Optimizing Apixaban Intermediate Production via Novel Lactone Amidation Routes
Optimizing Apixaban Intermediate Production via Novel Lactone Amidation Routes
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value anticoagulants like Apixaban, a direct Factor Xa inhibitor widely marketed under the trade name Eliquis. A critical bottleneck in the supply chain has historically been the efficient production of key intermediates, specifically the 5,6-dihydropyridin-2(1H)-one derivatives. Patent CN111170992B introduces a groundbreaking preparation method for these derivatives, specifically targeting compounds where the R substituent is either chlorine or a morpholine-4-yl group. This innovation addresses the longstanding reliance on expensive iodo-substances and harsh chlorinating agents, proposing a greener, cost-effective alternative starting from p-acetamino aniline. By leveraging delta-valerolactone and 2,2-dichloro-delta-valerolactone, this technology enables the synthesis of 1-(piperidine-2-ketone-1-yl)-4-(5,6-dihydro-3-R substituent pyridine-2(1H)-ketone-1-yl) benzene with exceptional purity and yield.
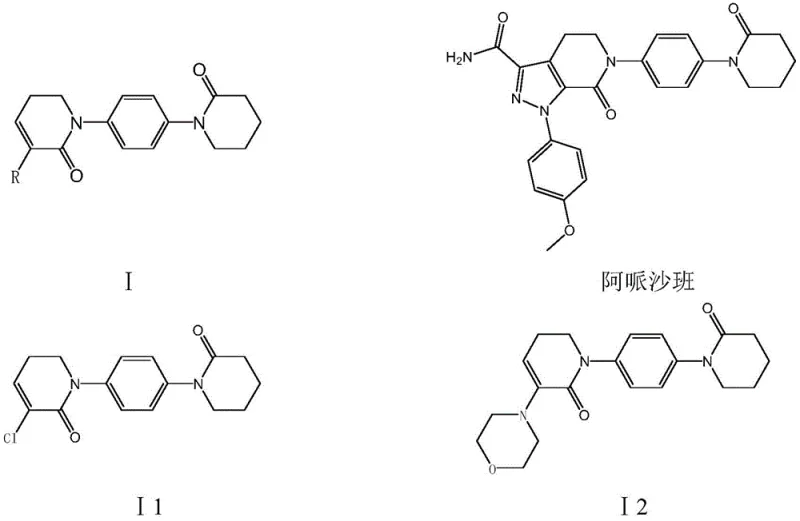
The structural complexity of Apixaban necessitates precise intermediate synthesis to ensure final drug efficacy and safety. The intermediates described in this patent, labeled as I1 and I2, serve as the foundational scaffolds upon which the pyrazolo-pyridine core is built. Traditional methods often struggle with regioselectivity and the management of toxic byproducts, leading to supply chain volatility. The novel approach detailed in CN111170992B not only streamlines the molecular construction but also aligns with modern green chemistry principles by reducing the generation of hazardous waste. For R&D directors and procurement managers, understanding this shift from iodine-based chemistry to lactone-based amidation is crucial for securing a stable, long-term supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those disclosed in WO2003049681 and WO2004083177, rely heavily on 1-(4-iodophenyl)-2-piperidone or its dichloro-derivatives as starting materials. These iodo-substances are inherently expensive due to the cost of iodine and the complex multi-step synthesis required to introduce the iodine atom onto the phenyl ring. Furthermore, one prominent conventional route utilizes phosphorus pentachloride (PCl5) for chlorination, a reaction known for its low selectivity and the generation of substantial amounts of phosphorus-containing wastewater, posing significant environmental disposal challenges. Another route employs a cuprous bromide-triphenylphosphine catalyst for coupling reactions, which requires high temperatures and adds heavy metal contamination risks that necessitate costly purification steps. Consequently, the total yield of Apixaban via these traditional pathways is often dismally low, reported at merely 5.2% in some instances, rendering them economically unviable for large-scale commercial production.
