Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazol-3-one Compounds for Commercial Scale-up
Introduction: A Breakthrough in Heterocyclic Synthesis
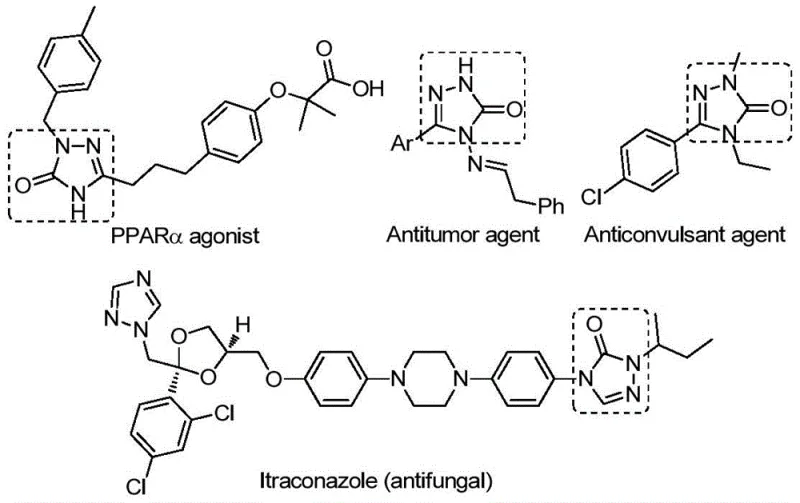
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic routes. A recent technological advancement detailed in patent CN112538054B introduces a highly versatile preparation method for 1,2,4-triazole-3-ketone compounds. These five-membered nitrogen-containing heterocycles are critical scaffolds in medicinal chemistry, renowned for their broad spectrum of biological activities including antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant properties. As illustrated in the structural diversity of bioactive molecules below, the ability to access these cores efficiently is paramount for drug discovery pipelines.
Traditionally, the synthesis of such complex heterocycles has been plagued by operational complexities and safety concerns. However, this new methodology leverages a transition metal palladium-catalyzed carbonylation tandem cyclization reaction. By utilizing cheap and readily available starting materials such as chlorinated hydrazones and sodium azide, this process not only simplifies the operational workflow but also significantly enhances reaction efficiency. For R&D directors and procurement managers alike, this represents a shift towards more robust supply chains for high-purity pharmaceutical intermediates, enabling the rapid prototyping of new drug candidates with diverse substitution patterns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthetic landscape for 1,2,4-triazol-3-one compounds was characterized by significant bottlenecks that hindered both research speed and commercial viability. Conventional literature reports typically rely on methods such as the cyclization of benzoyl hydrazide with urea under strong basic conditions (e.g., potassium hydroxide), or the tandem cyclization of hydrazides with isocyanates. These legacy approaches suffer from severe drawbacks: they often require harsh reaction conditions that limit functional group tolerance, involve complicated multi-step sequences that reduce overall atom economy, and necessitate the pre-activation of substrates which adds cost and waste. Furthermore, methods involving thioamides or acyl isocyanates frequently result in low yields and narrow substrate scopes, making them unsuitable for the diverse library synthesis required in modern drug discovery. The reliance on unstable or toxic reagents like isocyanates also poses significant safety and environmental compliance challenges for large-scale manufacturing facilities.
The Novel Approach
In stark contrast, the method disclosed in CN112538054B offers a streamlined, one-pot solution that elegantly bypasses these historical limitations. The core of this innovation lies in a palladium-catalyzed carbonylation cascade that transforms simple chlorohydrazones directly into the target triazolone core. As depicted in the general reaction scheme below, the process integrates the formation of the carbon-nitrogen bonds and the carbonyl insertion in a single operational sequence. This approach eliminates the need for pre-formed isocyanates or harsh basic cyclization conditions. Instead, it employs a safe solid CO surrogate (TFBen) and sodium azide, allowing for the synthesis of variously substituted 1,2,4-triazole-3-ketones with high efficiency. The reaction demonstrates exceptional substrate compatibility, tolerating a wide range of functional groups including halogens, alkyls, and aryls, thereby providing medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies without the burden of complex protecting group strategies.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogues. The reaction is believed to proceed through a sophisticated catalytic cycle initiated by the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate (Structure II). This forms a key divalent palladium intermediate. Simultaneously, the CO surrogate, TFBen (1,3,5-tricarboxylic acid phenol ester), undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, generating an acyl-palladium species. Subsequently, the acyl-palladium intermediate reacts with sodium azide to form an acyl azide compound. This unstable intermediate rapidly undergoes a Curtius rearrangement to generate an isocyanate intermediate in situ, which immediately participates in an intramolecular nucleophilic addition with the adjacent hydrazine nitrogen. This final cyclization step closes the ring to yield the thermodynamically stable 1,2,4-triazole-3-one product (Structure I). The elegance of this mechanism lies in its tandem nature, where multiple bond-forming events occur sequentially without isolating reactive intermediates.

