Advanced One-Pot Synthesis of Quinolone Carboxylic Acid for Commercial Pharma Manufacturing
Introduction to Novel Quinolone Synthesis Technology
The pharmaceutical industry continuously seeks robust methodologies for producing critical antibiotic intermediates, and Patent CN1030911A presents a transformative approach to synthesizing quinolone carboxylic acid derivatives. This intellectual property discloses a highly efficient one-pot reaction strategy that consolidates multiple synthetic transformations into a single continuous process, thereby eliminating the need for isolating unstable or sensitive intermediate species. By integrating acylation, amine exchange, cyclization, and hydrolysis into a unified workflow, this technology addresses longstanding inefficiencies in the manufacturing of 4-oxo-3-quinoline-carboxylic acid scaffolds. The method specifically targets the production of compounds where R1 represents cyclopropyl groups, which are pivotal structural motifs in second-generation fluoroquinolone antibiotics. For R&D directors and process chemists, this patent offers a compelling alternative to traditional multi-step routes that often suffer from cumulative yield losses and excessive solvent usage. The ability to generate high-purity intermediates through a telescoped sequence represents a significant leap forward in process intensification, aligning perfectly with modern green chemistry principles and cost-effective manufacturing goals.
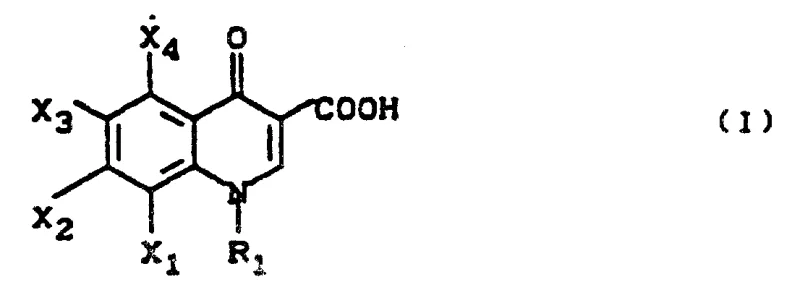
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinolone carboxylic acids typically involve discrete, isolated steps that require extensive purification between each transformation. In conventional processes, the acylation of enamines followed by cyclization often necessitates the isolation of the acylated intermediate, which can be thermally unstable or prone to hydrolysis upon exposure to ambient conditions. This fragmentation of the synthesis leads to substantial material loss during transfer, filtration, and drying operations, significantly impacting the overall mass balance and economic viability of the process. Furthermore, the requirement for multiple solvent exchanges and drying cycles increases the operational footprint, demanding larger reactor volumes and extended production timelines. The accumulation of impurities at each isolation stage complicates the final purification, often requiring recrystallization or chromatography that further erodes yield. From a supply chain perspective, these discontinuous processes introduce variability and potential bottlenecks, making it difficult to guarantee consistent batch-to-batch quality and timely delivery for large-scale API production.
The Novel Approach
In stark contrast, the methodology described in CN1030911A utilizes a telescoped one-pot reaction design that maintains the reaction mixture in a continuous fluid state throughout the critical transformation phases. By avoiding the isolation of the intermediate acylated enamine (Formula IV), the process minimizes exposure to degradative conditions and prevents the loss of material associated with solid handling. The reaction proceeds seamlessly from the initial acylation of dimethylamino acrylate to the subsequent amine exchange with cyclopropylamine, all within the same reactor vessel. This continuity allows for the immediate utilization of the reactive intermediate, driving the equilibrium towards the desired cyclized product with higher efficiency. The elimination of intermediate work-ups not only reduces the consumption of auxiliary materials like filtration aids and drying agents but also significantly lowers the volume of waste solvent generated. For procurement managers, this streamlined approach translates directly into reduced operational expenditures and a more sustainable manufacturing profile, as the simplified workflow requires less energy input and fewer unit operations to achieve the final target molecule.
