Advanced Semi-Synthetic Route for Paclitaxel and Docetaxel Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical oncology therapeutics, and patent CN100560576C presents a transformative approach to the semi-synthesis of paclitaxel and docetaxel. This specific intellectual property outlines a refined condensation strategy that utilizes protected 10-deacetylbaccatin 10-DAB and a synthesized chiral C-13 side chain to construct the complex taxane core with unprecedented efficiency. Unlike traditional extraction methods which rely on scarce plant resources, this chemical synthesis offers a sustainable and scalable alternative for producing high-purity pharmaceutical intermediates. The core innovation lies in the ability to manipulate stereochemistry during the coupling phase, thereby streamlining the entire production workflow. For global procurement teams and R&D directors, understanding this technological leap is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent quality and volume demands. This report analyzes the technical merits and commercial implications of adopting this advanced semi-synthetic methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
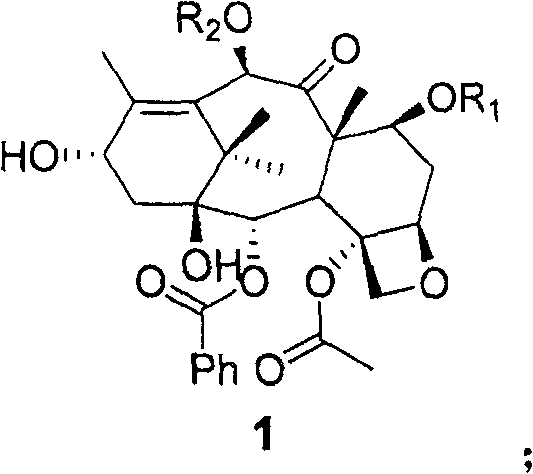
Prior art technologies, such as those described in patent PCT/CN2006/003150, often suffer from excessive synthetic complexity that hinders efficient commercial scale-up of complex polymer additives and API intermediates. These legacy routes typically require a multi-step sequence where the chiral side chain is attached first, followed by a distinct and often low-yielding step to invert the stereochemistry at the 2' position after deprotection. This separation of tasks introduces significant opportunities for material loss, increases the consumption of solvents and reagents, and extends the overall manufacturing lead time. Furthermore, each additional isolation and purification stage adds to the operational expenditure and complicates the impurity profile, making it challenging to meet the rigorous purity specifications required for oncology drugs. The cumulative effect of these inefficiencies results in a lower overall yield, historically hovering around 35.8%, which directly impacts the cost of goods sold and supply chain stability.
The Novel Approach
The methodology disclosed in CN100560576C fundamentally reengineers this workflow by integrating the stereochemical inversion directly into the initial condensation reaction. By optimizing the docking conditions between the novel chiral side chain and the protected 10-DAB scaffold, the process achieves the correct configuration at the 2' position in situ, effectively eliminating the need for a dedicated inversion step post-deprotection. This strategic consolidation shortens the synthetic route by three distinct steps, which drastically simplifies the manufacturing process and reduces the potential for side reactions. The result is a significant enhancement in overall yield, climbing to 55.9%, which represents a major breakthrough in cost reduction in API manufacturing. This streamlined approach not only improves economic viability but also enhances the environmental profile of the synthesis by reducing waste generation and solvent usage, aligning with modern green chemistry principles.
Mechanistic Insights into DCC-DMAP Catalyzed Condensation
The heart of this technological advancement is the precise control over the esterification reaction between the C-13 side chain and the C-13 hydroxyl group of the baccatin core. The process employs dicyclohexylcarbodiimide (DCC) as the condensing agent and N,N-dimethylaminopyridine (DMAP) as the catalyst, operating within a temperature range of 0 to 100 degrees Celsius. During this critical phase, the reaction conditions are tuned to facilitate not just the formation of the ester bond but also the simultaneous inversion of chirality at the 2' position of the side chain. This dual-functionality is achieved through careful selection of protecting groups such as TBS, Troc, or ketal derivatives, which stabilize the intermediate species while allowing the necessary stereochemical rearrangement to occur. The ability to manage this complex transformation in a single pot demonstrates a sophisticated understanding of organic reaction mechanisms and provides a robust platform for consistent batch-to-batch quality.
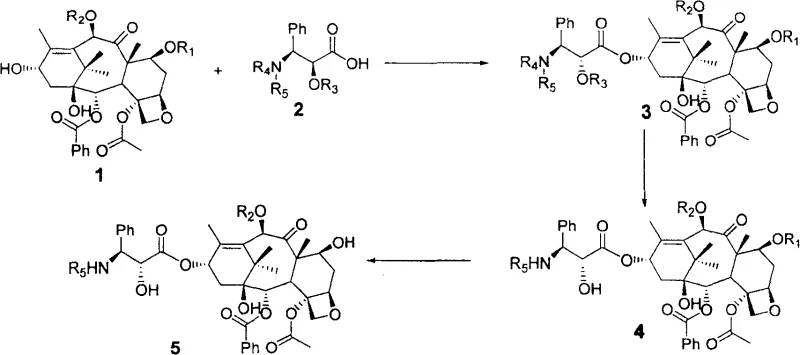
Controlling the impurity profile is paramount when synthesizing potent anti-cancer agents, and this method offers inherent advantages in this regard. By reducing the number of discrete chemical transformations, the process minimizes the generation of by-products and degradation compounds that often arise during prolonged exposure to reactive conditions. The use of mild reaction parameters ensures that the sensitive oxetane ring and the ester linkages within the taxane core remain intact, preserving the biological activity of the final product. Furthermore, the specific choice of protecting groups allows for selective deprotection in subsequent steps, enabling the divergent synthesis of either paclitaxel or docetaxel from a common intermediate. This flexibility is crucial for manufacturers who need to adapt quickly to market demands for different taxane variants without retooling their entire production line, ensuring a steady supply of high-purity OLED material or pharmaceutical grades.
