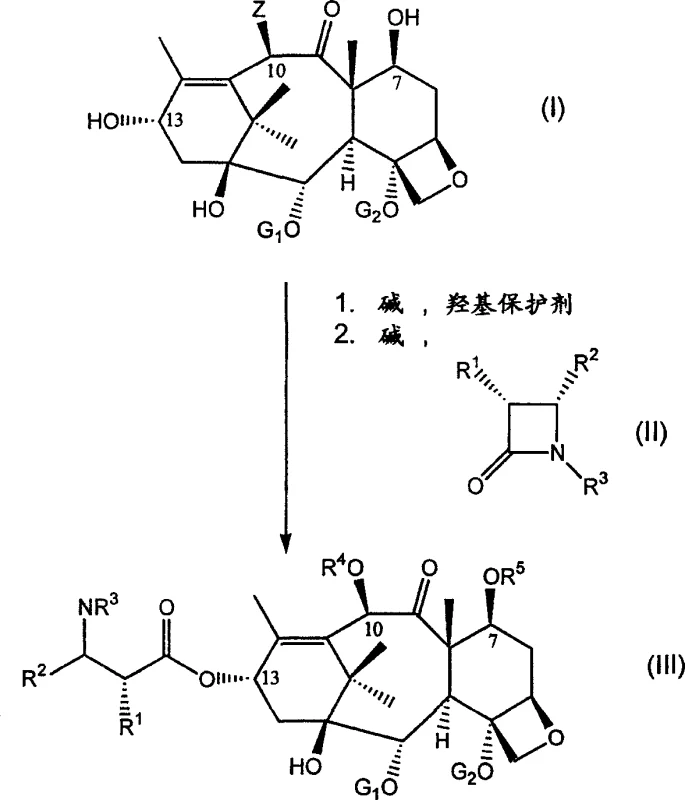
Impurity control is inherently improved in this one-pot system because the intermediate species are not exposed to external environments or purification media where degradation or contamination could occur. The continuous presence of reagents ensures that any transient species formed during the protection phase are immediately consumed in the side-chain attachment phase, minimizing the accumulation of by-products. Furthermore, the use of specific beta-lactam precursors allows for the introduction of the necessary stereochemistry at the C-13 side chain with high fidelity, which is critical for the biological activity of the final API. The patent highlights the versatility of using various protecting groups, enabling manufacturers to tailor the process to their specific downstream deprotection capabilities. This flexibility in mechanistic design ensures that the process can be adapted to different regulatory requirements or facility constraints without necessitating a complete re-engineering of the synthetic route. For technical teams, this means a robust platform technology that can be validated once and applied to multiple taxane analogs with minimal modification.
How to Synthesize C-13 Protected Taxane Intermediates Efficiently
Implementing this synthesis route requires a disciplined approach to reagent addition and temperature management to maximize the yield of the protected intermediate. The process begins with dissolving the taxane substrate in an anhydrous organic solvent under an inert atmosphere, followed by the controlled addition of base and protecting agent at low temperatures to initiate the protection phase. Once the protection is complete, as monitored by analytical methods, the side-chain precursor is introduced without quenching the reaction, allowing the second transformation to proceed seamlessly in the same vessel. Detailed standardized synthesis steps are essential to ensure reproducibility and safety, particularly when handling reactive organometallic bases or sensitive beta-lactam reagents. The following guide outlines the critical operational parameters derived from the patent data to assist technical teams in process development.
- Protect the free hydroxyl groups at the C-7 and/or C-10 positions of the taxane substrate using a suitable hydroxyl protecting agent and base.
- Without isolating the protected intermediate, add a side chain precursor such as a beta-lactam and additional base to attach the side chain at the C-13 position.
