Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazol-3-one Compounds for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazol-3-one Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-rich heterocycles, particularly those with proven biological significance. Patent CN112538054B introduces a groundbreaking preparation method for 1,2,4-triazole-3-ketone compounds, a privileged scaffold found in numerous bioactive molecules ranging from antifungal agents like Itraconazole to potent antitumor and anticonvulsant drugs. As depicted in the structural diversity shown below, these cores are critical for modern drug discovery. This patent details a transition metal palladium-catalyzed carbonylation tandem cyclization reaction that utilizes inexpensive chlorohydrazones and sodium azide as starting materials. By leveraging a sophisticated catalytic system involving Pd2(dba)3 and Xantphos, this technology offers a streamlined pathway that addresses long-standing challenges in yield, operational simplicity, and substrate compatibility, positioning it as a vital asset for any reliable pharmaceutical intermediate supplier.
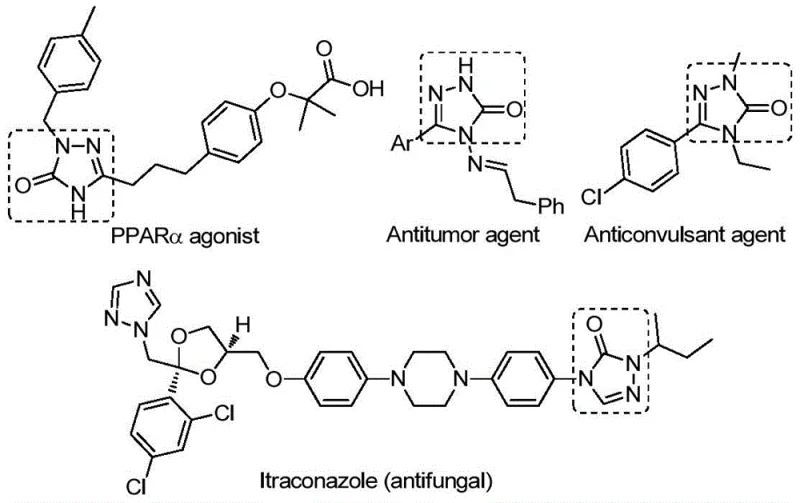
The strategic importance of this synthesis lies in its ability to access complex heterocyclic architectures efficiently. The 1,2,4-triazol-3-one motif is not merely a synthetic curiosity but a cornerstone of medicinal chemistry, appearing in tyrosinase inhibitors, CB receptor modulators, and NK1 antagonists. The ability to synthesize these structures with high purity and minimal impurity profiles is paramount for R&D directors focused on accelerating lead optimization. This patent provides a versatile platform where various substituents (R1 and R2) can be introduced, allowing for the rapid generation of analog libraries. For procurement managers and supply chain heads, the implication is clear: a method that relies on commodity chemicals rather than exotic, unstable reagents translates directly to cost reduction in API manufacturing and reduced risk of supply chain disruption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazol-3-one ring has been fraught with synthetic inefficiencies that hinder large-scale production. Traditional literature reports describe methods such as the cyclization of benzoyl hydrazide with urea under strong basic conditions, or the tandem reaction of hydrazides with isocyanates. These legacy processes suffer from severe drawbacks, including the requirement for harsh reaction conditions that can degrade sensitive functional groups, multi-step sequences that lower overall throughput, and the necessity for pre-activation of substrates which adds cost and waste. Furthermore, methods involving thioamides often require high temperatures that limit substrate scope, while reactions with acyl isocyanates pose significant safety hazards due to the toxicity and volatility of the reagents. Consequently, these conventional routes often result in low yields and narrow substrate tolerance, making them unsuitable for the rigorous demands of modern commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112538054B represents a paradigm shift towards green and efficient synthesis. By employing a palladium-catalyzed carbonylation strategy, this novel approach bypasses the need for hazardous gaseous carbon monoxide or toxic isocyanates. Instead, it utilizes TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid, safe, and easy-to-handle carbon monoxide substitute that releases CO in situ under thermal conditions. The reaction couples readily available chlorohydrazones with sodium azide in a one-pot tandem process. This not only simplifies the operational workflow but also dramatically improves atom economy. The use of a robust Pd2(dba)3/Xantphos catalyst system ensures high turnover and excellent functional group tolerance, allowing for the synthesis of diverse derivatives substituted with alkyl, aryl, halogen, or alkoxy groups. This flexibility is crucial for developing high-purity OLED material precursors or specialized agrochemical intermediates where specific substitution patterns are required.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is essential for R&D teams aiming to optimize the process further. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the TFBen additive undergoes thermal decomposition to release carbon monoxide within the reaction medium. This in situ generated CO then undergoes migratory insertion into the carbon-palladium bond, forming a key acyl-palladium species. This step is critical as it installs the carbonyl functionality required for the triazolone ring without external gas cylinders. Subsequently, the acyl-palladium intermediate reacts with sodium azide to generate an acyl azide compound, which spontaneously undergoes a Curtius rearrangement to yield an isocyanate intermediate. Finally, an intramolecular nucleophilic addition of the hydrazine nitrogen to the isocyanate carbon closes the ring, delivering the final 1,2,4-triazol-3-one product with high regioselectivity.