How to Synthesize Paclitaxel Efficiently
The synthesis pathway described in the patent provides a clear roadmap for producing these critical oncology ingredients with high efficiency and reproducibility. The process begins with the condensation of the protected baccatin core and the chiral side chain, followed by sequential deprotection steps that reveal the active pharmacophore. Detailed standardized synthesis steps are provided below to guide technical teams in implementing this route.
- Condense protected 10-deacetylbaccatin 10-DAB with a novel chiral C-13 side chain using DCC and DMAP to form the intermediate ester while simultaneously inverting the 2' chirality.
- Perform deprotection of the side chain protecting groups on the intermediate compound to reveal the necessary hydroxyl functionalities for the final structure.
- Execute final deprotection of the 7-position for Paclitaxel or both 7 and 10 positions for Docetaxel using zinc powder in acetic acid to yield the final active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this semi-synthetic route translates into tangible strategic benefits that extend beyond simple unit cost metrics. The reduction in synthetic steps directly correlates with a decrease in operational complexity, which enhances the reliability of the supply chain and reduces the risk of production bottlenecks. By eliminating three steps from the traditional workflow, manufacturers can significantly reduce the consumption of raw materials, solvents, and energy, leading to substantial cost savings in the overall manufacturing budget. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, creating a win-win scenario for both producers and buyers in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The primary driver of cost optimization in this process is the drastic simplification of the synthetic route, which removes the need for expensive and time-consuming stereochemical inversion steps. By achieving the correct chirality during the initial condensation, the process avoids the yield losses associated with additional isolation and purification stages, thereby maximizing the output from every kilogram of starting material. This efficiency leads to a lower cost per gram of the final active ingredient, enabling significant financial advantages for downstream drug manufacturers who are constantly pressured to reduce healthcare costs. Additionally, the use of common reagents like DCC and DMAP ensures that material costs remain stable and predictable, avoiding the volatility associated with specialized catalysts.
- Enhanced Supply Chain Reliability: A shorter and more robust synthetic pathway inherently reduces the lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more agilely to fluctuations in market demand. The simplified process flow decreases the likelihood of batch failures and deviations, which are common causes of supply disruptions in complex chemical manufacturing. With fewer unit operations to monitor and control, quality assurance teams can maintain stricter oversight on critical parameters, ensuring consistent product quality and regulatory compliance. This reliability is essential for pharmaceutical companies that require uninterrupted access to key ingredients to maintain their own production schedules and meet patient needs globally.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced solvent usage make this process highly scalable from laboratory benchtop to multi-ton commercial production without significant engineering hurdles. The elimination of multiple workup and purification steps reduces the volume of chemical waste generated, simplifying waste treatment and lowering the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only meets increasingly stringent environmental regulations but also enhances the corporate social responsibility profile of the supply chain partners. Companies prioritizing sustainability will find this method particularly attractive as it supports their goals for reducing carbon emissions and resource consumption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this semi-synthetic technology. These answers are derived directly from the patent specifications and provide clarity on the operational advantages and chemical feasibility of the route. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing supply networks.
Q: How does this semi-synthetic method improve upon previous patent technologies?
A: This method eliminates the need for a separate stereochemical inversion step after deprotection, which was required in prior art like PCT/CN2006/003150. By integrating the inversion directly into the condensation reaction, the synthetic route is shortened by three steps, significantly reducing processing time and material loss.
Q: What are the typical yield improvements observed with this novel route?
A: Experimental data indicates a substantial increase in overall yield, rising from approximately 35.8% in conventional methods to 55.9% with this optimized process. This improvement is driven by the reduction in unit operations and the mild reaction conditions that preserve the integrity of the complex taxane core.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild, operating between 0 and 100 degrees Celsius, and utilize common reagents like DCC and DMAP. The simplification of the workflow and the robustness of the condensation step make it highly amenable to commercial scale-up for reliable API intermediate supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Supplier
The technical potential of this semi-synthetic route is immense, offering a pathway to more affordable and accessible cancer treatments through improved manufacturing efficiency. NINGBO INNO PHARMCHEM stands ready as a premier CDMO partner with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of paclitaxel or docetaxel meets the highest international standards. We understand the critical nature of these supply chains and are committed to delivering consistency and quality in every shipment.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the economic impact of switching to this advanced route. Our experts are available to provide specific COA data and route feasibility assessments tailored to your volume and timeline needs, ensuring a seamless transition to a more efficient supply model.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
