Advanced Paclitaxel Semi-Synthesis: Oxazolidine Coupling for Commercial Scale-up
The pharmaceutical industry continuously seeks robust methodologies for the production of complex anticancer agents, and patent CN1158126A presents a significant advancement in the semi-synthetic preparation of paclitaxel and its derivatives. This technology addresses the critical challenge of attaching the C-13 side chain to the baccatin III nucleus with high stereochemical fidelity, a step that has historically limited the economic viability of total synthesis. By leveraging novel oxazolidine and oxazoline intermediates, the process ensures that the biological activity essential for tumor suppression is maintained without the formation of inactive epimers. 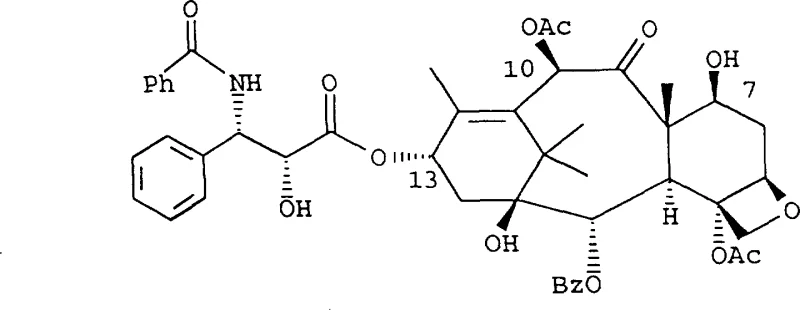 . The strategic implementation of this route allows manufacturers to bypass the ecological constraints associated with extracting sufficient quantities from natural yew sources, thereby securing a more stable supply of this vital medication for global healthcare systems.
. The strategic implementation of this route allows manufacturers to bypass the ecological constraints associated with extracting sufficient quantities from natural yew sources, thereby securing a more stable supply of this vital medication for global healthcare systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional esterification methods for linking the side chain to the baccatin core often rely on harsh conditions that compromise the integrity of the sensitive molecular structure. For instance, protocols utilizing excess DCC and DMAP at elevated temperatures frequently result in the unwanted epimerization at the C-2' carbon, leading to a mixture of diastereomers that complicates downstream purification. This lack of selectivity not only reduces the overall yield of the active pharmaceutical ingredient but also necessitates costly and time-consuming chromatographic separation steps to remove impurities. Furthermore, the steric hindrance surrounding the C-13 hydroxyl group on the baccatin nucleus poses a significant kinetic barrier, requiring aggressive reagents that can degrade other functional groups within the complex taxane ring system. These inefficiencies create substantial bottlenecks in manufacturing, driving up the cost of goods and limiting the ability to meet large-scale demand consistently.
The Novel Approach
In contrast, the methodology described in the patent utilizes protected oxazolidine or oxazoline thioesters that react under much milder conditions to achieve superior coupling efficiency. 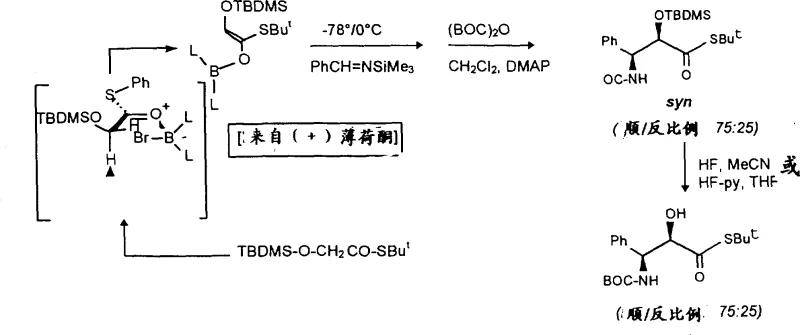 . This approach effectively shields the stereocenters during the bond-forming event, ensuring that the desired 2'(R) configuration is preserved with high enantiomeric excess. The use of thioester derivatives facilitates a nucleophilic substitution mechanism that is inherently more selective than direct acylation, significantly minimizing the formation of byproducts. Additionally, the synthetic route for the side chain itself is streamlined, often requiring only two to four steps from simple starting materials, which drastically reduces the material footprint and operational complexity. This innovation represents a paradigm shift towards more sustainable and economically feasible production of high-value taxane intermediates.