From an impurity control perspective, this mechanism offers distinct advantages over traditional routes. The tandem nature of the reaction minimizes the accumulation of stable intermediates that could otherwise become difficult-to-remove impurities. The use of Xantphos, a bidentate ligand with a large bite angle, stabilizes the palladium center and prevents the formation of palladium black, ensuring consistent catalytic activity throughout the 16 to 30-hour reaction window. Furthermore, the choice of aprotic solvents like 1,4-dioxane is pivotal; protic solvents or polar aprotic solvents like DMF were found to inhibit the reaction or lower efficiency. By strictly controlling the solvent environment and maintaining the temperature between 100°C and 120°C, the process ensures that side reactions such as hydrolysis of the acyl azide are minimized. This precise control over the reaction parameters results in a cleaner crude profile, reducing the burden on downstream purification and enhancing the overall yield of high-purity pharmaceutical intermediates.
How to Synthesize 1,2,4-Triazol-3-one Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The procedure involves charging a reaction vessel, such as a Schlenk tube, with the palladium catalyst, ligand, TFBen, chlorohydrazone, and sodium azide in an organic solvent. The mixture is then heated to facilitate the tandem cyclization. The beauty of this protocol lies in its simplicity: it avoids the need for high-pressure equipment typically associated with carbonylation reactions, as the CO source is solid and released gently upon heating. Following the reaction, the workup is straightforward, involving filtration to remove inorganic salts and catalyst residues, followed by standard silica gel chromatography. This operational simplicity makes it an attractive candidate for reducing lead time for high-purity pharmaceutical intermediates, as it requires minimal specialized training for operators and fits into existing infrastructure.
- Charge a reaction vessel with Pd2(dba)3 catalyst, Xantphos ligand, TFBen (CO source), chlorohydrazone substrate, and sodium azide in an aprotic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to allow for the carbonylation tandem cyclization to proceed to completion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the high-purity 1,2,4-triazol-3-one target.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers tangible strategic benefits beyond mere chemical elegance. The primary driver for cost optimization here is the replacement of expensive, hazardous, or difficult-to-source reagents with commodity chemicals. Sodium azide and chlorohydrazones are widely available in the global market at competitive price points, unlike specialized isocyanates or gaseous CO which require stringent safety protocols and specialized logistics. By eliminating the need for high-pressure CO reactors, the capital expenditure (CAPEX) for setting up production lines is significantly reduced. Furthermore, the high reaction efficiency and broad substrate scope mean that fewer batches are rejected due to poor yield or purity issues, leading to substantial cost savings in raw material consumption and waste disposal. This efficiency directly translates to a more competitive pricing structure for the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic viability of this process is anchored in the use of TFBen as a surrogate for carbon monoxide gas. Handling gaseous CO requires expensive mass flow controllers, leak detection systems, and specialized high-pressure autoclaves, all of which drive up operational costs. By switching to a solid CO source, the process eliminates these capital and operational expenditures. Additionally, the catalyst loading is relatively low (2.5 mol%), and the ligand Xantphos, while specialized, is used in stoichiometric amounts that are manageable at scale. The elimination of pre-activation steps for substrates further reduces labor and utility costs, creating a leaner manufacturing process that enhances margin potential for high-value fine chemicals.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical in the post-pandemic era, and this synthesis route bolsters security of supply. The key reagents—sodium azide, palladium catalysts, and simple aromatic chlorohydrazones—are produced by multiple vendors globally, reducing the risk of single-source bottlenecks. Unlike methods relying on unstable intermediates that must be synthesized immediately prior to use, the starting materials here are shelf-stable solids that can be stocked in bulk. This inventory flexibility allows manufacturers to respond rapidly to surges in demand for antiviral or antifungal APIs without waiting for complex precursor synthesis. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in utility quality, ensuring consistent output and reliable delivery schedules for downstream partners.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental and safety challenges, but this methodology is inherently designed for expansion. The absence of toxic gas feeds simplifies the permitting process for new production facilities, as the environmental footprint regarding volatile organic compounds (VOCs) and hazardous air pollutants is lower. The reaction generates nitrogen gas as a benign byproduct during the azide decomposition, avoiding the formation of noxious sulfur-containing wastes common in thioamide-based routes. Moreover, the high conversion rates minimize the volume of solvent required for purification, aligning with green chemistry principles. This environmental compatibility facilitates smoother regulatory approvals and supports corporate sustainability goals, making it an ideal choice for the commercial scale-up of complex polymer additives or specialty chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the operational feasibility and strategic value for potential licensees or manufacturing partners. Understanding these nuances is key to evaluating the technology for integration into existing production portfolios.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that require harsh conditions, pre-activation of substrates, or toxic isocyanates, this novel approach utilizes cheap and readily available chlorohydrazones and sodium azide. It operates under milder conditions with a broader substrate scope and significantly higher reaction efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability. The use of stable solid reagents like TFBen as a carbon monoxide substitute eliminates the need for handling hazardous CO gas, making the process safer and more amenable to commercial scale-up in standard reactor setups.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a safe and effective carbon monoxide substitute. Under heating conditions, it releases CO in situ, which inserts into the carbon-palladium bond to form the necessary acyl-palladium intermediate for the subsequent cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazol-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just a recipe; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the laboratory are faithfully reproduced in the plant. We understand the critical nature of stringent purity specifications in the pharmaceutical sector, which is why our rigorous QC labs employ advanced analytical techniques to monitor every batch for trace impurities and residual metals. Whether you are developing a novel antifungal agent or a targeted oncology therapy, our infrastructure is ready to support your journey from clinical trial material to full commercial supply.
We invite you to leverage our technical capabilities to optimize your supply chain for 1,2,4-triazol-3-one derivatives. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us help you secure a competitive edge through superior chemistry and reliable manufacturing execution.