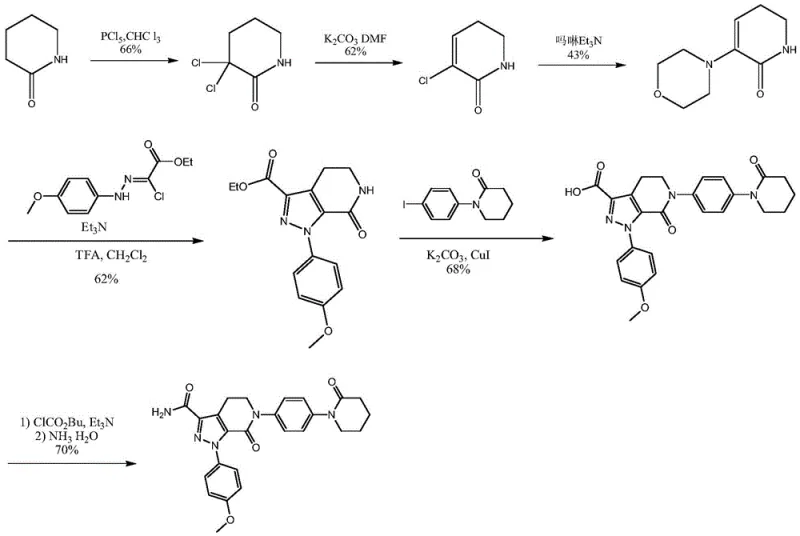
The Novel Approach
In stark contrast, the method disclosed in CN111170992B circumvents these economic and environmental hurdles by utilizing p-acetamino aniline and delta-valerolactone as primary feedstocks. These raw materials are commodity chemicals, widely available in the global market at a fraction of the cost of iodo-precursors. The new process eliminates the need for phosphorus pentachloride and expensive transition metal catalysts entirely. Instead, it employs a sequence of amidation, halogenation (or sulfonylation), and intramolecular condensation reactions that proceed under mild conditions. This strategic shift results in a dramatic improvement in overall efficiency, with the patent reporting a total yield reaching up to 72%. By avoiding high-temperature coupling reactions and utilizing specific functional group transformations, the new route ensures high reaction selectivity, minimizing the formation of difficult-to-remove impurities and significantly lowering the operational expenditure for manufacturers.
Mechanistic Insights into Lactone-Mediated Cyclization and Amidation
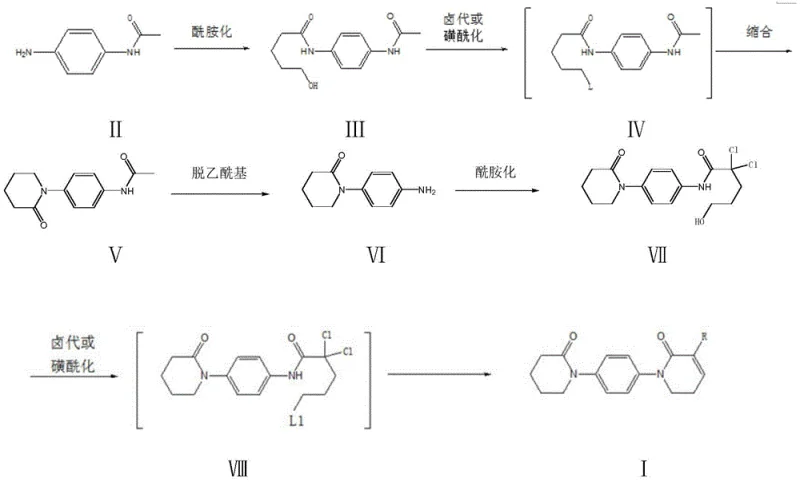
The core of this innovative synthesis lies in the precise manipulation of lactone rings to construct the piperidinone and dihydropyridinone moieties. The mechanism initiates with the nucleophilic attack of the amino group of p-acetamino aniline on the carbonyl carbon of delta-valerolactone. This amidation reaction, typically conducted in solvents like toluene or DMF at temperatures between 90°C and 120°C, opens the lactone ring to form a hydroxy-amide intermediate (Compound III). The subsequent activation of the hydroxyl group is critical; it is converted into a good leaving group via halogenation with thionyl chloride or sulfonylation with sulfonyl chlorides. This activation primes the molecule for an intramolecular nucleophilic substitution, where the amide nitrogen attacks the activated carbon, closing the ring to form the 4-(piperidin-2-one-1-yl) structure (Compound V). This cyclization is highly favored entropically and is driven by the removal of the leaving group under basic conditions.
Following the formation of the first ring, the acetyl protecting group is removed under acidic conditions to reveal the free aniline nitrogen, which then undergoes a second amidation with 2,2-dichloro-delta-valerolactone. This step introduces the necessary dichloro-substituted chain required for the final heterocyclic construction. The final transformation involves another activation of the terminal hydroxyl group followed by a base-mediated elimination or substitution. If morpholine is present, a substitution occurs to yield the morpholino-derivative (I2); otherwise, elimination yields the chloro-derivative (I1). This stepwise construction ensures that each functional group reacts at a specific site, preventing cross-reactions and ensuring the high purity observed in the examples. The chemoselectivity is paramount, as the reaction conditions are tuned to affect only the intended hydroxyl or amino groups without disturbing the existing amide bonds or the aromatic ring.
How to Synthesize 5,6-Dihydropyridin-2-one Derivatives Efficiently
The synthesis of these critical Apixaban intermediates requires strict adherence to the reaction parameters outlined in the patent to maximize yield and purity. The process is divided into five distinct operational stages, beginning with the solvent selection and temperature control for the initial amidation. Operators must ensure that the molar ratio of delta-valerolactone to p-acetamino aniline is maintained between 1.2:1 and 2.0:1 to drive the reaction to completion without excessive reagent waste. The subsequent halogenation or sulfonylation steps require careful temperature management, typically keeping the reaction between 0°C and 70°C depending on the specific reagent used, to prevent degradation of the sensitive intermediates. Detailed standard operating procedures regarding work-up, such as hot filtration and recrystallization from isopropanol, are essential to achieve the reported liquid phase purities of >99.5%.
