Revolutionizing Alpha-Aminonitrile Production: A Metal-Free Visible Light Protocol for Scalable Pharma Intermediates
Introduction to Next-Generation Cyanation Technology
The landscape of organic synthesis is undergoing a profound transformation driven by the urgent need for greener, safer, and more cost-effective manufacturing protocols. A pivotal advancement in this domain is detailed in Chinese Patent CN112390696B, which discloses a novel method for preparing alpha-aminonitriles, a class of compounds serving as critical building blocks for amino acids, alkaloids, and various nitrogen-containing heterocycles. Unlike conventional approaches that rely on hazardous cyanide sources or expensive transition metal catalysts, this invention leverages visible light irradiation to drive the coupling of tertiary amines with benzoyl cyanide. This metal-free photocatalytic strategy operates under remarkably mild conditions, typically at room temperature, and achieves high yields without the necessity for external photocatalysts in many instances. For R&D directors and process chemists, this represents a paradigm shift towards sustainable chemistry that does not compromise on efficiency or product quality.
The significance of this technology extends beyond the laboratory bench, offering tangible benefits for supply chain stability and regulatory compliance. By replacing toxic inorganic cyanides with a safer organic alternative and eliminating heavy metal residues, the process simplifies purification workflows and reduces the environmental footprint of pharmaceutical intermediate production. As the industry moves towards stricter impurity controls and greener manufacturing standards, adopting such innovative methodologies becomes not just an option but a strategic imperative for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-aminonitriles has been dominated by the Strecker reaction and oxidative coupling methods, both of which present substantial challenges for modern industrial application. The classic Strecker reaction typically involves the condensation of aldehydes and amines followed by cyanation using reagents like sodium cyanide (NaCN) or potassium cyanide (KCN). These inorganic salts are acutely toxic, posing severe safety risks to personnel and requiring specialized containment infrastructure to prevent environmental release. Furthermore, the handling of such hazardous materials necessitates rigorous waste treatment protocols, significantly driving up operational costs and complicating regulatory approvals.
Alternatively, dehydrogenative coupling methods often employ stoichiometric oxidants and transition metal catalysts, such as copper or palladium complexes, to activate the amine substrate. While effective, these metal-catalyzed routes introduce the risk of heavy metal contamination in the final product, a critical concern for pharmaceutical intermediates destined for API synthesis. The removal of trace metals to meet stringent ppm limits often requires additional purification steps, such as scavenging or recrystallization, which erode overall yield and increase production time. Moreover, the reliance on noble metals exposes the supply chain to price volatility and geopolitical sourcing risks, making long-term cost planning difficult for procurement managers.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN112390696B introduces a streamlined, catalyst-free pathway that utilizes visible light as the sole energy source for activation. The core innovation lies in the direct use of benzoyl cyanide as a cyanating agent, which reacts with tertiary amines under irradiation from purple LEDs (390-400 nm). This approach completely bypasses the need for toxic inorganic cyanides and expensive metal catalysts, thereby addressing both safety and cost pain points simultaneously. The reaction proceeds efficiently at room temperature, eliminating the energy consumption associated with heating or cooling large-scale reactors.
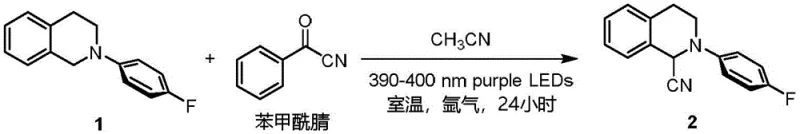
The versatility of this method is demonstrated by its broad substrate scope, accommodating various substituted tetrahydroisoquinolines and other cyclic amines with excellent functional group tolerance. As illustrated in the reaction scheme, the process delivers the target alpha-aminonitrile in high yields, often exceeding 80%, without the formation of complex byproduct mixtures typical of radical oxidation pathways. This simplicity translates directly into easier downstream processing, as the crude reaction mixtures are cleaner and require less intensive chromatographic separation. For manufacturing teams, this means faster batch turnover times and higher throughput capacity without the need for capital-intensive equipment upgrades.
Mechanistic Insights into Visible Light-Promoted Cyanation
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring robust process control. The reaction likely proceeds through a photo-induced electron transfer (PET) mechanism or the formation of an electron donor-acceptor (EDA) complex between the tertiary amine and benzoyl cyanide. Upon absorption of visible light photons, the excited state species facilitates the homolytic cleavage of the alpha-C-H bond adjacent to the nitrogen atom, generating an alpha-amino radical intermediate. This highly reactive radical species then undergoes rapid coupling with the cyano group derived from the benzoyl cyanide, leading to the formation of the C-CN bond.
The absence of an external photocatalyst suggests that either the substrates themselves possess sufficient absorption in the visible region or that a transient charge-transfer complex is formed in situ, which acts as the photoactive species. This self-sensitized nature of the reaction is a significant advantage, as it removes the variable of catalyst loading and potential catalyst degradation over time. Furthermore, the use of benzoyl cyanide serves a dual purpose: it acts as both the cyanide source and potentially as an electron acceptor in the initial excitation step, streamlining the reagent profile. The mild conditions prevent the decomposition of sensitive functional groups, ensuring high chemical fidelity in the final product.