From an impurity control perspective, this mechanism offers distinct advantages. The use of a solid CO source like TFBen avoids the handling of gaseous CO, which can lead to inconsistent pressure and side reactions in traditional carbonylations. Furthermore, the intramolecular nature of the final cyclization step ensures high regioselectivity, minimizing the formation of isomeric byproducts that are common in intermolecular condensations. The choice of ligand, specifically Xantphos, plays a critical role in stabilizing the palladium center and facilitating the reductive elimination steps, ensuring high turnover numbers. The protocol specifies a molar ratio of Pd2(dba)3 to Xantphos of 1:2, which is optimized to maintain catalyst activity throughout the 16 to 30-hour reaction window at 100-120°C. This precise control over the catalytic environment allows for the synthesis of complex derivatives, such as those shown below (I-1 to I-5), with high purity and minimal downstream purification burden.

How to Synthesize 1,2,4-Triazol-3-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and safety. The patent outlines a robust protocol where the chlorohydrazone, sodium azide, palladium catalyst, ligand, and CO source are combined in an aprotic solvent such as 1,4-dioxane. The reaction is heated to 100°C for approximately 24 hours. Detailed standard operating procedures regarding stoichiometry, solvent volume, and workup techniques are essential for reproducibility. For a comprehensive guide on executing this synthesis with optimal results, please refer to the standardized protocol below.
- Charge a reaction vessel with Pd2(dba)3 catalyst, Xantphos ligand, TFBen (CO source), chlorohydrazone substrate, and sodium azide in an aprotic solvent like 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, perform silica gel treatment, and purify the crude product via column chromatography to isolate the high-purity 1,2,4-triazol-3-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond mere chemical curiosity. The shift from multi-step, hazardous legacy processes to this streamlined one-pot carbonylation addresses several critical pain points in the sourcing of pharmaceutical intermediates. By simplifying the synthetic pathway, manufacturers can reduce the number of unit operations, which directly correlates to lower capital expenditure and reduced operational complexity. The use of commodity chemicals like sodium azide and chlorohydrazones, which are widely available from global chemical suppliers, mitigates the risk of raw material shortages that often plague specialized reagent supply chains. Furthermore, the elimination of harsh reagents and the use of a solid CO surrogate enhance workplace safety and simplify regulatory compliance, reducing the administrative burden associated with handling toxic gases or corrosive bases.
- Cost Reduction in Manufacturing: The economic implications of this technology are profound. By consolidating multiple synthetic steps into a single pot, the process drastically reduces solvent consumption, energy usage, and labor costs associated with intermediate isolation and purification. The avoidance of expensive and unstable reagents like isocyanates further drives down the bill of materials. Additionally, the high reaction efficiency and broad substrate scope mean that fewer batches are rejected due to poor yield or purity issues, leading to substantial cost savings in quality control and waste disposal. The ability to use standard stainless steel reactors without the need for high-pressure CO equipment also lowers the barrier to entry for contract manufacturing organizations.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on stable, shelf-stable starting materials. Unlike gaseous carbon monoxide, which requires specialized logistics and storage infrastructure, TFBen is a solid that can be easily transported and stored. This flexibility allows for more agile inventory management and reduces the lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions (100-120°C) ensures consistent output even with minor variations in utility supply, guaranteeing on-time delivery to downstream API manufacturers. This reliability is crucial for maintaining continuous production schedules in the fast-paced pharmaceutical industry.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a greener alternative to traditional synthesis. The atom economy is improved by avoiding the generation of stoichiometric amounts of salt waste typical in base-mediated cyclizations. The simplified workup procedure, involving filtration and chromatography, minimizes the volume of aqueous waste streams. As regulatory pressures on pharmaceutical manufacturing intensify, adopting processes with a lower environmental footprint is not just a compliance requirement but a competitive advantage. The scalability of this reaction, demonstrated from milligram to gram scales in the patent data, suggests a clear path to multi-ton commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that require harsh conditions, pre-activated substrates, or toxic reagents like isocyanates, this novel approach utilizes cheap chlorohydrazones and sodium azide under mild conditions (100-120°C) with excellent functional group tolerance and higher yields.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability. The use of commercially available reagents, simple post-treatment (filtration and chromatography), and robust reaction conditions make it highly amenable to commercial scale-up from gram to multi-ton levels.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a safe and effective carbon monoxide (CO) substitute. Under heating conditions, it releases CO in situ, which inserts into the carbon-palladium bond to form the necessary acyl-palladium intermediate without requiring high-pressure CO gas cylinders.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazol-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the Pd-catalyzed carbonylation described in CN112538054B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle complex organometallic reactions with stringent purity specifications, supported by rigorous QC labs that guarantee the quality of every batch. We understand that in the competitive landscape of API manufacturing, consistency and compliance are non-negotiable, and our processes are designed to meet the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with reliable, high-quality intermediates produced via the most efficient chemistry available.