. This approach effectively shields the stereocenters during the bond-forming event, ensuring that the desired 2'(R) configuration is preserved with high enantiomeric excess. The use of thioester derivatives facilitates a nucleophilic substitution mechanism that is inherently more selective than direct acylation, significantly minimizing the formation of byproducts. Additionally, the synthetic route for the side chain itself is streamlined, often requiring only two to four steps from simple starting materials, which drastically reduces the material footprint and operational complexity. This innovation represents a paradigm shift towards more sustainable and economically feasible production of high-value taxane intermediates.
Mechanistic Insights into Oxazolidine-Mediated Coupling
The core of this technological breakthrough lies in the precise control of stereochemistry during the aldol condensation and subsequent cyclization steps that generate the side chain precursor. 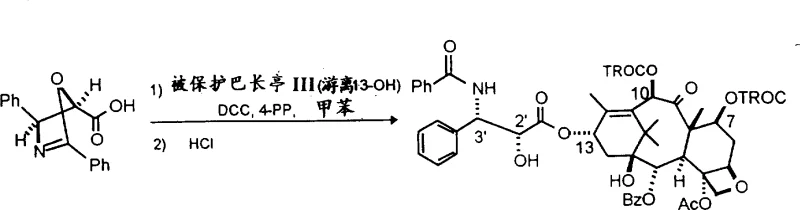 . By employing chiral boron reagents derived from menthone, the reaction dictates the spatial arrangement of atoms during the formation of the carbon-carbon bonds, leading to a predominant syn-configuration in the intermediate. This high level of diastereoselectivity is crucial because it eliminates the need for difficult separations of unwanted isomers later in the process. The transition state is stabilized by the chelation of the boron atom, which locks the conformation of the enolate and ensures that the incoming imine attacks from the less hindered face. Such mechanistic precision is rare in the synthesis of complex natural products and underscores the sophistication of this patented process.
. By employing chiral boron reagents derived from menthone, the reaction dictates the spatial arrangement of atoms during the formation of the carbon-carbon bonds, leading to a predominant syn-configuration in the intermediate. This high level of diastereoselectivity is crucial because it eliminates the need for difficult separations of unwanted isomers later in the process. The transition state is stabilized by the chelation of the boron atom, which locks the conformation of the enolate and ensures that the incoming imine attacks from the less hindered face. Such mechanistic precision is rare in the synthesis of complex natural products and underscores the sophistication of this patented process.
Furthermore, the coupling of the side chain to the baccatin nucleus proceeds through a nucleophilic attack by the C-13 hydroxyl group on the thioester carbonyl, facilitated by specific condensing agents like lithium hexamethyldisilazide. This reaction pathway avoids the formation of reactive acyl ammonium intermediates that are prone to racemization in traditional peptide coupling methods. The oxazolidine ring acts as a temporary protecting group that stabilizes the beta-hydroxy amide linkage until the final deprotection stage. This stability allows the reaction to be conducted at low temperatures, typically ranging from -78°C to 0°C, which further suppresses side reactions and degradation. The result is a clean reaction profile that yields the protected paclitaxel derivative in high purity, ready for the final global deprotection steps to reveal the active drug substance.
How to Synthesize Paclitaxel Efficiently
The standardized synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory-scale experiments to industrial production environments. The process begins with the preparation of the chiral side chain thioester, followed by its coupling with 10-deacetylbaccatin III under inert atmospheric conditions to prevent moisture sensitivity issues. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these high-yield results. . Adhering to these parameters ensures that the critical quality attributes of the intermediate, such as optical rotation and impurity profile, remain within strict specifications required for regulatory approval. This structured approach minimizes batch-to-batch variability and supports the consistent supply of materials needed for clinical and commercial formulations.
