Advanced Synthesis of Renin Inhibitor Intermediates for Commercial Scale Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic pathways for active pharmaceutical ingredients, particularly for cardiovascular treatments such as hypertension management. Patent CN1089604A introduces a groundbreaking approach to synthesizing amino acid derivatives with renin inhibiting activity, addressing critical bottlenecks in existing manufacturing protocols. This technical insight report analyzes the novel synthetic intermediate (II) and its application in producing the target compound (I), offering a strategic advantage for supply chain optimization. The disclosed method leverages specific peptide condensation techniques and protecting group strategies to enhance overall process efficiency. By shifting away from traditional, cost-prohibitive precursors, this technology enables the production of high-purity Pharmaceutical Intermediates with improved economic viability. For R&D Directors and Procurement Managers, understanding the mechanistic advantages of this route is essential for evaluating long-term supplier partnerships and cost-reduction strategies in the competitive landscape of antihypertensive drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, specifically those disclosed in documents like EP-A-300189, rely heavily on expensive starting materials that significantly inflate the cost of goods sold for the final active ingredient. The conventional synthesis typically utilizes compound (VI) as a key building block, which is not only costly to procure but also limits the scalability of the process due to supply chain constraints. Furthermore, historical data indicates that these traditional routes often suffer from suboptimal yields, frequently ranging between 40-50%, which necessitates larger batch sizes to meet production targets and generates increased chemical waste. The reliance on such inefficient pathways creates substantial pressure on procurement budgets and complicates inventory management for large-scale pharmaceutical manufacturers. Additionally, the purification steps required to remove by-products from these older methods are often labor-intensive and require specialized chromatography, further adding to the operational expenditure. For Supply Chain Heads, these inefficiencies translate into longer lead times and reduced reliability in meeting market demand for critical hypertension medications.
The Novel Approach
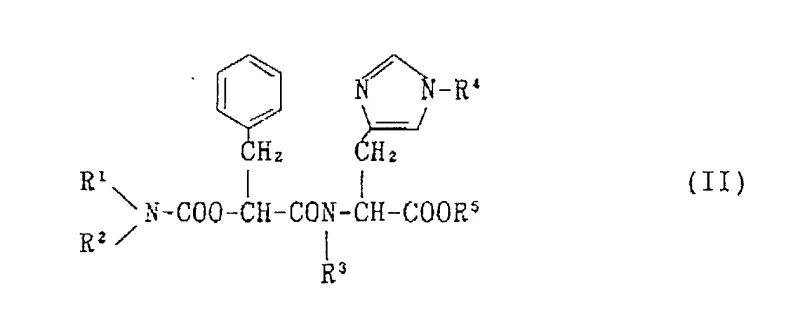
The innovative methodology presented in CN1089604A circumvents these historical challenges by introducing a novel intermediate (II) that serves as a superior building block for the final assembly of the target molecule. This new route utilizes more economically accessible raw materials, effectively decoupling production costs from the volatility of specialized reagent markets. The strategic design of intermediate (II) allows for a more streamlined condensation process, which inherently reduces the number of purification steps required and minimizes material loss during synthesis. By optimizing the reaction conditions and selecting appropriate protecting groups, the new method achieves significantly higher yields compared to the 40-50% benchmark of the prior art. This improvement in efficiency directly correlates to a reduction in solvent usage and waste generation, aligning with modern environmental compliance standards. For procurement teams, this translates to a more stable pricing structure and a reliable Pharmaceutical Intermediates supplier capable of sustaining long-term commercial contracts without the risk of raw material shortages.
Mechanistic Insights into Peptide Condensation and Protecting Group Strategy
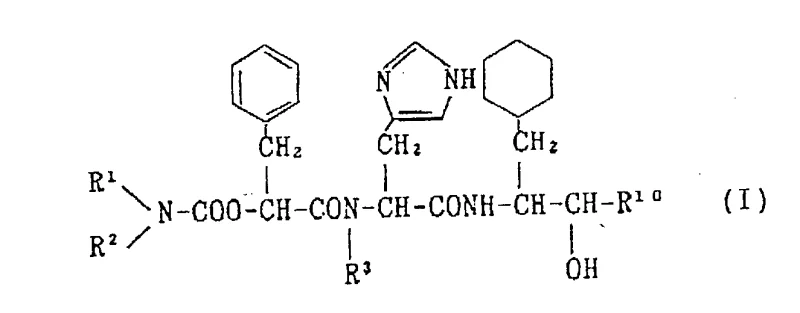
The core of this synthetic advancement lies in the precise control of peptide bond formation and the strategic manipulation of nitrogen-protecting groups to ensure stereochemical integrity. The synthesis of intermediate (II) involves the condensation of compound (IV) and compound (V), where the carboxyl group of one component reacts with the amino group of the other under carefully controlled conditions. The use of activating agents such as carbodiimides or active esters facilitates this coupling, ensuring high conversion rates while minimizing racemization at the chiral centers. The selection of protecting groups, such as the trityl group for the imidazole nitrogen and various alkoxycarbonyl groups for the alpha-amino functions, is critical for preventing side reactions during the multi-step sequence. These protecting groups are stable under the reaction conditions but can be selectively removed in the final stages without compromising the structural integrity of the sensitive peptide backbone. This level of chemical precision is vital for R&D Directors who must ensure that the impurity profile of the intermediate meets stringent regulatory specifications for downstream drug substance manufacturing.