Mechanistic Insights into One-Pot Telescoped Cyclization
The core of this innovative synthesis lies in the precise control of reaction conditions that allow distinct chemical transformations to occur sequentially without interference. The process initiates with the acylation of a dimethylamino acrylate derivative (Formula III) using a substituted benzoyl chloride (Formula II) in the presence of a tertiary amine base. This step generates the key enaminone intermediate (Formula IV) in situ, which serves as the precursor for the subsequent ring closure. The choice of solvent, such as toluene or xylene, and the temperature range of 50-150°C are critical for ensuring complete conversion while managing the exothermic nature of the acylation. Following acylation, the reaction mixture undergoes an amine exchange where cyclopropylamine displaces the dimethylamino group. This nucleophilic substitution is facilitated by heating the mixture to 50-120°C, driving off dimethylamine gas and shifting the equilibrium towards the formation of the cyclopropyl-substituted enaminone. The seamless transition between these steps is crucial for maintaining the integrity of the reactive species.
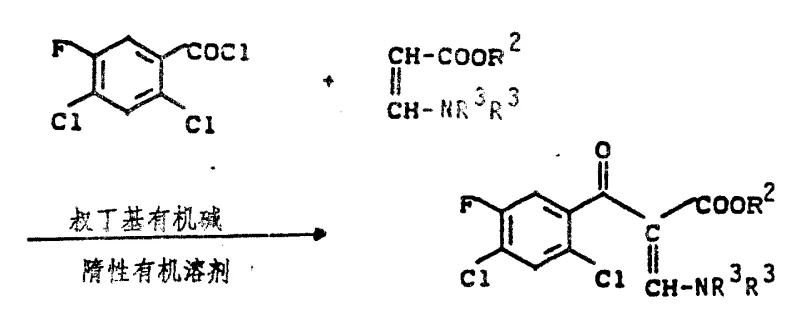
Following the amine exchange, the reaction mixture is subjected to cyclization conditions involving a strong inorganic base such as potassium carbonate or sodium hydride in a high-boiling solvent like butyl glycol. Heating the mixture to temperatures between 80-180°C promotes the intramolecular nucleophilic attack of the amine nitrogen onto the ester carbonyl, closing the quinolone ring system. This cyclization step is immediately followed by hydrolysis, where the addition of water and subsequent acidification precipitates the final quinolone carboxylic acid product. The ability to perform hydrolysis in the same pot without isolating the cyclic ester intermediate is a testament to the robustness of this chemical design. Impurity control is inherently managed by the continuous nature of the process; side products formed in early stages are either consumed in subsequent steps or remain soluble in the mother liquor during the final precipitation. This mechanistic elegance ensures that the final product meets stringent purity specifications required for pharmaceutical applications, minimizing the need for extensive downstream purification.
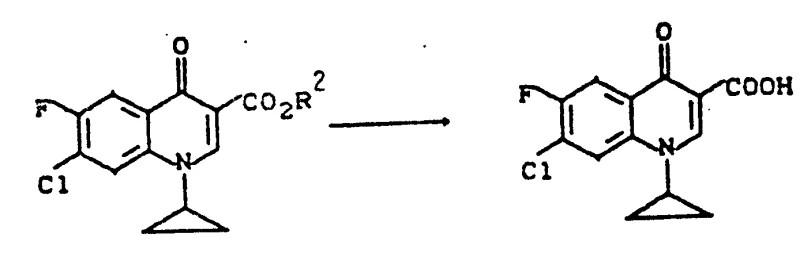
How to Synthesize Quinolone Carboxylic Acid Efficiently
The implementation of this one-pot synthesis requires careful attention to reagent addition rates and temperature profiles to maximize yield and safety. The process begins with the preparation of the acylation mixture, followed by the direct addition of the amine source without cooling or filtering the intermediate. Detailed operational parameters, including specific molar ratios of base to acid chloride and precise heating ramps for the cyclization phase, are essential for reproducibility. The patent provides multiple embodiments demonstrating the versatility of this method across different solvent systems and base choices, offering flexibility for scale-up. For process engineers looking to adopt this technology, understanding the thermal profile of the exothermic acylation and the gas evolution during amine exchange is vital for reactor design. The standardized protocol outlined in the patent serves as a robust foundation for developing a GMP-compliant manufacturing process that balances efficiency with safety.
- Perform acylation of dimethylamino acrylate with substituted benzoyl chloride in the presence of a tertiary amine base at 50-150°C.
- Conduct amine exchange by adding cyclopropylamine directly to the reaction mixture at 50-120°C to replace the dimethylamino group.