- Perform amidation between p-acetamino aniline and delta-valerolactone in solvent A at 90-120°C to form compound III.
- Subject compound III to halogenation or sulfonylation, followed by condensation with alkali to cyclize into compound V.
- Remove the acetyl protecting group from compound V using acid in alcohol to obtain compound VI.
- React compound VI with 2,2-dichloro-delta-valerolactone to form the second amide linkage, yielding compound VII.
- Activate compound VII via halogenation or sulfonylation, then perform condensation-elimination or substitution with morpholine to yield the final derivative I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN111170992B represents a strategic opportunity to optimize the cost structure of API manufacturing. The primary driver for cost reduction is the substitution of high-value iodo-compounds with low-cost commodity chemicals like p-acetamino aniline and valerolactone. This shift drastically reduces the raw material bill of materials (BOM) and mitigates the price volatility associated with iodine markets. Furthermore, the elimination of expensive catalysts such as cuprous bromide-triphenylphosphine removes a significant line item from the production budget. The simplified process flow, characterized by fewer purification steps and higher yields, translates directly into lower processing costs per kilogram of finished intermediate, enhancing the overall margin profile for downstream API producers.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived from the fundamental chemistry rather than marginal process tweaks. By avoiding the use of phosphorus pentachloride, manufacturers eliminate the need for specialized corrosion-resistant equipment and the costly treatment of phosphorus-laden wastewater. The high selectivity of the reactions means that solvent recovery rates are improved, and the loss of product to side-reactions is minimized. This efficiency allows for a significant reduction in the cost of goods sold (COGS) without compromising on quality. The ability to achieve high yields in each step cumulatively boosts the final output, meaning less raw material is required to produce the same amount of active intermediate, further driving down unit costs.
- Enhanced Supply Chain Reliability: Reliance on specialized iodo-intermediates often creates single points of failure in the supply chain, as few suppliers possess the capability to produce them at scale. By switching to a route based on widely available anilines and lactones, manufacturers can diversify their supplier base and reduce lead times. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, ensures that production can be maintained consistently across different manufacturing sites. This flexibility is crucial for maintaining business continuity and meeting the rigorous delivery schedules demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental footprint of this process is substantially smaller than that of conventional methods. The reduction in hazardous waste generation simplifies regulatory compliance and lowers disposal fees. The process is inherently safer, operating at moderate temperatures and avoiding the handling of large quantities of reactive phosphorus halides. This safety profile facilitates easier scale-up from pilot plant to commercial tonnage, as the thermal hazards are well-managed. For companies aiming to meet sustainability goals, this green chemistry approach offers a tangible way to reduce the environmental impact of their manufacturing operations while maintaining high productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and beneficial effects described in patent CN111170992B, providing clarity on how this technology compares to established industry standards. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements.
Q: Why is the new synthesis route for Apixaban intermediates considered superior to prior art?
A: The new route described in CN111170992B avoids the use of expensive and unstable iodo-substituted raw materials like 1-(4-iodophenyl)-2-piperidone. Instead, it utilizes cheap and readily available p-acetamino aniline and delta-valerolactone. Furthermore, it eliminates the need for harsh phosphorus pentachloride chlorination and expensive copper catalysts, resulting in significantly higher total yields (up to 72%) compared to previous methods which yielded as low as 5.2%.
Q: How does this process improve impurity control and product purity?
A: The process achieves high purity through highly selective unit reactions. The amidation occurs specifically between the amino group and the lactone ester, while subsequent halogenation targets only the hydroxyl group. This chemoselectivity minimizes side reactions. Additionally, the intramolecular condensation steps are optimized to prevent polymerization, ensuring liquid phase purities consistently above 99.5% for key intermediates.
Q: Is this manufacturing method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial scalability. It operates under mild reaction conditions (typically 50-120°C) that are easy to realize in standard reactors. The process generates minimal wastewater, particularly avoiding the large amounts of phosphorus-containing waste associated with PCl5 chlorination, making it environmentally compliant and safer for green manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-Dihydropyridin-2-one Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at implementing complex organic syntheses, such as the lactone-mediated route described in CN111170992B, ensuring that every batch meets stringent purity specifications. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every lot, guaranteeing that our 5,6-dihydropyridin-2-one derivatives are perfectly suited for the synthesis of Apixaban. Our commitment to quality assurance ensures that our clients receive materials that facilitate smooth downstream processing and final API registration.
We invite global partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us to obtain specific COA data for our intermediates and to receive comprehensive route feasibility assessments tailored to your production requirements. Let us collaborate to build a more resilient and cost-effective supply chain for next-generation anticoagulants.
