Advanced Manufacturing of Roxadustat Intermediate via Efficient Cyclization Technology
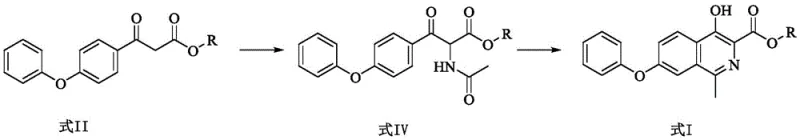
The pharmaceutical industry is constantly seeking more efficient pathways for the synthesis of critical therapeutic agents, particularly for treatments addressing chronic conditions like renal anemia. Patent CN111499572A introduces a groundbreaking preparation method for a key Roxadustat intermediate, specifically 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline formate. This innovation represents a significant leap forward in process chemistry, shifting away from the cumbersome, multi-step syntheses of the past towards a streamlined, high-yield approach. By utilizing 3-oxo-3-(4-phenoxyphenyl) propionate as a strategic starting material, the disclosed method achieves the target molecule through a concise sequence involving nitrosation, acetylation, and a robust cyclization reaction. This technical advancement not only promises superior purity profiles essential for regulatory compliance but also addresses the urgent market demand for cost-effective and environmentally sustainable manufacturing processes in the realm of hypoxia-inducible factor prolyl hydroxylase inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roxadustat and its precursors has been plagued by significant inefficiencies that hinder large-scale commercial viability. Early routes, such as those described in world patent WO2004108681, relied on 3,4-dicyanonitrobenzene as a starting material, necessitating an arduous 11-step sequence that included hazardous reagents and complex purification stages. Furthermore, subsequent optimizations like those in WO2013013609 attempted to shorten the route but still depended heavily on expensive palladium catalysts for methylation steps, which notoriously suffered from low yields of approximately 33%. These conventional methodologies impose severe bottlenecks on supply chains, as the reliance on precious metals increases raw material costs and introduces risks associated with heavy metal residue removal. Additionally, the use of high-risk reagents and the need for multiple protection and deprotection cycles in alternative routes create substantial waste streams, making them less attractive from both an economic and an environmental compliance perspective for modern pharmaceutical manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN111499572A offers a radically simplified synthetic strategy that bypasses the need for precious metal catalysis and excessive step counts. The core innovation lies in the construction of the isoquinoline skeleton directly from a functionalized ketone ester via a cleverly designed cyclization cascade. This route leverages the inherent reactivity of the 3-oxo-3-(4-phenoxyphenyl) propionate backbone, allowing for the introduction of the necessary nitrogen functionality through a mild nitrosation followed by acetylation. The subsequent ring closure is achieved efficiently using phosphorus oxychloride, a reagent that is both cost-effective and widely available in the fine chemical industry. By eliminating the palladium-catalyzed methylation step entirely, this new pathway drastically reduces the complexity of the impurity profile and enhances the overall atom economy. The result is a process that is not only safer and more environmentally friendly but also inherently more scalable, providing a reliable foundation for the consistent supply of high-quality pharmaceutical intermediates.
Mechanistic Insights into POCl3-Mediated Cyclization
The heart of this synthetic breakthrough is the mechanistic elegance of the cyclization step, which transforms the linear acetamido-keto ester into the rigid isoquinoline structure. The process begins with the formation of an imidoyl chloride species through the reaction of the amide carbonyl with phosphorus oxychloride (POCl3). This activation is crucial as it renders the carbon center highly electrophilic, facilitating an intramolecular nucleophilic attack by the electron-rich aromatic ring. The presence of the phenoxy group at the 7-position plays a subtle yet vital role in directing the regioselectivity of this electrophilic aromatic substitution, ensuring that the ring closure occurs at the correct position to form the desired 4-hydroxy-1-methyl-7-phenoxy scaffold. The reaction conditions, typically involving reflux in polar aprotic solvents like DMF or non-polar solvents like toluene, provide the necessary thermal energy to overcome the activation barrier while maintaining the stability of the sensitive ester functionality. This precise control over the cyclization mechanism is what allows the process to achieve yields exceeding 90% in many embodiments, a stark improvement over the erratic outcomes often seen in traditional isoquinoline syntheses.
Furthermore, the upstream preparation of the cyclization precursor involves a highly controlled nitrosation and reduction sequence that ensures minimal formation of side products. The initial reaction with sodium nitrite in an acidic medium generates a hydroxylamino intermediate, which is subsequently converted to the acetamido derivative. This two-step functionalization is critical because it installs the nitrogen atom in the exact oxidation state required for the final ring closure without needing harsh oxidizing or reducing conditions that could degrade the sensitive ketone moiety. The use of metal powders like zinc or magnesium during the acetylation phase serves a dual purpose: it acts as a reducing agent to manage the oxidation state and potentially scavenges any trace oxidants that could lead to impurity formation. This meticulous attention to the redox balance throughout the synthesis ensures that the final crude product possesses a high degree of chemical purity, significantly reducing the burden on downstream purification units and crystallization steps.