- Execute cyclization using an inorganic base in a high-boiling solvent at 80-180°C, followed by hydrolysis and acid precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this one-pot synthesis methodology offers profound commercial benefits that extend beyond simple yield improvements, fundamentally altering the cost structure of quinolone intermediate manufacturing. By collapsing multiple synthetic steps into a single reactor charge, the process drastically reduces the capital expenditure required for equipment, as fewer vessels are needed for reaction, isolation, and drying. This consolidation also leads to a significant reduction in labor costs and utility consumption, as the extended processing times associated with intermediate work-ups are eliminated. For procurement managers, the simplified supply chain means fewer raw materials are required for auxiliary processes, such as filtration media and drying agents, leading to direct cost savings. Furthermore, the reduced solvent usage aligns with increasingly strict environmental regulations, potentially lowering waste disposal fees and mitigating regulatory risks associated with volatile organic compound emissions. The overall effect is a leaner, more agile manufacturing operation capable of responding quickly to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for expensive filtration and drying equipment, directly lowering the fixed and variable costs associated with production. By avoiding the loss of material during transfer and purification of intermediates, the overall mass efficiency of the process is improved, meaning less starting material is required to produce the same amount of final product. Additionally, the ability to recover and recycle high-boiling solvents like butyl glycol further enhances the economic viability of the process. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), providing a competitive edge in the pricing of generic antibiotic intermediates.
- Enhanced Supply Chain Reliability: A simplified process with fewer unit operations inherently reduces the number of potential failure points in the manufacturing chain. The risk of batch rejection due to intermediate specification failures is minimized, as the crude intermediate is not isolated and tested but rather consumed immediately in the next step. This continuity ensures a more predictable production schedule, allowing supply chain heads to commit to tighter delivery windows with greater confidence. Moreover, the use of common, commercially available reagents such as cyclopropylamine and substituted benzoyl chlorides ensures that raw material sourcing remains stable and不受 geopolitical disruptions. The robustness of the chemistry supports consistent output, fostering stronger relationships with downstream API manufacturers.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis is inherently scalable, as it avoids the handling of large quantities of potentially hazardous dry intermediates. The process generates less solid waste and wastewater compared to traditional multi-step routes, simplifying effluent treatment and reducing the environmental footprint of the facility. The ability to operate at atmospheric pressure in standard glass-lined reactors facilitates easy technology transfer from pilot plant to commercial scale. This scalability ensures that production can be ramped up rapidly to meet surges in demand for fluoroquinolone antibiotics without requiring major infrastructure investments. Compliance with green chemistry metrics is improved, enhancing the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented one-pot synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance to industrial application. These insights help clarify the operational boundaries and critical success factors for achieving high-quality output consistently.
Q: What are the primary advantages of the one-pot synthesis method for quinolone derivatives?
A: The primary advantage is the elimination of intermediate isolation steps, which drastically reduces solvent consumption, processing time, and waste generation while maintaining high yields.
Q: Which solvents are compatible with the cyclization step of this process?
A: High-boiling solvents such as butyl glycol, ethylene glycol, DMF, DMSO, and N-Methyl pyrrolidone are suitable for facilitating the cyclization reaction at elevated temperatures.
Q: How is the final product purified in this patented method?
A: The final quinolone carboxylic acid is purified through precipitation by adding mineral or organic acids to the hydrolyzed reaction mixture, followed by filtration and washing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolone Carboxylic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to translate complex patent methodologies like CN1030911A into robust commercial realities. Our team of seasoned process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. We understand the critical importance of maintaining stringent purity specifications for pharmaceutical intermediates, and our rigorous QC labs are equipped to monitor every stage of the synthesis to guarantee product integrity. By leveraging our state-of-the-art facilities and deep knowledge of heterocyclic chemistry, we can optimize this one-pot process to maximize yield and minimize waste, delivering a cost-effective solution for your supply chain needs.
We invite you to collaborate with us to explore how this advanced synthesis route can enhance your product portfolio and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to reach out to request specific COA data and route feasibility assessments that demonstrate our capability to deliver high-purity quinolone derivatives reliably. Partnering with NINGBO INNO PHARMCHEM means gaining access to a supply chain partner committed to innovation, quality, and long-term value creation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
