Scalable Production of High-Purity Schiff Base Intermediates for Pharmaceutical and Optical Applications
The landscape of functional organic materials is continuously evolving, driven by the demand for compounds that bridge the gap between optical utility and biological activity. A recent technological advancement documented in patent CN109824647B highlights a novel Schiff base derivative, specifically 4‑((E)‑phenyl(((Z)‑1‑(thiophen‑2‑yl) ethylene) hydrazino) methyl) phenol, which exhibits dual functionality as a fluorescent marker and a potential anticancer agent. This development is particularly significant for R&D directors seeking robust intermediates for drug discovery and optical sensor fabrication. The synthesis described offers a streamlined pathway that bypasses the complex multi-step sequences often associated with heterocyclic Schiff bases, presenting a compelling opportunity for industrial adoption. By leveraging a direct condensation strategy, manufacturers can achieve high structural fidelity while minimizing process complexity, a critical factor for maintaining supply chain resilience in the specialty chemical sector.
For procurement specialists and supply chain leaders, the implications of this technology extend beyond mere chemical novelty. The ability to produce high-purity pharmaceutical intermediates through a simplified workflow directly correlates with reduced operational expenditures and enhanced throughput. As global regulations tighten around solvent usage and waste disposal, the flexibility of this protocol to operate under solvent-free conditions provides a strategic advantage. This report analyzes the technical merits and commercial viability of this synthesis route, offering actionable insights for stakeholders aiming to optimize their sourcing strategies for advanced fine chemical intermediates. Understanding the mechanistic underpinnings and scalability potential is essential for integrating this material into existing production pipelines effectively.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing complex Schiff base architectures often rely on cumbersome protection-deprotection strategies or require harsh reaction conditions that compromise yield and purity. Conventional methodologies frequently necessitate the use of expensive transition metal catalysts to drive the condensation of hydrazones with ketones, introducing the risk of heavy metal contamination in the final product. For pharmaceutical applications, removing trace metal residues to meet stringent regulatory standards adds significant cost and time to the downstream purification process. Furthermore, many established protocols depend on volatile organic compounds in large volumes, creating environmental liabilities and increasing the burden on waste management systems. These inefficiencies create bottlenecks in commercial scale-up of complex polymer additives and pharmaceutical intermediates, limiting the ability of manufacturers to respond rapidly to market demand fluctuations.
The Novel Approach
In contrast, the methodology outlined in the referenced patent introduces a remarkably efficient one-step condensation process that eliminates the need for exotic catalysts or extreme thermal conditions. By utilizing readily available starting materials such as 4-hydroxybenzophenone hydrazone and 2-acetylthiophene, the reaction proceeds smoothly in common organic solvents or even via solid-state grinding. This approach drastically simplifies the operational workflow, allowing for precise control over the reaction stoichiometry without the interference of side reactions typical in metal-catalyzed systems. The resulting product crystallizes directly from the reaction mixture as yellow-green flakes, indicating a high degree of self-purification during formation. This inherent efficiency translates to substantial cost savings in fine chemical manufacturing by reducing energy consumption and minimizing the requirement for extensive chromatographic purification steps.
Mechanistic Insights into Hydrazone-Thiophene Condensation
The core of this synthesis lies in the nucleophilic attack of the hydrazine nitrogen on the carbonyl carbon of the thiophene derivative, facilitated by the electronic properties of the substituents. The presence of the hydroxyl group on the benzophenone moiety enhances the stability of the resulting imine bond through potential intramolecular hydrogen bonding interactions, which contributes to the observed thermal stability and sharp melting point of 159.6‑160.1℃. This structural rigidity is crucial for maintaining the fluorescence quantum yield, as it restricts non-radiative decay pathways that often plague flexible molecular rotors. For R&D teams, understanding this electronic interplay is vital for tuning the emission wavelengths for specific optical applications or optimizing the binding affinity for biological targets. The reaction mechanism avoids the formation of unstable intermediates that typically degrade under prolonged heating, ensuring consistent batch-to-batch reproducibility essential for GMP compliance.
Impurity control is inherently managed through the crystallization behavior of the target molecule, which favors the exclusion of unreacted starting materials and side products from the crystal lattice. The specific geometry of the thiophene ring and the planar nature of the hydrazone linkage promote tight packing in the solid state, as evidenced by the monoclinic crystal system parameters. This self-assembly property means that simple filtration and recrystallization are often sufficient to achieve the high-purity OLED material or pharmaceutical intermediate standards required by end-users. By avoiding the introduction of external catalysts, the impurity profile is significantly cleaner, consisting primarily of unreacted precursors that are easily removed by washing. This mechanistic advantage reduces the analytical burden on quality control laboratories and accelerates the release of materials for clinical or industrial testing phases.