How to Synthesize Roxadustat Intermediate Efficiently
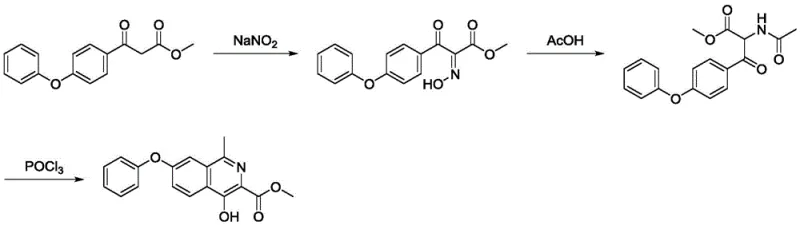
Implementing this advanced synthesis route requires a clear understanding of the operational parameters that drive high yield and purity. The process is designed to be robust, utilizing common laboratory and plant equipment without the need for specialized high-pressure reactors often associated with hydrogenation steps in other routes. The following guide outlines the critical phases of the synthesis, emphasizing the transition from the keto-ester starting material to the final isoquinoline product. Operators should pay close attention to temperature control during the nitrosation phase to prevent diazotization side reactions, and ensure thorough mixing during the POCl3 addition to manage exotherms effectively. For a comprehensive breakdown of the standardized operating procedures, including specific molar ratios and workup protocols, please refer to the detailed technical guide below.
- Perform sodium nitrite hydroxylamination on 3-oxo-3-(4-phenoxyphenyl) propionate in glacial acetic acid and water at 0°C to form the hydroxylamino intermediate.
- Convert the hydroxylamino intermediate to 2-acetamido-3-oxo-3-(4-phenoxyphenyl) propionate via acetylation using acetic anhydride and metal powder reduction.
- Execute the final cyclization reaction using phosphorus oxychloride (POCl3) in a solvent like toluene or DMF under reflux to obtain the target isoquinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic advantages that go beyond simple unit price reductions. The primary benefit lies in the drastic simplification of the supply chain for raw materials; by avoiding the need for custom-synthesized, multi-step starting materials like dicyanonitrobenzene derivatives, manufacturers can source bulk commodities that are stable and widely available. This shift significantly mitigates the risk of supply disruptions caused by the bottleneck of a single specialized vendor. Moreover, the elimination of palladium catalysts removes a major cost volatility factor, as precious metal prices can fluctuate wildly, impacting the final cost of goods sold. The process's inherent safety profile, characterized by mild reaction temperatures and the absence of high-pressure hydrogenation, also lowers insurance premiums and facility maintenance costs, contributing to a more resilient and cost-efficient manufacturing operation overall.
- Cost Reduction in Manufacturing: The economic impact of this route is profound, primarily driven by the removal of expensive catalytic systems and the reduction of total processing time. By bypassing the palladium-catalyzed methylation step, which historically suffered from poor yields and required costly metal recovery processes, the new method achieves a much higher throughput per batch. The use of inexpensive reagents like sodium nitrite, acetic anhydride, and phosphorus oxychloride further drives down the variable cost of production. Additionally, the high purity of the crude product means that solvent consumption for recrystallization and chromatography is significantly reduced, leading to lower waste disposal costs and improved overall margin potential for the final API.
- Enhanced Supply Chain Reliability: From a logistics perspective, this synthesis route offers superior stability and predictability. The starting material, 3-oxo-3-(4-phenoxyphenyl) propionate, is chemically stable and can be stockpiled without significant degradation, allowing manufacturers to build strategic inventory buffers against market fluctuations. The robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supply or operator technique, resulting in more consistent batch-to-batch quality. This reliability is crucial for maintaining uninterrupted supply to downstream API manufacturers, ensuring that clinical trials and commercial launches are not delayed due to intermediate shortages, thereby strengthening the partnership between intermediate suppliers and pharmaceutical developers.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the absence of complex unit operations like high-pressure hydrogenation or cryogenic reactions. The reactions proceed well in standard glass-lined or stainless steel reactors, facilitating rapid technology transfer. Environmentally, the process aligns with green chemistry principles by minimizing the use of toxic heavy metals and reducing the E-factor (mass of waste per mass of product). The aqueous workups and the ability to recover and recycle solvents like toluene or DMF further enhance the sustainability profile, helping companies meet increasingly stringent global environmental regulations and corporate social responsibility goals without compromising on production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Roxadustat intermediate synthesis. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic benefits of the technology. Understanding these details is essential for technical teams evaluating the route for potential licensing or contract manufacturing opportunities.
Q: How does this new synthesis route improve upon conventional methods for Roxadustat intermediates?
A: Unlike conventional routes that rely on expensive palladium-catalyzed methylation with low yields (around 33%) or lengthy 11-step sequences, this novel method utilizes a direct cyclization strategy from readily available ketone esters. This significantly reduces step count, eliminates precious metal catalysts, and improves overall process safety and yield.
Q: What are the key reaction conditions for the cyclization step?
A: The critical cyclization step employs phosphorus oxychloride (POCl3) as the dehydrating agent and cyclization promoter. The reaction is typically conducted in solvents such as toluene, xylene, or DMF under reflux conditions, ensuring high conversion rates and excellent purity of the final isoquinoline product without requiring extreme pressures.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It avoids high-risk reagents and operates under mild temperatures (often between -10°C to 30°C for early steps). The use of common solvents and the elimination of complex protection/deprotection sequences make it highly robust for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving drugs like Roxadustat depends on a supply chain that is both agile and technically卓越. Our team of expert chemists has extensively analyzed the CN111499572A patent and possesses the capability to optimize this route for maximum efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from development to market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline formate meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable, high-quality supply of this critical intermediate for your global operations.
