Optimized Synthetic Pathway for Ticagrelor Intermediates Driving Commercial Scalability and Purity
Optimized Synthetic Pathway for Ticagrelor Intermediates Driving Commercial Scalability and Purity
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, with a persistent demand for more efficient and environmentally sustainable manufacturing processes for critical active pharmaceutical ingredients (APIs). Patent CN103288837A discloses a novel preparation method for Ticagrelor, a potent P2Y12 receptor antagonist, which represents a significant departure from legacy synthetic strategies. This innovation focuses on constructing the core triazolo-pyrimidine scaffold through a streamlined cyclization of 5-amino-1,4-disubstituted-1,2,3-triazole derivatives, thereby bypassing several cumbersome steps found in earlier literature. By prioritizing green chemistry principles, this route not only enhances the chemical and chiral purity of the final product but also mitigates the safety risks associated with traditional halogenation reagents. For global stakeholders seeking a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for securing a robust supply chain.
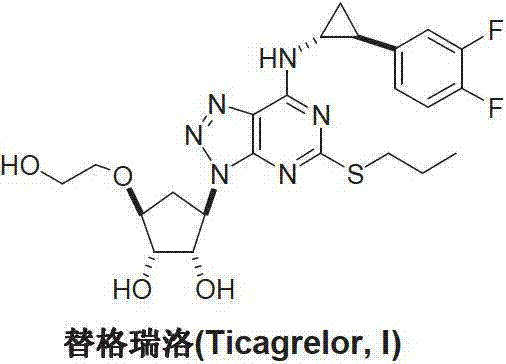
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Ticagrelor, as documented in various international patents such as WO97/03084 and WO2012/138981, often rely on complex sequences involving early-stage introduction of functional groups that require extensive protection and deprotection cycles. A major bottleneck in these conventional methodologies is the frequent utilization of aggressive chlorinating agents, specifically phosphorus oxychloride (POCl3), to activate pyrimidine rings for subsequent nucleophilic substitutions. This reliance introduces significant operational hazards, generates corrosive waste streams that complicate environmental compliance, and often leads to the formation of difficult-to-remove chlorinated impurities that compromise the purity profile of the API. Furthermore, the necessity to protect hydroxyl groups on the cyclopentyl ring early in the synthesis adds unnecessary molecular weight and step count, reducing overall atom economy and increasing production costs.
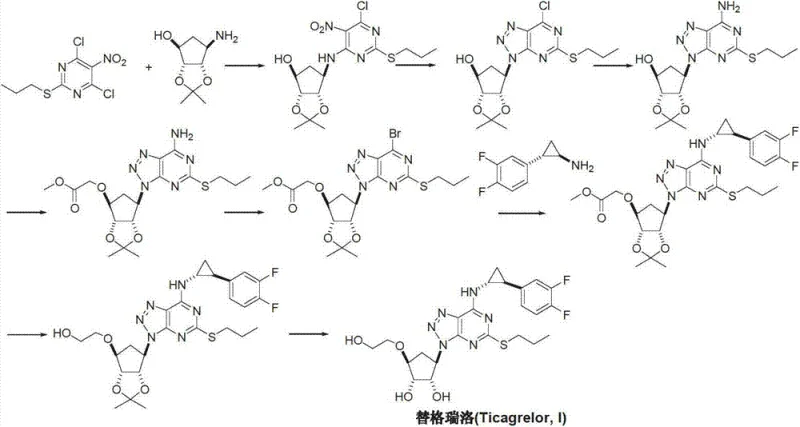
The Novel Approach
In stark contrast, the methodology outlined in CN103288837A proposes a convergent strategy that constructs the heterocyclic core with greater precision and fewer hazardous inputs. The process initiates with a cyclization reaction between a substituted triazole and a sulfur-containing agent, directly forming the 8-azaguanine skeleton without the need for initial harsh activation. This is followed by a controlled substitution with halogenated propane to install the propylthiol moiety, and finally, a condensation with the chiral cyclopropylamine. This sequence effectively eliminates the direct chlorination step found in older patents, thereby reducing the impurity burden and simplifying the purification workflow. The result is a more linear and predictable synthesis that aligns perfectly with the requirements for cost reduction in pharmaceutical intermediates manufacturing by minimizing reagent costs and waste disposal fees.
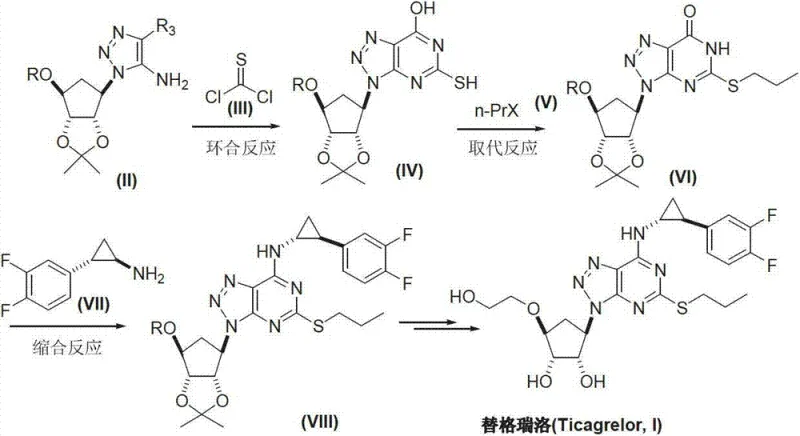
Mechanistic Insights into Triazole-Based Cyclization and Substitution
The cornerstone of this improved synthesis lies in the efficient cyclization of the 5-amino-1,4-disubstituted-1,2,3-triazole precursor. In this mechanism, the amino group acts as a nucleophile attacking the sulfur-containing cyclizing agent, such as thiophosgene or isothiocyanates, under basic conditions. This reaction forms the six-membered ring fused to the existing triazole, creating the stable triazolo[4,5-d]pyrimidine system. The choice of base, ranging from potassium carbonate to organic amines like DBU, allows for fine-tuning the reaction kinetics to maximize yield while suppressing side reactions. This specific cyclization pathway is advantageous because it avoids the high-energy intermediates typically associated with nitro-reduction pathways found in other patents, leading to a cleaner reaction profile and higher stereochemical integrity of the adjacent chiral centers.
