Revolutionizing Planar Chiral Metallocene Synthesis for Commercial Scale-up and High-Purity Applications
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly enantiopure compounds in the pharmaceutical and fine chemical sectors. Patent CN114560893A introduces a groundbreaking preparation method for planar chiral metallocene compounds, addressing long-standing challenges in stereocontrol and synthetic efficiency. This technology leverages a sophisticated rhodium-catalyzed kinetic resolution strategy, utilizing easily obtained chiral phosphine ligands to transform racemic substrates into valuable planar chiral architectures. For R&D directors and procurement specialists, this represents a significant shift from traditional stoichiometric methods to more sustainable, catalytic processes. The ability to synthesize these complex scaffolds with high regioselectivity and enantioselectivity opens new avenues for developing advanced chiral ligands, which are indispensable tools in modern drug discovery and material science applications.
Furthermore, the versatility of this synthetic route allows for the incorporation of diverse functional groups, enhancing the utility of the resulting metallocene derivatives across various chemical domains. By establishing a robust pathway for creating these challenging structures, the patent provides a foundation for optimizing supply chains dependent on high-performance chiral intermediates. The methodology not only improves the purity profile of the final products but also streamlines the overall production workflow, making it an attractive option for manufacturers seeking to enhance their competitive edge in the global market for specialty chemicals and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of planar chiral metallocene compounds has been fraught with significant technical hurdles that impede large-scale adoption and cost-effective manufacturing. Traditional strategies often rely on the use of chiral prosthetic groups or equivalent amounts of chiral bases, which inherently limit the atom economy and generate substantial chemical waste. These methods frequently require harsh reaction conditions, including the use of air-sensitive metal reagents that demand specialized handling infrastructure and increase operational risks. Moreover, the substrate universality of conventional approaches is often poor, restricting the range of substituents that can be introduced onto the metallocene scaffold without compromising stereochemical integrity. Such limitations result in prolonged development timelines and inflated production costs, posing serious challenges for procurement managers aiming to secure reliable sources of high-quality intermediates.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent employs an asymmetric catalytic carbon-hydrogen bond activation method that dramatically simplifies the synthetic landscape. By utilizing a kinetic resolution strategy with a rhodium catalyst and chiral phosphine ligands, this method efficiently differentiates between enantiomers of the racemic starting material. This catalytic cycle allows for the direct arylation of the metallocene core under mild conditions, typically in organic solvents like tetrahydrofuran or toluene at moderate temperatures. 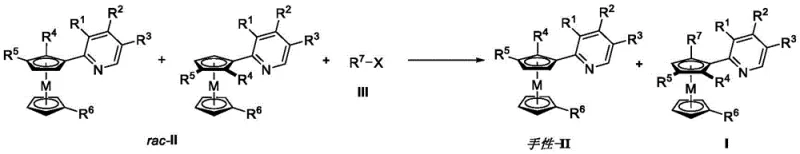 The process not only yields the desired planar chiral product with high selectivity but also enables the recovery of the unreacted enantiomer of the starting material, effectively doubling the utility of the raw materials. This dual-output capability significantly enhances resource efficiency and reduces the overall cost burden associated with raw material consumption, offering a compelling value proposition for supply chain optimization.
The process not only yields the desired planar chiral product with high selectivity but also enables the recovery of the unreacted enantiomer of the starting material, effectively doubling the utility of the raw materials. This dual-output capability significantly enhances resource efficiency and reduces the overall cost burden associated with raw material consumption, offering a compelling value proposition for supply chain optimization.
Mechanistic Insights into Rhodium-Catalyzed Kinetic Resolution
At the heart of this technological advancement lies a meticulously engineered catalytic cycle that ensures precise stereocontrol during the bond-forming event. The rhodium catalyst, activated by the chiral phosphine ligand, coordinates with the racemic metallocene substrate to form a transient intermediate complex. The chiral environment created by ligands such as Taddol-derived phosphites or Feringa ligands dictates the spatial orientation of the incoming arylating agent, thereby favoring the formation of one planar chiral isomer over the other. This discrimination is crucial for achieving high enantiomeric excess (ee) values, which are often reported to exceed 90% in specific embodiments described within the patent documentation. The mechanistic pathway avoids the formation of unwanted by-products that typically complicate downstream purification processes, thus ensuring a cleaner impurity profile for the final active pharmaceutical ingredients.