- Quench the reaction with acid, perform aqueous workup, and purify the resulting C-13 protected taxane intermediate via chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot technology offers substantial strategic benefits that extend beyond mere chemical efficiency to impact the entire supply chain ecosystem. By consolidating multiple reaction steps into a single operation, manufacturers can drastically reduce the consumption of solvents and reagents, which are significant cost drivers in fine chemical production. This reduction in material usage translates directly into lower variable costs per kilogram of product, enhancing the margin profile for high-volume API manufacturing. Additionally, the shortened processing time allows for faster turnover of reactor assets, increasing the overall capacity of the production facility without the need for capital-intensive expansion. For procurement managers, this means a more stable pricing structure and the ability to secure long-term supply agreements with reduced risk of cost volatility. The technology also supports sustainability goals by minimizing waste generation, which is increasingly important for meeting environmental compliance standards in global markets.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for extensive filtration, drying, and purification operations that typically consume significant energy and labor resources. By avoiding these unit operations, the process reduces the demand for auxiliary equipment and the associated maintenance costs, leading to a leaner operational model. The ability to use crude or partially purified starting materials further lowers the entry cost for raw materials, as expensive high-purity precursors are not strictly required for the reaction to proceed efficiently. This qualitative improvement in process economics allows for a more competitive pricing strategy in the global market for oncology intermediates. Consequently, the overall cost of goods sold is significantly optimized, providing a buffer against market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The flexibility to process various taxane mixtures, including waste streams from extraction processes, diversifies the sources of raw materials and reduces dependency on single suppliers of high-purity starting materials. This resilience is critical for maintaining continuous production schedules, especially when facing disruptions in the supply of natural extracts or synthetic precursors. The robust nature of the one-pot reaction also means that the process is less sensitive to minor variations in starting material quality, ensuring consistent output even with fluctuating feedstock specifications. For supply chain heads, this translates to reduced lead time for high-purity taxane intermediates and a more predictable delivery schedule for downstream API manufacturers. The ability to valorize waste streams also creates a circular economy aspect to the supply chain, enhancing long-term sustainability and resource security.
- Scalability and Environmental Compliance: Scaling this process is inherently simpler because it involves fewer transfer steps and unit operations, reducing the complexity of technology transfer from pilot to commercial scale. The reduced solvent volume and waste generation align with green chemistry principles, making it easier to obtain environmental permits and maintain compliance with increasingly strict regulations. The compact nature of the one-pot process allows for higher production density within existing facilities, maximizing the return on invested capital for manufacturing infrastructure. This scalability ensures that the commercial scale-up of complex taxane intermediates can be achieved rapidly to meet surging market demand without compromising quality. Furthermore, the reduced environmental footprint enhances the corporate social responsibility profile of the manufacturing entity, which is a key factor for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot taxane synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational capabilities and limitations of the process for potential partners. Understanding these details is crucial for making informed decisions about process adoption and supply chain integration. The responses highlight the versatility of the method and its alignment with modern manufacturing standards for pharmaceutical intermediates.
Q: What starting materials are compatible with this one-pot taxane synthesis process?
A: The process is highly versatile and can utilize 10-deacetylbaccatin III, baccatin III, and even complex mixtures found in spent taxane solutions or crude extracts, allowing for the valorization of waste streams.
Q: Which side chain precursors can be used for the C-13 attachment?
A: The method supports various precursors including beta-lactams, oxazolidines, and oxazolines, providing flexibility in sourcing and enabling the synthesis of both paclitaxel and docetaxel derivatives.
Q: How does the one-pot approach impact overall process efficiency?
A: By eliminating the isolation and purification steps between protection and side-chain attachment, the process significantly reduces solvent consumption, processing time, and material loss, leading to higher overall throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic technologies to deliver high-quality taxane intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for oncology drug manufacturing. Our commitment to process excellence means that we can leverage the efficiencies of the one-pot synthesis described in patent CN1942460A to offer competitive advantages to our clients. By partnering with us, you gain access to a supply chain that is both robust and adaptable to the evolving needs of the pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your specific supply chain requirements. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this streamlined manufacturing route. Our experts are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us collaborate to engineer a supply solution that balances cost, quality, and reliability for your critical API needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