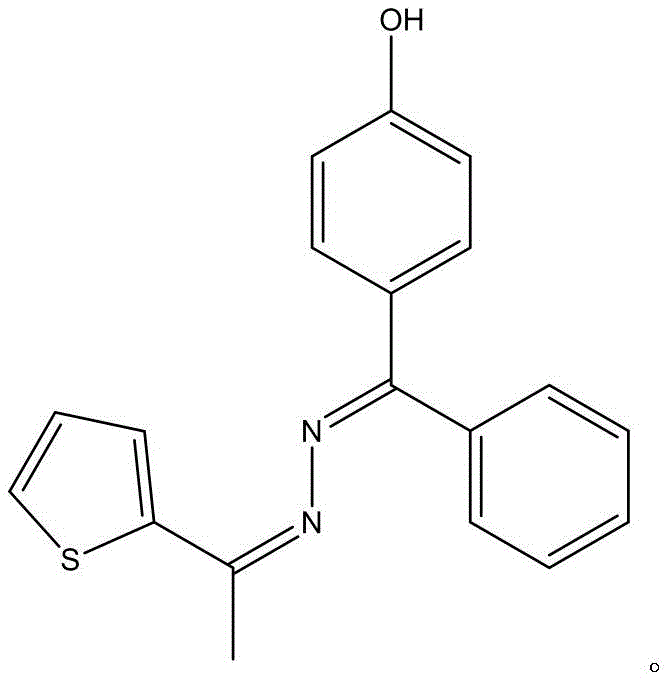
How to Synthesize 4-hydroxybenzophenone hydrazone-2-acetylthiophene Schiff Base Efficiently
Implementing this synthesis route requires careful attention to solvent selection and mixing dynamics to maximize yield and crystal quality. The process is designed to be adaptable, allowing operators to choose between solution-phase reflux for rapid kinetics or solid-phase grinding for greener processing. Detailed standard operating procedures regarding specific molar ratios and temperature gradients are critical for ensuring the formation of the desired E/Z isomeric configuration. The following guide outlines the fundamental steps derived from the patent data to assist technical teams in replicating this high-value transformation. Please refer to the standardized protocol below for execution details.
- Dissolve 4-hydroxybenzophenone hydrazone in a suitable organic solvent such as acetonitrile or ethanol under stirring.
- Add 2-acetylthiophene to the solution maintaining a molar ratio between 1: 4 and 4:1 depending on optimization requirements.
- Heat under reflux or grind at room temperature for 1 to 10 hours, then filter and recrystallize to obtain yellow-green flaky crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthesis route offers profound benefits that align with the goals of cost reduction in electronic chemical manufacturing and pharmaceutical production. The reliance on commodity chemicals as starting materials insulates the supply chain from the volatility associated with specialized reagents, ensuring consistent availability even during market disruptions. The elimination of expensive catalysts not only lowers the direct material cost but also removes the need for sophisticated metal scavenging technologies, which are often a hidden cost driver in fine chemical production. Additionally, the option to perform the reaction without solvents opens up opportunities for continuous manufacturing techniques, further enhancing throughput and reducing the facility footprint required for production. These factors collectively contribute to a more resilient and economically viable supply model for high-value intermediates.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing a catalyst-free system that removes the expense of precious metal procurement and recovery. By simplifying the workup procedure to basic filtration and volatilization, labor hours and utility consumption are drastically reduced compared to traditional multi-step syntheses. The high yield and purity obtained directly from crystallization minimize the loss of valuable material during purification, maximizing the return on raw material investment. This streamlined approach allows for competitive pricing structures without compromising on the quality specifications required for sensitive applications.
- Enhanced Supply Chain Reliability: Sourcing stability is improved as the key reactants, 4-hydroxybenzophenone hydrazone and 2-acetylthiophene, are widely produced and available from multiple global vendors. The robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality or environmental factors, ensuring consistent output. Reducing lead time for high-purity pharmaceutical intermediates is achieved through the shortened reaction timeline and simplified isolation steps, enabling faster response to urgent customer orders. This reliability is crucial for maintaining uninterrupted production schedules in downstream drug formulation or device assembly lines.
- Scalability and Environmental Compliance: The versatility of the method supports seamless scale-up from laboratory grams to multi-tonne commercial batches without requiring fundamental process redesign. The potential for solvent-free operation significantly lowers the volume of hazardous waste generated, facilitating easier compliance with increasingly strict environmental regulations. Reduced solvent usage also lowers the risk of fire and exposure hazards in the plant, improving overall operational safety profiles. These environmental and safety advantages position the manufacturer as a preferred partner for sustainability-conscious clients seeking green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this Schiff base derivative. Answers are derived directly from the experimental data and beneficial effects reported in the underlying intellectual property. This section aims to clarify uncertainties regarding scalability, purity, and application suitability for potential partners. Comprehensive technical support is available to discuss specific customization requirements for your project needs.
Q: What are the primary applications of this Schiff base compound?
A: This compound exhibits significant fluorescence properties suitable for optical materials and demonstrates in vitro antitumor activity against A549 lung cancer and Hep G2 liver cancer cells, making it valuable for pharmaceutical research.
Q: Is the synthesis process environmentally friendly?
A: Yes, the patented method allows for solid-phase reactions without organic solvents, significantly reducing chemical waste and aligning with green manufacturing standards for fine chemicals.
Q: What is the purity profile of the final crystalline product?
A: The product forms distinct yellow-green flaky crystals with a sharp melting point range of 159.6‑160.1℃, indicating high structural integrity and suitability for sensitive analytical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-hydroxybenzophenone hydrazone-2-acetylthiophene Schiff Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative laboratory chemistry into reliable industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising attributes of this fluorescent Schiff base are preserved at scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical and optical applications. Our commitment to technical excellence ensures that the transition from pilot scale to full commercialization is smooth, efficient, and fully compliant with international quality norms.
We invite you to collaborate with us to optimize your supply chain for this high-performance intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product development timeline. Let us help you secure a stable, high-quality supply of this advanced material to drive your innovation forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