Furthermore, the mechanism includes specific steps for the introduction of alkyl groups and the subsequent deprotection sequences that define the final pharmacological activity of the derivative. The process allows for the flexibility to introduce various substituents at the R1, R2, and R3 positions, enabling the synthesis of a diverse library of analogues for structure-activity relationship studies. The deprotection steps, often involving acidic or basic hydrolysis or catalytic hydrogenation, are optimized to proceed cleanly, avoiding the formation of difficult-to-remove impurities. For example, the removal of the trityl group is managed under conditions that preserve the stereochemistry of the adjacent chiral centers, which is essential for maintaining the renin inhibiting potency. This robust mechanistic framework ensures that the commercial scale-up of complex Pharmaceutical Intermediates can be achieved with consistent quality, providing a solid foundation for regulatory filings and clinical trial material supply.
How to Synthesize Renin Inhibitor Intermediate Efficiently
The practical implementation of this synthesis route requires adherence to specific operational parameters to maximize yield and purity while ensuring safety during scale-up. The process begins with the preparation of the key intermediates, followed by a series of condensation and deprotection reactions that must be monitored closely for completion. Detailed standard operating procedures dictate the stoichiometry of reagents, the choice of solvents such as dichloromethane or tetrahydrofuran, and the precise temperature controls needed for exothermic steps. The following guide outlines the critical phases of the production workflow, emphasizing the points where quality control interventions are most effective. Operators must be trained to recognize the visual and analytical indicators of reaction progress to prevent the accumulation of by-products. This structured approach ensures that the high-purity Pharmaceutical Intermediates produced meet the rigorous standards expected by global pharmaceutical clients.
- Prepare the novel intermediate (II) by condensing compound (IV) and compound (V) using standard peptide coupling agents.
- React the resulting intermediate (II) with compound (III) under controlled temperature conditions to form the protected precursor.
- Perform deprotection of the nitrogen-protecting group to yield the final high-purity amino acid derivative (I).
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this novel synthesis route offers transformative benefits for the commercial viability of producing renin inhibitor intermediates, directly addressing the pain points of cost and reliability in the pharmaceutical supply chain. By eliminating the dependency on expensive and scarce raw materials like compound (VI), manufacturers can achieve substantial cost savings that can be passed down to the end client or reinvested into process optimization. The improved yield efficiency means that less raw material is required to produce the same amount of final product, reducing the overall material footprint and waste disposal costs associated with the manufacturing process. For Procurement Managers, this stability in raw material sourcing mitigates the risk of supply disruptions and price volatility, ensuring a consistent flow of goods to meet production schedules. The simplified purification process also reduces the consumption of solvents and chromatography media, further contributing to a leaner and more sustainable manufacturing operation.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of high-cost precursors with readily available, cheaper alternatives without sacrificing product quality. This shift fundamentally alters the cost structure of the synthesis, allowing for a more competitive pricing model in the global market for Pharmaceutical Intermediates. The reduction in step count and the avoidance of complex purification protocols further lower the operational expenses, making the process economically attractive for large-volume production. Additionally, the higher yield reduces the cost per kilogram of the active intermediate, providing a direct margin improvement for the manufacturer. These qualitative efficiencies ensure that the supply chain remains resilient against market fluctuations in reagent pricing.
- Enhanced Supply Chain Reliability: Sourcing raw materials that are commercially available in bulk quantities significantly enhances the reliability of the supply chain, reducing the lead time for high-purity Pharmaceutical Intermediates. Unlike specialized reagents that may have long procurement cycles, the starting materials for this novel route are standard chemical commodities with multiple global suppliers. This diversification of the supply base protects against single-source failures and ensures continuity of supply even during periods of high market demand. For Supply Chain Heads, this reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery deadlines imposed by pharmaceutical customers. The robustness of the supply chain directly translates to improved customer satisfaction and stronger long-term contractual relationships.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The avoidance of hazardous reagents and the reduction in waste generation align with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing facilities. The simplified work-up procedures minimize the use of volatile organic compounds, contributing to a safer working environment and lower emissions. This environmental stewardship is a key value proposition for pharmaceutical companies looking to partner with suppliers who prioritize sustainability. The ability to scale up complex Pharmaceutical Intermediates efficiently ensures that the technology can meet the growing global demand for antihypertensive medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its potential. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, ensuring accuracy and relevance. Understanding these details is crucial for making informed decisions about process adoption and supplier selection. The information covers aspects of yield, purity, and scalability that are of primary concern to technical and procurement teams. This section serves as a quick reference guide for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What is the primary advantage of the novel intermediate (II) over prior art methods?
A: The novel intermediate (II) allows for the use of significantly cheaper raw materials compared to the expensive compound (VI) used in traditional methods, while simultaneously achieving higher overall yields.
Q: How does this synthesis route impact impurity profiles for R&D teams?
A: By utilizing specific protecting groups like trityl and optimized condensation conditions, the method minimizes side reactions, resulting in a cleaner impurity profile that simplifies downstream purification.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process avoids extremely harsh conditions and utilizes readily available reagents, making it highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino Acid Derivative Supplier
The technical potential of the synthesis route described in CN1089604A represents a significant opportunity for optimizing the production of renin inhibitor intermediates, and NINGBO INNO PHARMCHEM is uniquely positioned to capitalize on this innovation. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Amino Acid Derivative meets the highest international standards. We understand the critical nature of supply chain continuity in the pharmaceutical industry and are committed to delivering consistent quality and reliability. Partnering with us means gaining access to a team of experts dedicated to solving complex synthetic challenges and driving cost efficiency.
We invite you to initiate a dialogue with our technical procurement team to explore how this novel synthesis route can benefit your specific product portfolio. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient manufacturing process. Our team is ready to provide specific COA data and route feasibility assessments tailored to your requirements. By collaborating with NINGBO INNO PHARMCHEM, you secure a reliable Amino Acid Derivative supplier who is committed to your success. Let us help you reduce lead time for high-purity Pharmaceutical Intermediates and achieve your commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