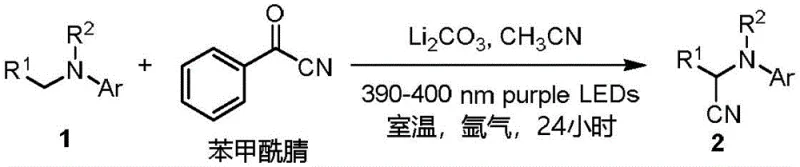
Impurity control is inherently superior in this system due to the specificity of the radical generation. Unlike thermal oxidation methods that can lead to over-oxidation or N-dealkylation side reactions, the photochemical pathway is highly selective for the alpha-position. The patent data indicates that even with diverse substituents on the aromatic rings, such as halogens, methoxy groups, or trifluoromethyl groups, the reaction maintains high selectivity. This predictability is vital for R&D teams aiming to minimize the formation of genotoxic impurities or difficult-to-remove structural analogs. The ability to tune the reaction outcome by simply adjusting the wavelength of the light source or the solvent polarity offers an additional layer of process optimization that is not available in traditional thermal chemistry.
How to Synthesize Alpha-Aminonitrile Efficiently
Implementing this synthesis route in a pilot or production setting requires adherence to specific operational parameters to maximize yield and safety. The general procedure involves dissolving the tertiary amine and benzoyl cyanide in a polar aprotic solvent like acetonitrile, optionally adding a mild base such as lithium carbonate to facilitate proton transfer. The reaction mixture is then subjected to irradiation from 390-400 nm LEDs at room temperature for approximately 24 hours under an inert or oxygen atmosphere. Detailed standardized synthetic steps see the guide below.
- Combine the tertiary amine substrate and benzoyl cyanide in an organic solvent such as acetonitrile within a reaction vessel.
- Maintain the reaction mixture at room temperature under an inert atmosphere or oxygen, and irradiate with 390-400 nm purple LEDs for approximately 24 hours.
- Upon completion, remove the solvent via vacuum spin-drying and purify the crude residue using silica gel column chromatography to isolate the high-purity alpha-aminonitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light mediated protocol offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The elimination of noble metal catalysts represents a direct reduction in raw material costs, removing the dependency on fluctuating markets for palladium, rhodium, or iridium. Additionally, the removal of metal catalysts simplifies the purification process, reducing the consumption of silica gel, solvents, and metal scavengers, which collectively contribute to a significantly lower cost of goods sold (COGS). The use of benzoyl cyanide, a stable and commercially available solid, further enhances supply chain reliability compared to gaseous or highly toxic liquid cyanide sources that require special shipping and storage protocols.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the bill of materials. By removing the need for expensive transition metal catalysts and toxic inorganic cyanides, manufacturers can achieve substantial cost savings on raw material procurement. Furthermore, the mild reaction conditions reduce energy consumption, as there is no need for heating mantles or cryogenic cooling systems, leading to lower utility costs per kilogram of product. The simplified workup procedure also reduces labor hours and solvent waste disposal fees, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Sourcing high-purity reagents with consistent quality is a perennial challenge, but this method utilizes widely available commodity chemicals like acetonitrile and benzoyl cyanide. The robustness of the reaction against moisture and oxygen variations (as shown in the patent examples with both argon and oxygen atmospheres) ensures consistent batch-to-batch performance, minimizing the risk of production delays due to failed runs. This reliability allows for more accurate demand forecasting and inventory management, ensuring that downstream API synthesis schedules are met without interruption.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a bottleneck, but advancements in LED technology and flow chemistry have made this increasingly viable. The low thermal load of this reaction makes it ideal for scale-up, as heat dissipation is minimal compared to exothermic thermal reactions. From an environmental perspective, the avoidance of heavy metals and highly toxic cyanides aligns perfectly with green chemistry principles, facilitating easier regulatory approval and reducing the liability associated with hazardous waste disposal. This positions the manufacturer as a leader in sustainable chemical production, a key differentiator in today's eco-conscious market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light cyanation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios.
Q: What are the primary safety advantages of using benzoyl cyanide over traditional cyanation reagents?
A: Traditional methods often rely on highly toxic inorganic salts like sodium cyanide (NaCN) or volatile organic cyanides. This patent utilizes benzoyl cyanide, which is a solid reagent with significantly lower toxicity and volatility, drastically reducing handling risks and environmental hazards during large-scale manufacturing.
Q: Does this photocatalytic method require expensive transition metal catalysts?
A: No, one of the most significant breakthroughs of this technology is that it operates without the need for additional photocatalysts or noble metal catalysts. The reaction proceeds efficiently under visible light irradiation alone, eliminating the cost of precious metals and the complex downstream removal processes associated with metal contamination.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the process operates at room temperature and atmospheric pressure using standard visible light sources, avoiding the extreme conditions often required by thermal methods. These mild parameters, combined with the use of common solvents like acetonitrile, make the protocol highly adaptable for commercial scale-up in standard reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Aminonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free photocatalytic synthesis in the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112390696B can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of alpha-aminonitrile meets the exacting standards required for API synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel alpha-aminonitrile derivatives or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
