Advanced Atmospheric Pressure Synthesis of Roxadustat Intermediates for Commercial Scale-Up
Introduction to Novel Isoquinolinone Manufacturing Technology
The pharmaceutical industry is constantly seeking more efficient and safer pathways for synthesizing complex heterocyclic intermediates, particularly for hypoxia-inducible factor prolyl hydroxylase inhibitors like Roxadustat. Patent CN112679431B introduces a groundbreaking method for preparing isoquinolinone compounds that fundamentally shifts the paradigm from high-pressure hydrogenation to atmospheric active metal reduction. This technology addresses critical bottlenecks in the existing supply chain by eliminating the need for expensive noble metal catalysts and hazardous high-pressure equipment. By utilizing common active metals such as zinc or iron in acidic media, the process achieves high yields and exceptional purity while operating under standard atmospheric conditions. This innovation represents a significant leap forward for manufacturers aiming to optimize their production lines for cost and safety without compromising on the stringent quality standards required for API intermediates.
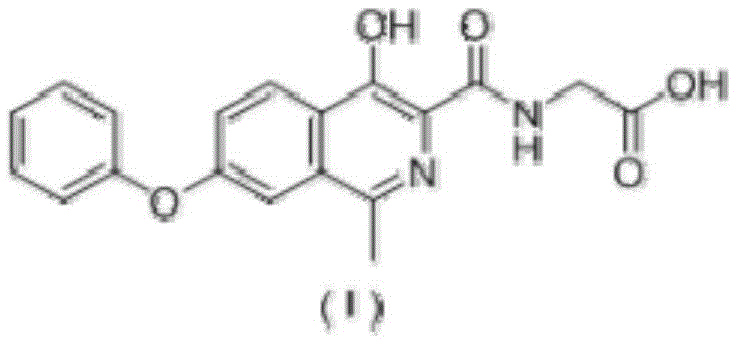
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing 1-methyl isoquinolinone derivatives often rely heavily on catalytic hydrogenation using palladium on carbon (Pd/C) under elevated pressures. As illustrated in comparative data from prior art, these conventional methods necessitate the use of sealed reaction vessels or autoclaves to maintain the required hydrogen pressure, which imposes severe constraints on equipment selection and operational safety. The reliance on noble metals not only drives up the raw material costs significantly but also introduces complex downstream processing challenges related to removing trace heavy metal residues to meet pharmacopeial limits. Furthermore, the harsh conditions often lead to the formation of various byproducts, complicating the purification process and resulting in lower overall yields that negatively impact the economic viability of large-scale manufacturing.
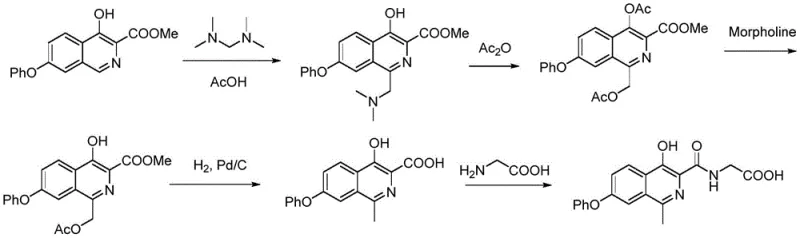
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a strategic aminomethylation followed by a reductive conversion using active metals like zinc or iron. This method cleverly bypasses the need for molecular hydrogen and high-pressure reactors by employing a chemical reduction strategy that proceeds smoothly at atmospheric pressure. The process involves converting a 1-aminomethyl intermediate into the desired 1-methyl product through a robust reduction step that is both operationally simple and highly effective. This shift allows for the use of standard industrial reactors, drastically lowering the barrier to entry for production and enhancing the safety profile of the facility. The ability to perform these transformations under mild thermal conditions further preserves the integrity of sensitive functional groups, ensuring a cleaner reaction profile and superior product quality.
Mechanistic Insights into Active Metal Catalytic Conversion
The core of this technological advancement lies in the reductive cleavage of the carbon-nitrogen bond in the aminomethyl side chain using active metals in an acidic environment. When the compound of Formula 3, which contains a dialkylaminomethyl group at the 1-position, is treated with zinc or iron powder in the presence of acids like acetic acid or hydrochloric acid, a single-electron transfer mechanism facilitates the removal of the amine moiety. This results in the formation of a stable methyl group at the 1-position of the isoquinolinone ring system. The choice of acid and metal is critical; for instance, zinc in acetic acid provides a balanced reduction potential that minimizes over-reduction of other sensitive parts of the molecule, such as the phenoxy ether linkage or the amide bond. This selectivity is paramount for maintaining the structural fidelity of the complex heterocyclic scaffold during the transformation.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based high-temperature processes. The controlled nature of the metal-acid reduction limits the generation of free radicals that typically lead to polymerization or ring-opening side reactions. Consequently, the crude reaction mixture contains significantly fewer impurities, simplifying the isolation of the final product. The patent data indicates that this method consistently achieves high purity levels, often exceeding 98% by HPLC, directly after standard workup procedures like filtration and crystallization. This inherent cleanliness reduces the burden on downstream purification units, such as chromatography columns, thereby streamlining the entire manufacturing workflow and reducing solvent consumption.