Moreover, the reaction conditions are optimized to maintain catalyst stability and activity throughout the transformation. The use of alkali bases, such as lithium tert-butoxide, facilitates the deprotonation steps necessary for the catalytic turnover without degrading the sensitive organometallic species. 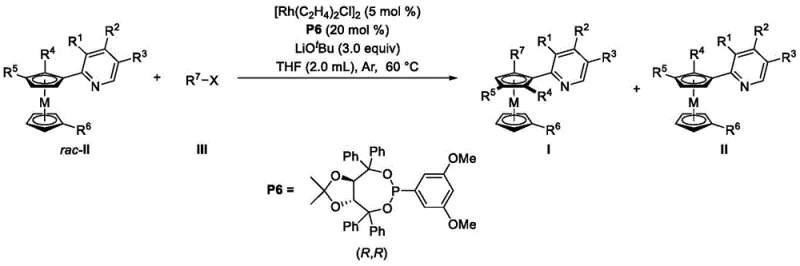 Detailed analysis of the reaction parameters reveals that maintaining a molar concentration of the substrate between 0.1 to 0.2 mol/L and a catalyst loading of approximately 5 mol% strikes an optimal balance between reaction rate and cost efficiency. This level of control over the reaction kinetics is essential for R&D teams focused on scaling up processes from laboratory benchtop to pilot plant operations, ensuring that the high selectivity observed in small-scale experiments translates reliably to commercial production volumes.
Detailed analysis of the reaction parameters reveals that maintaining a molar concentration of the substrate between 0.1 to 0.2 mol/L and a catalyst loading of approximately 5 mol% strikes an optimal balance between reaction rate and cost efficiency. This level of control over the reaction kinetics is essential for R&D teams focused on scaling up processes from laboratory benchtop to pilot plant operations, ensuring that the high selectivity observed in small-scale experiments translates reliably to commercial production volumes.
How to Synthesize Planar Chiral Metallocene Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding atmospheric control and reagent quality to maximize yield and stereoselectivity. The detailed standardized synthesis steps involve precise measurement of the rhodium catalyst and chiral ligand ratios, followed by careful monitoring of the reaction progress via analytical techniques such as HPLC or TLC. The process concludes with a workup procedure that includes dilution, separation, and purification via column chromatography to isolate the target compound with the required purity specifications.
- Prepare the reaction system under a protective gas atmosphere using an organic solvent such as tetrahydrofuran or toluene.
- Add the racemic metallocene substrate, halogenated aromatic coupling partner, rhodium catalyst, and chiral phosphine ligand.
- Introduce the alkali base and maintain the reaction temperature between 50 to 70 degrees Celsius to achieve high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers transformative benefits that directly address the pain points of procurement managers and supply chain heads. The shift from stoichiometric chiral auxiliaries to a catalytic system fundamentally alters the cost structure of manufacturing these high-value intermediates. By eliminating the need for expensive, single-use chiral reagents, the process achieves substantial cost savings in raw material procurement. Furthermore, the ability to recover and recycle the unreacted enantiomer of the starting material minimizes waste disposal costs and maximizes the return on investment for every kilogram of substrate purchased. This efficiency gain is critical for maintaining competitive pricing in the volatile market for specialty fine chemicals.
- Cost Reduction in Manufacturing: The catalytic nature of the reaction significantly lowers the consumption of high-cost chiral ligands relative to the product output. Unlike traditional methods that require equivalent amounts of chiral inducers, this approach uses sub-stoichiometric quantities of the catalyst system, leading to a drastic reduction in the bill of materials. Additionally, the mild reaction temperatures reduce energy consumption requirements for heating and cooling systems, further contributing to lower operational expenditures. The streamlined purification process also reduces solvent usage and labor hours associated with isolation, compounding the overall economic benefits for large-scale production facilities.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials and common organic solvents mitigates the risk of supply disruptions often associated with exotic or proprietary reagents. Since the key components such as rhodium catalysts and phosphine ligands can be sourced from established chemical suppliers, procurement teams can build more resilient supply networks. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification products. This reliability is paramount for pharmaceutical companies that require uninterrupted access to critical intermediates to meet regulatory filing deadlines and market demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations and safety protocols familiar to industrial chemists. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, simplifying the compliance burden for manufacturing sites. The ability to operate at moderate temperatures and pressures reduces the engineering complexity required for scale-up, allowing for faster technology transfer from R&D to production. This ease of scale-up ensures that supply chain heads can confidently plan for increased capacity without encountering unforeseen technical bottlenecks or safety hazards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of adopting this method. Understanding these details is essential for stakeholders evaluating the integration of this process into their existing manufacturing portfolios.
Q: What are the advantages of this rhodium-catalyzed method over traditional synthesis?
A: This method utilizes a kinetic resolution strategy with chiral phosphine ligands, avoiding the need for stoichiometric chiral auxiliaries and enabling the recovery of optically pure starting materials.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the reaction conditions involve commercially available solvents and moderate temperatures, facilitating commercial scale-up of complex organometallic intermediates.
Q: What level of enantioselectivity can be achieved with this technology?
A: The patent data demonstrates high enantioselectivity, often exceeding 90% ee, which is critical for producing high-purity chiral ligands and active pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Planar Chiral Metallocene Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthesis technology to deliver superior planar chiral metallocene compounds to our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by the pharmaceutical and agrochemical sectors. Our commitment to quality assurance means that clients can trust us to provide consistent, high-performance intermediates that drive their own product development success.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this cutting-edge technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your strategic goals. Let us help you navigate the complexities of chiral synthesis and secure a competitive advantage in your market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