- Prepare the oxazolidine or oxazoline thioester side chain precursor using chiral boron reagents for high stereocontrol.
- React the side chain precursor with protected 10-deacetylbaccatin III in the presence of a condensing agent like LiN[Si(Me)3]2.
- Perform deprotection steps using acid treatment and zinc reduction to yield the final paclitaxel compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits related to cost stability and operational reliability. The elimination of expensive transition metal catalysts in favor of more accessible reagents significantly reduces the raw material costs associated with the manufacturing process. Cost Reduction in Manufacturing: By simplifying the purification workflow and reducing the number of synthetic steps required for the side chain, the overall production expense is substantially lowered without compromising quality. The high coupling yield means that less starting material is wasted, leading to a more efficient use of resources and a reduction in the environmental footprint of the facility. This economic efficiency translates into a more competitive pricing structure for the final API, allowing pharmaceutical companies to better manage their budget allocations for oncology portfolios.
- Cost Reduction in Manufacturing: The process eliminates the need for extensive chromatographic purification in the early stages, which is a major cost driver in fine chemical manufacturing. By relying on crystallization and simple solvent extraction, the operational expenditure related to silica gel and solvent consumption is drastically simplified. This reduction in downstream processing complexity allows for faster batch turnover and higher throughput in existing production suites. Consequently, the cost of goods sold is optimized, providing a significant margin advantage in the highly competitive generic and branded pharmaceutical markets.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as 10-deacetylbaccatin III and simple aldehydes, are readily available from established suppliers, reducing the risk of raw material shortages. The robustness of the reaction conditions means that the process is less susceptible to variations in environmental factors, ensuring consistent output regardless of seasonal changes. This reliability is critical for maintaining continuous production schedules and meeting the strict delivery timelines demanded by global drug manufacturers. A stable supply of high-quality intermediates prevents production delays and safeguards the continuity of care for patients relying on these essential medications.
- Scalability and Environmental Compliance: The use of mild reaction conditions and the avoidance of hazardous heavy metals align with modern green chemistry principles and regulatory environmental standards. Scaling this process from kilograms to metric tons is facilitated by the straightforward workup procedures, which do not require specialized equipment for handling toxic waste streams. This ease of scale-up ensures that production capacity can be rapidly expanded to meet surges in demand without significant capital investment in new infrastructure. Furthermore, the reduced generation of chemical waste simplifies compliance with environmental protection regulations, mitigating the risk of fines and operational shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in a production setting. . These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing pipelines.
Q: How does the oxazolidine method improve stereochemical control compared to DCC coupling?
A: The oxazolidine ring structure prevents epimerization at the C-2' position during the esterification process, which is a common issue with traditional DCC methods under strong conditions.
Q: What are the key advantages of using thioester intermediates in this synthesis?
A: Thioester intermediates allow for high coupling yields through nucleophilic substitution and enable simpler purification processes without the need for extensive chromatography in early stages.
Q: Is this process suitable for large-scale commercial production of paclitaxel?
A: Yes, the method utilizes readily available starting materials and mild reaction conditions, making it highly scalable for industrial manufacturing of complex anticancer agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate complex patented methodologies like CN1158126A into commercial reality for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of paclitaxel intermediate meets the highest industry standards. Our commitment to quality assurance means that you can rely on us as a long-term strategic partner for your oncology drug development programs.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements with a Customized Cost-Saving Analysis. By leveraging our manufacturing capabilities, you can secure a stable source of specific COA data and route feasibility assessments tailored to your timeline. Let us help you optimize your supply chain and accelerate the delivery of life-saving treatments to patients worldwide through our advanced semi-synthetic solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