How to Synthesize Roxadustat Intermediate Efficiently
The synthesis of the target isoquinolinone compound is achieved through a streamlined three-step sequence that maximizes atom economy and operational simplicity. The process begins with the amidation of a methyl isoquinoline carboxylate with glycine, followed by an electrophilic substitution to install the aminomethyl handle, and concludes with the pivotal active metal reduction. This route is designed to be telescoped where possible, minimizing the number of isolation steps and reducing the overall processing time. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and conditions optimized for reproducibility and scale-up.
- React methyl 4-hydroxy-7-phenoxyisoquinoline-3-carboxylate with glycine or glycine ester in the presence of an organic base to form the amide intermediate.
- Perform an aminomethylation reaction on the amide intermediate using an aminal reagent under acid catalysis to introduce the aminomethyl group at the 1-position.
- Subject the aminomethylated intermediate to active metal catalytic conversion using zinc or iron powder in an acidic solution to obtain the final 1-methyl isoquinolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this atmospheric pressure synthesis route offers transformative benefits that extend far beyond simple yield improvements. By replacing expensive palladium catalysts with abundant and low-cost metals like zinc or iron, the direct material costs are substantially reduced, creating a more competitive pricing structure for the final intermediate. Additionally, the elimination of high-pressure equipment requirements means that production can be outsourced to a wider range of contract manufacturing organizations (CMOs) that possess standard reactor capabilities, thereby diversifying the supply base and mitigating the risk of production bottlenecks. This flexibility is crucial for maintaining supply continuity in a volatile global market.
- Cost Reduction in Manufacturing: The substitution of noble metal catalysts with base metals like zinc creates a profound impact on the bill of materials, effectively removing the volatility associated with precious metal pricing. Furthermore, the simplified workup procedure, which avoids complex heavy metal scavenging steps, reduces the consumption of auxiliary chemicals and lowers waste disposal costs. The overall process efficiency is enhanced by the ability to run reactions in standard vessels, avoiding the depreciation and maintenance costs associated with specialized high-pressure autoclaves.
- Enhanced Supply Chain Reliability: Operating under atmospheric pressure significantly de-risks the manufacturing process, as it removes the potential for catastrophic equipment failure associated with high-pressure hydrogenation. This safety improvement translates to fewer unplanned shutdowns and a more predictable production schedule. Moreover, the reagents used, such as glycine and common mineral acids, are commodity chemicals with robust global supply chains, ensuring that raw material availability remains stable even during market fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable because it relies on unit operations that are well-understood and easily replicated from pilot plant to commercial tonnage scales. The use of less toxic metals and the generation of simpler waste streams facilitate easier compliance with increasingly stringent environmental regulations. This green chemistry advantage not only reduces the regulatory burden but also aligns with the sustainability goals of modern pharmaceutical companies, making the supply chain more resilient to future regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel isoquinolinone synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production portfolios.
Q: What are the primary advantages of the active metal reduction method over traditional palladium catalysis?
A: The active metal reduction method eliminates the need for expensive noble metal catalysts like palladium on carbon, significantly reducing raw material costs and removing the complex heavy metal removal steps required for pharmaceutical grade purity. Furthermore, this process operates effectively under atmospheric pressure, removing the safety risks and equipment costs associated with high-pressure hydrogenation reactors.
Q: Can this synthesis route be scaled for industrial production without specialized pressure equipment?
A: Yes, the patent explicitly states that the reaction proceeds under normal atmospheric pressure, meaning it does not require sealed tubes or autoclaves. This allows for the use of standard glass-lined or stainless steel reactors commonly found in fine chemical facilities, greatly facilitating commercial scale-up and reducing capital expenditure.
Q: How does this method impact the purity profile of the final isoquinolinone compound?
A: By avoiding transition metal catalysts that can leave trace residues, the method simplifies the purification process. The examples in the patent demonstrate high HPLC purity levels exceeding 98% directly after workup, indicating a cleaner reaction profile with fewer side products compared to high-temperature or high-pressure alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technology described in CN112679431B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this atmospheric pressure reduction strategy, leveraging our rigorous QC labs and stringent purity specifications to deliver isoquinolinone intermediates that meet the highest international standards. Our commitment to process optimization ensures that we can offer this complex intermediate with consistent quality and reliability.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your supply chain efficiency and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization efforts, ensuring a seamless transition to this superior manufacturing platform.