Following the core formation, the substitution of the sulfo-group with a propylthiol chain via halogenated propane is a critical step for establishing the pharmacophore's lipophilicity. The mechanism involves a nucleophilic attack by the sulfur anion on the alkyl halide, facilitated by mild acid binders. This step is particularly robust, tolerating a variety of solvent systems including acetonitrile and dichloromethane. The subsequent condensation with the chiral amine is mediated by coupling agents like BOP or HBTU, ensuring high fidelity in the formation of the C-N bond at the 6-position. This mechanistic control is vital for maintaining the high-purity pharmaceutical intermediates required for regulatory approval, as it minimizes the formation of regioisomers that are common in less selective coupling reactions.
How to Synthesize Ticagrelor Efficiently
The execution of this synthesis requires precise control over reaction parameters to ensure the successful transformation of the triazole precursor into the final API. The process begins with the careful addition of the sulfur-containing agent to the triazole derivative at low temperatures to manage exothermicity, followed by a room temperature stir to complete the ring closure. Subsequent steps involve standard workup procedures such as extraction and crystallization, which are well-suited for industrial equipment. The final deprotection of the acetonide group is achieved under mild acidic conditions, preserving the sensitive glycol functionality. For a detailed breakdown of the specific molar ratios, temperature profiles, and isolation techniques, please refer to the standardized protocol below.
- Perform a cyclization reaction between 5-amino-1,4-disubstituted-1,2,3-triazole and a sulfur-containing agent to form the 8-azaguanine core.
- Execute a nucleophilic substitution using halogenated propane to introduce the propylthiol group at the 2-position.
- Condense the intermediate with trans-(1R,2S)-2-(3,4-difluorophenyl)cyclopropylamine to install the chiral side chain.
- Remove the acetonide protecting group under acidic conditions to yield the final Ticagrelor API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical elegance. By restructuring the synthesis to avoid hazardous chlorinating agents and complex protection groups, the process inherently reduces the dependency on specialized corrosion-resistant equipment and expensive waste treatment protocols. This simplification translates directly into a more resilient supply chain, as the raw materials required—such as substituted triazoles and simple alkyl halides—are commodity chemicals with stable availability. Consequently, manufacturers can achieve commercial scale-up of complex pharmaceutical intermediates with greater confidence, knowing that the process is less susceptible to disruptions caused by the scarcity of exotic reagents or stringent environmental regulations regarding toxic effluent.
- Cost Reduction in Manufacturing: The elimination of phosphorus oxychloride and the reduction in protection-deprotection steps significantly lower the operational expenditure associated with reagent procurement and waste management. Without the need for extensive neutralization of acidic byproducts or the removal of heavy metal catalysts often used in alternative cross-coupling methods, the downstream processing becomes markedly more economical. This efficiency allows for a substantial reduction in the cost of goods sold (COGS), providing a competitive edge in the global market for cardiovascular drugs while maintaining high margin potential for suppliers.
- Enhanced Supply Chain Reliability: The streamlined nature of this four-step sequence reduces the overall lead time required for batch production compared to longer, multi-step legacy routes. Fewer unit operations mean fewer opportunities for batch failure or quality deviations, ensuring a consistent output of material that meets strict specifications. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to maintain leaner inventory levels and respond more agilely to fluctuations in market demand for Ticagrelor formulations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior due to its alignment with green chemistry principles. The avoidance of highly toxic reagents and the use of recyclable solvents like acetonitrile facilitate easier regulatory approval for new manufacturing sites. This environmental compatibility ensures long-term operational continuity, as facilities adopting this method are better positioned to meet increasingly stringent global emissions standards, thereby securing the supply continuity essential for long-term commercial contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this route compares to established industry standards. Understanding these nuances is essential for technical teams evaluating process transfer feasibility.
Q: How does this new synthesis route improve upon conventional Ticagrelor manufacturing methods?
A: Unlike traditional routes that often rely on harsh chlorinating agents like phosphorus oxychloride (POCl3) and complex multi-step protection strategies, this patented method utilizes a direct cyclization of triazole derivatives. This significantly simplifies the workflow, reduces hazardous waste generation, and improves overall chemical purity by minimizing side reactions associated with aggressive chlorination.
Q: What are the key intermediates involved in this specific preparation method?
A: The process centers on three critical intermediates: first, the formation of 9-substituted-2-sulfo-6-oxo-8-azaguanine via cyclization; second, the conversion to 9-substituted-2-propylthiol-6-oxo-8-azaguanine through substitution; and third, the condensation product containing the cyclopropylamine side chain before final deprotection.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. By avoiding dangerous reagents and utilizing robust reaction conditions such as mild alkaline promoters and standard organic solvents, the process offers enhanced safety profiles and easier downstream processing, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Supplier
As the demand for next-generation antiplatelet therapies grows, the ability to source high-quality intermediates through innovative synthetic routes becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in heterocyclic chemistry to deliver solutions that balance performance with sustainability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Ticagrelor intermediate meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for this critical cardiovascular medication. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our advanced manufacturing capabilities can support your long-term strategic goals in the pharmaceutical sector.
